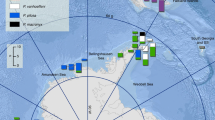
Abstract
Sea spiders are conspicuous, and often abundant, members of the Antarctic benthic community. Nymphonidae (Pycnogonida) in Southern Ocean waters comprise over 240 species which are often difficult to assign due to their intraspecific ‘highly variable’ morphology. In particular, Nymphon australe, the numerically dominant species in Antarctic waters is known to have a high level of phenotypic variation in external morphology and is also reported to have a circumpolar distribution. Circumpolarity seems contradictory to the pycnogonid’s brooding lifestyle and presumably limited dispersal. Here we examine the genetic diversity of several Nymphon species collected in the Antarctic Peninsular region. Concomitantly, we assess the genetic structure of N. australe to gain insight into Nymphon dispersal capacity. Cytochrome c oxidase subunit I (COI) and 16S ribosomal gene data suggest a recent common history and/or recent gene-flow of N. australe populations across nearly 800 km of the Antarctic Peninsula. Furthermore, these data support that the Antarctic Peninsula region may hold two previously unrecognized species of Nymphon.



Similar content being viewed by others
References
Ament AS (1979) Geographic variation in relation to life history in three species of the marine gastropod genus Crepidula: growth rates of newly hatched larvae and juveniles. In: Stanyck E (ed) Reproductive ecology of marine invertebrates. University of South Carolina Press, Columbia, pp 61–76
Arango CP (2002) Morphological phylogenetics of the sea spiders (Arthropoda: Pycnogonida). Org Divers Evol 2:107–125. doi:https://doi.org/10.1078/1439-6092-00035
Arango CP (2003) Molecular approach to the phylogenetics of sea spiders (Pycnogonida, Arthropoda) using nuclear ribosomal DNA and morphology. Mol Phylogenet Evol 28:588–600. doi:https://doi.org/10.1016/S1055-7903(03)00069-1
Arango CP, Wheeler WC (2007) Phylogeny of the sea spiders (Arthropoda, Pycnogonida) based on direct optimization of six loci and morphology. Cladistics 23:1–39. doi:https://doi.org/10.1111/j.1096-0031.2007.00143.x
Aris-Brosou S, Excoffier L (1996) The impact of population expansion and mutation rate heterogeneity on DNA sequence polymorphism. Mol Biol Evol 13:494–504
Arnaud F, Bamber RN (1987) The biology of Pycnogonida. Adv Mar Biol 24:1–95
Arndt A, Smith J (1998) Genetic diversity and population structure in two species of sea cucumber: differing patterns according to mode of development. Mol Ecol 7:1053–1064. doi:https://doi.org/10.1046/j.1365-294x.1998.00429.x
Avise JC (2000) Phylogeography: the history and formation of species. Harvard University Press, Cambridge
Avise JC, Arnold J, Ball RM Jr, Bermingham E, Lamb T, Neigel JE et al (1987) Intraspecific phylogeography: the mitochondrial DNA bridge between population genetics and systematics. Annu Rev Ecol Syst 18:489–522
Ayre DJ, Hughes TP (2000) Genotypic diversity and gene flow in brooding and spawning corals along the Great Barrier Reef, Australia. Evol Int J Org Evol 54:1590–1605
Bamber RN, El Nagar A (2008) Pycnobase: pycnogonida world database. http://www.marinespecies.org/pycnobase/
Bargelloni L, Lorenzo Z, Derome N, Lecointre G, Patarnello T (2000) Molecular zoography of Antarctic euphasiids and notothenioids: from species phylogenies to intraspecific patterns of genetic variation. Ant Sci 12(3):259–268. doi:https://doi.org/10.1017/S0954102000000328
Berger EM (1973) Gene-enzyme variation in three sympatric species of Littorina. Biol Bull 145:83–90. doi:https://doi.org/10.2307/1540349
Berger EM (1977) Gene-enzyme variation in three sympatric species of Littorina. II. The Roscoff population, with a note on the origin of North American L. littorea. Biol Bull 153:255–264. doi:https://doi.org/10.2307/1540433
Bogomolova EV, Malakhov VV (2003) Larvae of sea spiders (Arthropoda, Pycnogonida) from the White Sea. Entomol Rev (Engl Transl) 83(2):222–236
