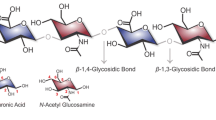
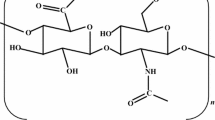
Abstracts
Hyaluronic acid (HA) is a commercially valuable medical biopolymer increasingly produced through microbial fermentation. Viscosity limits product yield and the focus of research and development has been on improving the key quality parameters, purity and molecular weight. Traditional strain and process optimisation has yielded significant improvements, but appears to have reached a limit. Metabolic engineering is providing new opportunities and HA produced in a heterologous host is about to enter the market. In order to realise the full potential of metabolic engineering, however, greater understanding of the mechanisms underlying chain termination is required.






Similar content being viewed by others
References
Abbe K, Takahashi S, Yamada T (1982) Involvement of oxygen-sensitive pyruvate-formate-lyase in mixed acid fermentation by Streptococcus mutans under strictly anaerobic conditions. J Bacteriol 152:175–182
Armstrong DC (1997) The molecular weight properties of hyaluronic acid produced Streptococcus zooepidemicus. PhD. Thesis, University of Queensland
Armstrong DC, Johns MR (1997) Culture conditions affect the molecular weight properties of hyaluronic acid produced by Streptococcus zooepidemicus. Appl Environ Microbiol 63:2759–2764
Asari A, Miyauchi S (2000) Medical applications of Hyaluronan. Glycoforum - Hyaluronan Today. http://www.glycoforum.gr.jp/science/hyaluronan/HAIZ/HAIZE.html
Ashbaugh CD, Alberti S, Wessels MR (1998) Molecular analysis of the capsule gene region of group A streptococcus: the hasAB genes are sufficient for capsule expression. J Bacteriol 180:4955–4959
Balazs EA, Leshchiner E, Larsen NE, Band P (1993) Application of hyaluronan and its derivatives. In: Gebelein CG (ed) Biotechnological polymers. Technomic, Lancaster, pp 41–65
Cleary PP, Larkin A (1979) Hyaluronic acid capsule: strategy for oxygen resistance in group A streptococci. J Bacteriol 140:1090–1097
Cooney MJ, Goh LT, Lee PL, Johns MR (1999) Structured model-based analysis and control of the hyaluronic acid fermentation by Streptococcus zooepidemicus: Physiological implications of glucose and complex nitrogen-limited growth. Biotechnol Prog 15:898–910
Crater DL, Dougherty BA, van de Rijn I (1995) Molecular characterization of hasC from an operon required for hyaluronic acid synthesis in group A streptococci. Demonstration of UDP- glucose pyrophosphorylase activity. J Biol Chem 270:28676–28680
Cywes C, Wessels MR (2001) Group A Streptococcus tissue invasion by CD44-mediated cell signalling. Nature 414:648–652
DeAngelis PL, Papaconstantinou J, Weigel PH (1993a) Isolation of a Streptococcus-Pyogenes gene locus that directs hyaluronan biosynthesis in acapsular mutants and in heterologous bacteria. J Biol Chem 268:14568–14571
DeAngelis PL, Papaconstantinou J, Weigel PH (1993b) Molecular cloning, identification, and sequence of the hyaluronan synthase gene from group A Streptococcus pyogenes. J Biol Chem 268:19181–19184
DeAngelis PL, Weigel PH (1994) Immunochemical confirmation of the primary structure of streptococcal hyaluronan synthase and synthesis of high molecular weight product by the recombinant enzyme. Biochemistry 33:9033–9039
de Ruyter P, Kuipers OP, de Vos WM (1996) Controlled gene expression systems for Lactococcus lactis with the food-grade inducer nisin. Appl Environ Microbiol 62:3662–3667
Dougherty BA, van de Rijn I (1993) Molecular characterization of hasB from an operon required for hyaluronic acid synthesis in group A streptococci. Demonstration of UDP- glucose dehydrogenase activity. J Biol Chem 268:7118–7124
Dougherty BA, van de Rijn I (1994) Molecular characterization of hasA from an operon required for hyaluronic acid synthesis in group A streptococci. J Biol Chem 269:169–175
Ellwood DC, Evans CGT, Dunn GM, McInnes N et al (1995) Production of hyaluronic acid. US Patent 5411874
