Abstract
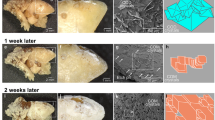
The in vitro study of calcium oxalate (CaOx) stone formation is usually based on crystallisation models but it is recognised that both healthy individuals and stone formers have crystalluria. We have established a robust in vitro stone growth model based on the principle of mixed suspension, mixed product removal system (MSMPR). Utilising this technique we studied the influence of CaOx crystallisation kinetics and the variation of calcium and oxalate concentrations on CaOx stone growth in vitro. Six stones received standard concentration of Ca (6 mM) and Ox (1.2 mM) in the medium while another six received variable concentrations of both Ca and Ox at various intervals. Stone mass was plotted against the experiment duration (typically 5–7 weeks). The stone growth was dependent on sufficient input calcium and oxalate concentrations and once triggered, stone growth could not be maintained at reduced calcium and oxalate inputs. The stone growth rate was positively correlated to the number of crystals in suspension around the stone and to the crystal nucleation rate and negatively correlated to the crystal growth rates. This leads to the conclusion that aggregation of crystals from the surrounding suspension was the dominant mechanism for stone enlargement.



Similar content being viewed by others
References
Kavanagh JP (2001) A critical appraisal of the hypothesis that urine is a saturated equilibrium with respect to stone-forming calcium salts. BJU Int 87:589–598
Ananth K, Kavanagh JP, Walton RC, Rao PN (2002) Enlargement of calcium oxalate stones to clinically significant size in an in-vitro stone generator. BJU Int 90:939–944
Chow K, Dixon J, Gilpin S, Kavanagh JP, Rao PN (2004) A stone farm: development of a method for simultaneous production of multiple calcium oxalate stones in vitro. Urol Res 32:55–60
Chow K, Dixon J, Gilpin S, Kavanagh JP, Rao PN (2004) Citrate inhibits growth of residual fragments in an in vitro model of calcium oxalate renal stones. Kidney Int 65:1724–1730
Vermeulen CW (1972) Calculogenesis and stone triggering. National Academy of Sciences, Washington
Finlayson B (1972) The concept of a continuous crystalliser. Its theory and application to in vivo and in vitro urinary tract models. Invest Urol 9:258–263
Kavanagh JP, Rao PN (2007) Lessons from a stone farm. AIP Conf Proc 900:159–169
Kavanagh JP, Jones L, Rao PN (1999) Calcium oxalate crystallization kinetics at different concentrations of human and artificial urine, with a constant calcium to oxalate ratio. Urol Res 27:231–237
Marengo S, Zhang A (2007) It pays to read the old dead guy’s papers:triggering doses of oxalate and calcium oxalte nephrocalcinosis. AIP Conf Proc 900:398–401
Kavanagh JP (1999) Enlargement of a lower pole calcium oxalate stone: A theoretical examination of the role of crystal nucleation, growth, and aggregation. J Endourol 13:605–610
Grases F, Costa-Bauza A, Garcia-Ferragut L (1998) Biopathological crystallization: a general view about the mechanisms of renal stone formation. Adv Colloid Interface Sci 74:169–194
Khan SR, Hackett RL (1993) Role of organic matrix in urinary stone formation—an ultrastructural-study of crystal matrix interface of calcium-oxalate monohydrate stones. J Urol 150:239–245
Daudon M, Bader CA, Jungers P (1993) Urinary calculi—review of classification methods and correlations with etiology. Scanning Microsc 7:1081–1106
Millan A (1997) Crystal morphology and texture in calcium oxalate monohydrate renal calculi. J Mater Sci Mater Med 8:247–250
Webber D, Chauvet MC, Ryall RL (2005) Proteolysis and partial dissolution of calcium oxalate: a comparative, morphological study of urinary crystals from black and white subjects. Urol Res 33:273–284
Fleming DE, Van Riessen A, Chauvet MC, Grover PK, Hunter B, Van Bronswijk W, Ryall RL (2003) Intracrystalline proteins and urolithiasis: a synchrotron X-ray diffraction study of calcium oxalate monohydrate. J Bone Miner Res 18:1282–1291
Niederberger M, Colfen H (2006) Oriented attachment and mesocrystals: Non-classical crystallization mechanisms based on nanoparticle assembly. Phy Chem Chem Phy 8:3271–3287
Hounslow MJ, Mumtaz HS, Collier AP, Barrick JP, Bramley AS (2001) A micro-mechanical model for the rate of aggregation during precipitation from solution. Chem Eng Sci 56:2543–2552
David R, Espitalier F, Cameirao A, Rouleau L (2001) Developments in the understanding and modeling of the agglomeration of suspended crystals in crystallization from solution. KONA 21:40–53
Sokol E, Nigmatulina E, Maksimova N, Chiglintsev A (2005) CaC2O4.H2O spherulites in human kidney stones: morphology, chemical composition, and growth regime. Eur J Mineral 17:285–295
Grases F, Costa-Bauza A, Ramis M, Montesinos V, Conte A (2002) Simple classification of renal calculi closely related to their micromorphology and etiology. Clin Chim Acta 322:29–36
Finlayson B, Reid F (1978) Expectation of free and fixed particles in urinary stone disease. Invest Urol 15:442–448
Kok DJ, Khan SR (1994) Calcium-oxalate nephrolithiasis, a free or fixed particle disease. Kidney Int 46:847–854
Acknowledgments
These studies were funded by Kidney Research UK and The Oxalosis and Hyperoxaluria Foundation. We also thank S. Gilpin and J. Dixon for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saw, N.K., Rao, P.N. & Kavanagh, J.P. A nidus, crystalluria and aggregation: key ingredients for stone enlargement. Urol Res 36, 11–15 (2008). https://doi.org/10.1007/s00240-007-0121-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00240-007-0121-5