Child AC (1995) Antarcic and subantarctic pycnogonida III. The family Nymphonidae. In: Cairns S (ed) Antarctic and subantarctic Pycnogonida: Nymphonidae, Colossendeidae, Rhynchothoracidae, Pycnogonidae, Endeididae, and Callipallenidae. American Geophysical Union, Washington DC, pp 69–111
Clarke A, Johnson NM (2003) Antarctic marine benthic diversity. Oceanogr Mar Biol Ann Rev 41:47–114
Clarke A, Barnes DKA, Hodgson DA (2005) How isolated is Antarctica? Trends Ecol Evol 20:1–3. doi:https://doi.org/10.1016/j.tree.2004.10.004
Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9:1657–1659. doi:https://doi.org/10.1046/j.1365-294x.2000.01020.x
Crandall KA, Templeton AR, Sing CF (1994) Intraspecific phylogenetics: problems and solutions. In: Scotland RW, Siebert DJ, Williams DM (eds) Models in phylogeny reconstruction. systematics association special, vol 52. Clarendon Press, Oxford, pp 273–297
Dayton PK, Robilliard GA, Paine RT (1970) Benthic faunal zonation as a result of anchor ice at McMurdo Sound, Antarctica. Academic Press, London
Dell RK (1972) Antarctic benthos. Adv Mar Biol 10:1–216. doi:https://doi.org/10.1016/S0065-2881(08)60416-2
Duffy JE (1993) Genetic population structure in two tropical sponge-dwelling shrimps that differ in dispersal potential. Mar Biol (Berl) 116:459–470. doi:https://doi.org/10.1007/BF00350063
Dunlop JA, Arango CP (2005) Pycnogonid affinities: a review. J Zool Syst Evol Res 43:8–21. doi:https://doi.org/10.1111/j.1439-0469.2004.00284.x
Farris JS, Kallersjo M, Kluge AG, Bult C (1995) Constructing a significance test for incongruence. Syst Biol 44(4):570–572. doi:https://doi.org/10.2307/2413663
Folmer O, Black M, Hoeh W, Lutz R, Vrijenhoek R (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Mar Biol Biotechnol 3:294–299
Fry WG, Hedgpeth JW (1969) Pycnogonida. Colossendeidae, Pycnogonidae, Endeidae, Ammotheidae. NZ Dep Sci Ind Res Bull Part 7:1–139
Fu X (1996) New statistical test of neutrality for DNA samples from a population. Genetics 143:557–570
Gordon I (1944) Pycnogonida. Reports of the British. Aust NZ Antarct Res Expedition B5:1–72
Grant WS, Silva-Tatley da FM (1997) Lack of genetically-subdivided population structure in Bullia digitalis, a southern African marine gastropod with lecithotrophic development. Mar Biol (Berl) 129:123–137. doi:https://doi.org/10.1007/s002270050153
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hedgpeth JW (1947) On the evolutionary significance of the Pycnogonida. Smithsonian miscellaneous Collection, 106:1–53, 1 pl
Hedgpeth JW (1955) Paleoisopus. In: Moore RC (ed) Treatise on invertebrate paleontology, Vol. Part P, Arthropoda 2. Geological Society of America and University of Kansas Press, Lawrence, Kansas, pp P171–P173
Hedgpeth JW (1962) Introduction to seashore life of the San Francisco Bay Region and the Coast of Northern California. University of California Press, Berkeley
Held C (2003) Molecular evidence for cryptic speciation within the widespread Antarctic crustacean Ceratoserolis trilobitoides (Crustacea, Isopoda). In: Huiskes AHL, Giekes WWC, Rozema J, Schorno RML, van der Vies SM, Wolff WJ (eds) Antarctic biology in a global context. Backhuys Publishers, Leiden, pp 135–139
Held C, Wägele J-W (2005) Cryptic speciation in the giant Antarctic isopod Glyptonotus antarcticus (Isopoda: Valvifera: Chaetiliidae). Sci Mar 69:175–181
Hellberg ME (1996) Dependence of gene flow on geographic distance in two solitary corals with different larval dispersal capabilities. Evol Int J Org Evol 28:1167–1175. doi:https://doi.org/10.2307/2410657
Hempel G (1985) On the biology of polar Seas, particularly the Southern Ocean. Wiley, New York
Hewitt GM (1999) Post-glacial re-colonization of European biota. Biol J Linn Soc 68:87–112
Hewitt GM (2001) Speciation, hybrid zones and phylogeography—or seeing genes in space and time. Mol Ecol 10(3):537–549. doi:https://doi.org/10.1046/j.1365-294x.2001.01202.x
Hoskin MG (1997) Effects of contrasting modes of larval development on the genetic structure of three species of prosobranch gastropods. Mar Biol (Berl) 127:647–656. doi:https://doi.org/10.1007/s002270050055