Ellwood DC, Evans CGT, Dunn GM, McInnes N et al (1996) Production of hyaluronic acid. US Patent 5411874
Fong Chong B (2002) Improving the cellular economy of Streptococcus zooepidemicus through metabolic engineering. PhD Thesis, The University of Queensland
Fong Chong B, Nielsen LK (2003a) Aerobic cultivation of Streptococcus zooepidemicus and the role of NADH oxidase. Biochem Eng J 16:153–162
Fong Chong B, Nielsen LK (2003b) Amplifying the cellular reduction potential of Streptococcus zooepidemicus. J Biotechnol 100:33–41
Forsee WT, Cartee RT, Yother J (2000) Biosynthesis of type 3 capsular polysaccharide in Streptococcus pneumoniae—enzymatic chain release by an abortive translocation process. J Biol Chem 275:25972–25978
Fouissac E, Milas M, Rinaudo M (1993) Shear rate, concentration, molecular weight and temperature viscosity dependences of hyaluronate, a wormlike polyelectrolytes. Macromolecules 26:6945–6951
Goh L-T (1998) Effect of culture conditions on rates of intrinsic hyaluronic acid production by Streptococcusequi subsp. zooepidemicus. PhD Thesis. University of Queensland
Heldermon C, DeAngelis PL, Weigel PH (2001) Topological organization of the hyaluronan synthase from Streptococcus pyogenes. J Biol Chem 276:2037–2046
Heldermon C, Kumari K, Tlapak-Simmons V, Weigel PH (2000) Streptococcal hyaluronan synthases and the synthesis of “designer” hyaluronan. In: Abatangelo G, Weigel PH (eds) New frontiers in medical sciences: redefining hyaluronan. Elsevier, New York
Jacques M, Graham L (1989) Improved preservation of bacterial capsule for electron microscopy. J Electron Microsc Tech 11:167–169
Johns MR, Goh LT, Oeggerli A (1994) Effect of pH, agitation and aeration on hyaluronic-acid production by Streptococcus-Zooepidemicus. Biotechnol Lett 16:507–512
Kim JH, Yoo SJ, Oh DK, Kweon YG et al. (1996) Selection of a Streptococcus equi mutant and optimization of culture conditions for the production of high molecular weight hyaluronic acid. Enzyme Microb Technol 19:440–445
Kitchen JR, Cysyk RL (1995) Synthesis and release of hyaluronic acid by Swiss 3T3 fibroblasts. Biochem J 309:649–656
Kumari K, Weigel PH (1997) Molecular cloning, expression, and characterization of the authentic hyaluronan synthase from group C Streptococcus equisimilis. J Biol Chem 272:32539–32546
Lander ES, Linton LM, Birren B, Nusbaum C et al. (2001) Initial sequencing and analysis of the human genome. Nature 409:860–921
Leonard BA, Woischnik M, Podbielski A (1998) Production of stabilized virulence factor-negative variants by group A streptococci during stationary phase. Infect Immun 66:3841–3847
Lertwerawat Y (1993) Hyaluronic acid production and its instability in Streptococcus zooepidemicus. PhD Thesis. University of Queensland
Maguin E, Prevost H, Ehrlich SD, Gruss A (1996) Efficient insertional mutagenesis in lactococci and other Gram-positive bacteria. J Bacteriol 178:931–935
Matsubara C, Kajiwara M, Akasaka H, Haze S (1991) Carbon-13 nuclear magnetic resonance studies on the biosynthesis of hyaluronic acid. Chem Pharm Bull 39:2446–2448
Mausolf A, Jungmann J, Robenek H, Prehm P (1990) Shedding of hyaluronate synthase from streptococci. Biochem J 267:191–196
Meyer K, Palmer JW (1934) The polysaccharide of the vitreous humor. J Biol Chem 107:629–634
O’Regan M, Martini I, Crescenzi F, De Luca C et al (1994) Molecular Mechanisms and genetics of hyaluron Biosynthesis. Int J Biol Macromol 16:283–286
Praest BM, Helmut G, Rudiger K (1997) Effects of oxygen-derived free radicals on the molecular weight and the polydispersity of hyaluronan solutions. Carbohydr Res 303:153–157
Prehm P (1984) Hyaluronate is synthesised at plasma membrane. Biochem J 220:597–600
Saettone MF, Giannaccini B, Chetoni P, Torracca MT et al (1991) Evaluation of high-molecular-weight and low-molecular-weight fractions of sodium hyaluronate and an ionic complex as adjuvants for topical ophthalmic vehicles containing pilocarpine. Int J Pharm 72:131–139