Huelsenbeck JP, Ronquist F (2001) MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 1:754–755. doi:https://doi.org/10.1093/bioinformatics/17.8.754
Hunt A (1993) Effects of contrasting patterns of larval dispersal on the genetic connectedness of local populations of two intertidal starfish, Pateriella calcar and Pateriella exigua. Mar Ecol Prog Ser 92:179–186. doi:https://doi.org/10.3354/meps092179
Hunter RL, Halanych KM (2008) Evaluating connectivity in the brooding brittle star Astrotoma agassizii across the Drake Passage in the Southern Ocean. J Hered 99(2):137–148. doi:https://doi.org/10.1093/jhered/esm119
Johannesson K (1988) The paradox of Rockall: why is a brooding gastropod (Littorina saxatilis) more widespread than one having a planktic larval dispersal stage (L. littorea). Mar Biol (Berl) 99:507–513. doi:https://doi.org/10.1007/BF00392558
Johannesson K, Warmoes T (1990) Rapid colonization of Belgian breakwaters by the direct developer, Littorina saxatilis (Olivi) (Prosobranchia, Mollusca). Hydrobiologia 193:99–108. doi:https://doi.org/10.1007/BF00028069
King PE (1973) Pycnogonids. Hutchinson, London
Kumar S, Tamura K, Nei M (2004) MEGA3: integrated software for molecular evolutionary genetics analysis and sequence alignment. Brief Bioinform 5:150–163. doi:https://doi.org/10.1093/bib/5.2.150
Kyle CJ, Boulding EG (2000) Comparative population genetic structure of marine gastropods (Littorina spp.) with and without pelagic larval dispersal. Mar Biol (Berl) 137:835–845. doi:https://doi.org/10.1007/s002270000412
Marko P (2004) “What’s larvae got to do with it?” Disparate patterns of post-glacial population structure in two benthic marine gastropods with identical dispersal potential. Mol Ecol 13:597–611. doi:https://doi.org/10.1046/j.1365-294X.2004.02096.x
Mauchline J (1984) Pycnogonids caught in bathypelagic samples from the Rockall Trough, Northeastern Atlantic-Ocean. J Nat Hist 18(2):315–322. doi:https://doi.org/10.1080/00222938400770241
Mayr E (1970) Populations, species and evolution: an abridgement of animal species and evolution. Harvard University Press, Cambridge
McMillian WO, Raff RA, Palumbi SR (1992) Population genetic consequences of developmental evolution in sea urchins (genus Heliocidaris). Evol Int J Org Evol 46:1299–1312. doi:https://doi.org/10.2307/2409938
Munilla T (2001) Synopsis of the pycnogonids from Antarctic and Subantarctic waters. Polar Biology 24:941–945
Nakamura K, Kano Y, Suzuki N, Namatame T, Kosaku A (2007) 18S rRNA phylogeny of sea spiders with emphasis on the position of Rhynchothoracidae. Mar Biol (Berl) 153:213–223. doi:https://doi.org/10.1007/s00227-007-0803-0
Nylander JAA (2004) MRMODELTEST v2. Program distributed by the author. Evolutionary Biology Centre, Uppsala University
Palumbi SR, Martin AP, Romano S, McMillan WO, Stice L, Grabowski G (1991) The simple fool’s guide to PCR. University of Hawaii, Honolulu
Pearse JS (1979) Polyplacophora. Academic Press, New York
Pearse JS, Bosch I (1994) Brooding in the Antarctic: Östergren had it nearly right. In: David B, Guille A, Feral J-P, Roux M (eds) Echinoderms through time. B. Balkema, Rotterdam, pp 111–120
Pechenik JA (1999) On the advantages and disadvantages of larval stages in benthic marine invertebrate life cycles. Mar Ecol Prog Ser 177:269–297. doi:https://doi.org/10.3354/meps177269
Picken GB (1980) Reproductive adaptations of Antarctic benthic invertebrates. Biol J Linn Soc 14:67–75. doi:https://doi.org/10.1111/j.1095-8312.1980.tb00098.x
Posada D (2006) Collapse: describing haplotypes from sequence alignments. http://darwin.uvigo.es/software/collapse.html
Poulin E, Feral J-P (1996) Why are there so many species of brooding antarctic echinoids? Evol Int J Org Evol 50:820–830. doi:https://doi.org/10.2307/2410854
Rozas J, Sánchez-DelBarrio JC, Messeguer X, Rozas R (2003) DNASP, DNA polymorphism analyses by the coalescent and other methods. Bioinformatics 19:2496–2497. doi:https://doi.org/10.1093/bioinformatics/btg359
Schneider S, Roessli D, ExcoYer L (2000) Arlequin: a software for population genetics data analysis. University of Geneva, Geneva
Simon C, Frati F, Beckenbach A, Crespi B, Liu H, Flook P (1994) Evolution, weighting and phylogenetic utility of mitochondrial gene sequence and a compilation of conserved polymerase chain reaction primers. Ann Entomol Soc Am 87:651–701
Simpson RD (1977) The reproduction of some littoral molluscs from Macquarie Island (sub-Antarctic). Mar Biol (Berl) 44:135–142. doi:https://doi.org/10.1007/BF00386953
Slatkin M (1985) Gene flow in natural populations. Annu Rev Ecol Syst 16:393–430. doi:https://doi.org/10.1146/annurev.ecolsys.16.1.393
Sotka EE, Palumbi SR (2006) The use of genetic clines to estimate dispersal distances of marine larvae. Ecology 87(5):1094–1103. doi:https://doi.org/10.1890/0012-9658(2006)87[1094:TUOGCT]2.0.CO;2
Stewart JR, Lister AM (2001) Cryptic northern refugia and the origins of the modern biota. Trends Ecol Evol 16(11):608–613. doi:https://doi.org/10.1016/S0169-5347(01)02338-2
Tajima F (1989) Statistical methods to test for nucleotide mutation hypothesis by DNA polymorphism. Genetics 123:585–595
Templeton AR (1998) Nested clade analysis of phylogeographic data: testing hypotheses about gene flow and population history. Mol Ecol 7:381–397. doi:https://doi.org/10.1046/j.1365-294x.1998.00308.x
Thajte S, Hillenbrand C-D, Larter R (2005) On the origin of Antarctic marine benthic community structure. Trends Ecol Evol 20:534–540. doi:https://doi.org/10.1016/j.tree.2005.07.010
Thiel M, Gutow L (2005) The ecology of rafting in the marine environment. II. The rafting organisms and community. Oceanogr Mar Biol Ann Rev 43:279–418
Vermeij GJ (1978) Biogeography and adaptation: patterns of marine life. Harvard University Press, Cambridge
Vermeij GJ, Palmer AR, Lindberg DR (1990) Range limits and dispersal of mollusks in the Aleutian Islands, Alaska. Veliger 33:346–354
Ward RD (1990) Biochemical genetic variation in the genus Littorina (Prosobranchia: Mollusca). Hydrobiologia 193:53–69. doi:https://doi.org/10.1007/BF00028066
Wares JP, Cunningham CW (2001) Phylogeography and historical ecology of the North Atlantic intertidal. Evol Int J Org Evol 55(12):2455–2469
Wilson NG, Hunter RL, Lockhard SH, Halanych KM (2007) Multiple lineages and absence of panmixia in the “circumpolar”crinoid Promachocrinus kerguelensis from the Atlantic sector of Antarctica. Mar Biol (Berl) 152(4):895–904. doi:https://doi.org/10.1007/s00227-007-0742-9
Acknowledgments
The Edison-Chouest Offshore crew, Raytheon personnel, and scientific participants of the ASRV Laurence M. Gould in the 2004 and 2006 Antarctic cruises (LMG 04-14 and LMG 06-05, respectively) are gratefully acknowledged for their help and logistical support. This work was supported by a National Science Foundation grant to KMH (OPP-0338218) and an Australian Biological Resources Study (ABRS) grant to CPA (204-61). This work is AU Marine Biology Program contribution #41.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by T. Reusch.
Electronic supplementary material
Below is the link to the electronic supplementary material.
227_2008_1029_MOESM1_ESM.eps
Supplementary Figure 1. Bayesian analysis of combined COI + 16S collapsed haplotype dataset using the GTR+I+G model of substitution, displaying only the clade containing Nymphon australe and the outgroup Nymphon paucituberculatum. Inset displays the entire tree topology. Haplotype designations (Roman numerals) correlate to data presented in Figure 3 and Supplementary Table 3 (EPS 878 kb)
Rights and permissions
About this article
Cite this article
Mahon, A.R., Arango, C.P. & Halanych, K.M. Genetic diversity of Nymphon (Arthropoda: Pycnogonida: Nymphonidae) along the Antarctic Peninsula with a focus on Nymphon australe Hodgson 1902. Mar Biol 155, 315–323 (2008). https://doi.org/10.1007/s00227-008-1029-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-008-1029-5