Sakamoto M, Komagata K (1996) Aerobic growth of and activities of NADH oxidase and NADH peroxidase in lactic acid bacteria. J Ferment Bioeng 82:210–216
Salzberg SL, White O, Peterson J, Eisen JA (2001) Microbial genes in the human genome: lateral transfer or gene loss? Science 292:1903–1906
Saso L, Bonanni G, Grippa E, Gatto MT et al (1999) Interaction of hyaluronic acid with mucin, evaluated by gel permeation chromatography. Res Commun Mol Pathol Pharmacol 104:277–284
Schmidt KH, Gunther E, Courtney HS (1996) Expression of both M protein and hyaluronic acid capsule by group A streptococcal strains results in a high virulence for chicken embryos. Med Microbiol Immunol 184:169–173
Scott JE, Heatley F (1999) Hyaluronan forms specific stable tertiary structures in aqueous solution: a C-13 NMR study. Proc Natl Acad Sci USA 96:4850–4855
Snoep JL, de Graef MR, de Mattos MJT, Neijssel OM (1992) Pyruvate catabolism during transient state conditions in chemostat cultures of Enterococcus faecalis NCTC 775: importance of internal pyruvate concentrations and NADH/NAD+ ratios. J Gen Microbiol 138:2015–2020
Spurlock SL, Spurlock GH, Bernstad S, Michanek P et al. (1999) Treatment of acute superficial flexor tendon injuries in performance horses with high molecular weight sodium hyaluronate. J Equine Vet Sci 19:338–344
Stangohl S (2000) Methods and means for the production of hyaluronic acid. US Patent 6090596
Stangohl S (2003) Methods and means for the production of hyaluronic acid. US Patent 6537795
Suzuki Y, Yamaguchi T (1993) Effect of hyaluronic acid on macrophage phagacytosis and active oxygen release. Agents Actions 38:32–37
Thomas EL, Pera KA (1983) Oxygen metabolism of Streptococcus mutans: uptake of oxygen and release of superoxide and hydrogen peroxide. J Bacteriol 154:1236–1244
Thomas TD, Ellwood DC, Longyear VMC (1979) Change from homo- to heterolactic fermentation by Streptococcus lactis resulting from glucose limitation in anaerobic chemostat cultures. J Bacteriol 138:109–117
Tlapak-Simmons VL, Baggenstoss BA, Clyne T, Weigel PH (1999a) Purification and lipid dependence of the recombinant hyaluronan synthases from Streptococcus pyogenes and Streptococcus equisimilis. J Biol Chem 274:4239–4245
Tlapak-Simmons VL, Baggenstoss BA, Kumari K, Heldermon C et al. (1999b) Kinetic characterization of the recombinant hyaluronan synthases from Streptococcus pyogenes and Streptococcus equisimilis. J Biol Chem 274:4246–4253
Tlapak-Simmons VL, Kempner ES, Baggenstoss BA, Weigel PH (1998) The active streptococcal hyaluronan synthases (HASs) contain a single HAS monomer and multiple cardiolipin molecules. J Biol Chem 273:26100–26109
van de Rijn I, Kessler RE (1980) Growth characteristics of group A streptococci in a new chemically defined medium. Infect Immun 27:444–448
Ward PN, Field TR, Ditcham WGF, Maguin E et al (2001) Identification and disruption of two discrete loci encoding hyaluronic acid capsule biosynthesis genes hasA, hasB, and hasC in Streptococcus uberis. Infect Immun 69:392–399
Weigel PH (2002) Functional characteristics and catalytic mechanisms of the bacterial hyaluronan synthases. IUBMB Life 54:201–211
Weigel PH, Hascall VC, Tammi M (1997) Hyaluronan synthases. J Biol Chem 272:13997–14000
Weissman B, Meyer K (1954) The structure of hyalobiuronic acid and of hyaluronic acid from umbilical cord. J Am Chem Soc 76:1753–1757
Wessels MR, Moses A, Goldberg JB, DiCesare TJ (1991) Hyaluronic acid capsule is a virulence factor for mucoid group A streptococci. Proc Natl Acad Sci USA 88:8317–8321
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chong, B.F., Blank, L.M., Mclaughlin, R. et al. Microbial hyaluronic acid production. Appl Microbiol Biotechnol 66, 341–351 (2005). https://doi.org/10.1007/s00253-004-1774-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-004-1774-4