Abstract
Rationale
Understanding naltrexone’s effect on motivation to drink and pattern of drinking is important for better treatment outcomes and for comparison with novel medications.
Objectives
Naltrexone’s effects on number and pattern of seeking, self-administration, and extinction responses were evaluated in two groups of baboons trained under a three-component chained schedule of reinforcement (CSR).
Methods
Alcohol (4 % w/v; n = 4; alcohol group) or a preferred nonalcoholic beverage (n = 4; control group) was available for self-administration only in component 3 of the CSR. Responses in component 2 provided indices of motivation to drink (seeking). Naltrexone (0.32–3.2 mg/kg) and saline were administered before drinking and component 2 extinction sessions.
Results
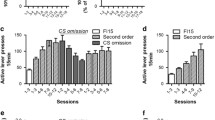
Acute doses of naltrexone significantly decreased total self-administration responses (p < 0.01), intake volume (p < 0.001), and grams per kilogram of alcohol (p < 0.01) in the alcohol group only. Pattern of drinking did not change, but the number of drinks during the initial drinking bout was decreased significantly by naltrexone for both groups (p < 0.05). During within-session extinction tests, acute naltrexone significantly decreased time to reach extinction (p < 0.01) and number of seeking responses (p < 0.05), particularly early in the extinction period in the alcohol group only. When administered chronically, naltrexone did not decrease progressive ratio breaking points to gain access to alcohol, but dose dependently reduced alcohol self-administration (p < 0.05) by decreasing the magnitude of the initial drinking bout.
Conclusions
The results support clinical observations that naltrexone may be most effective at reducing self-administration in the context of ongoing alcohol availability and may reduce motivation to drink in the presence of alcohol-related cues.




Similar content being viewed by others
References
Altshuler PPE, Feinhandler DA (1980) Alterations of ethanol self-administration by naltrexone. Life Sci 26:679–684
Anton RF, Swift RM (2003) Current pharmacotherapies of alcoholism: a U.S. perspective. Am J Addict 12(Suppl 1):S53–68
Anton RF, Moak DH, Waid LR, Latham PK, Malcolm RJ, Dias JK (1999) Naltrexone and cognitive behavioral therapy for the treatment of outpatient alcoholics: results of a placebo-controlled trial. Am J Psychiatry 156:1758–1764
Anton RF, O’Malley SS, Ciraulo DA, Cisler RA, Couper D, Donovan DM (2006) Combined pharmacotherapies and behavioral interventions for alcohol dependence: the COMBINE study: a randomized controlled trial. JAMA 295:2003–2017
Ator NA, Griffiths RR (1992) Oral self-administration of triazolam, diazepam and ethanol in the baboon: drug reinforcement and benzodiazepine physical dependence. Psychopharmacology (Berl) 108:301–312
Bienkowski P, Kostowski W, Koros E (1999) Ethanol-reinforced behaviour in the rat: effects of naltrexone. Eur J Pharmacol 374:321–327
Bouza C, Angeles M, Muno A, Amate JM (2004) Efficacy and safety of naltrexone and acamprosate in the treatment of alcohol dependence: a systematic review. Addiction 99:811–828
Boyle AE, Stewart RB, Macenski MJ, Spiga R, Johnson BA, Meisch RA (1998) Effects of acute and chronic doses of naltrexone on ethanol self-administration in rhesus monkeys. Alcohol Clin Exp Res 22:359–366
Bullock DH (1960) Repeated conditioning-extinction sessions as a function of the reinforcement schedule. J Exp Anal Behav 3:241–243
Bullock DH, Smith WC (1953) An effect of repeated conditioning-extinction upon operant strength. J Exp Psych 46:349–352
Childress A, Ehrman R, Rohsenow D, Robbins S, O’Brien C (1992) Classically conditioned factors in drug dependence. In: Lowinson P, Luiz P, Millman RB, Langard G (eds) Substance abuse: a comprehensive textbook. Williams & Wilkins, Baltimore, pp 56–69
Ciccocioppo R, Martin-Fardon R, Weiss F (2002) Effect of selective blockade of μ1 δ opioid receptors on reinstatement of alcohol-seeking behavior by drug-associated stimuli in rats. Neuropsychopharmacology 27:391–399
Clark GC, Taylor BW (1960) Effects of repeated extinction of an operant on characteristics of extinction curves. Psych Rep 6:226
Cohen C, Perrault G, Griebel G, Soubrie P (2005) Nicotine-associated cues maintain nicotine-seeking behavior in rats several weeks after nicotine withdrawal: reversal by the cannabinoid (CB1) receptor antagonist, rimonabant (SR141716). Neuropsychopharmacology 30:145–155
Collins B, Brandon T (2002) Effects of extinction context and retrieval cues on alcohol cue reactivity among nonalcoholic drinkers. J Consult Clin Psychol 70:390–397
Crombag H, Shaham Y (2002) Renewal of drug seeking by contextual cues after prolonged extinction in rats. Behav Neurosci 116:169–173
Czachowski CL, DeLory MJ (2010) Acamprosate and naltrexone treatment effects of ethanol and sucrose seeking and intake in ethanol-dependent and nondependent rats. Psychopharmacology 204:335–348
Davidson D, Palfai T, Bird C, Swift R (1999) Effects of naltrexone on alcohol self-administration in heavy drinkers. Alcohol Clin Exp Res 23:195–203
Dews PB (1976) Interspecies differences in drug effects: behavioral. In: Usdin E, Forrest IS (eds) Psychotherapeutic drugs, part I. Marcel Dekker, New York, pp 175–214
Egli M (2005) Can experimental paradigms and animal models be used to discover clinically effective medications for alcoholism? Addict Biol 10:309–319
Ferraro FM, Hill KG, Kaczmarek HJ, Coonfield DL, Kiefer SW (2002) Naltrexone modifies the palatability of basic tastes and alcohol in outbred male rats. Alcohol 27:107–114
Gracy KN, Dankiewicz LA, Weiss F, Koob GF (2000) Heroin-specific stimuli reinstate operant heroin-seeking behavior in rats after prolonged extinction. Pharmacol Biochem Behav 65:489–494
Grant KA, Bennett AJ (2003) Advances in nonhuman primate alcohol abuse and alcoholism research. Pharmacol Ther 100:235–255
Henningfield JE, Ator NA, Griffiths RR (1981) Establishment and maintenance of oral ethanol self-administration in the baboon. Drug Alcohol Depend 7:113–124
Hill KG, Kiefer SW (1997) Naltrexone treatment increases the aversiveness of alcohol for outbred rats. Alcohol Clin Exp Res 21:637–641
Jimenez-Gomez C, Shahan TA (2007) Resistance to change of alcohol self-administration: effects of alcohol-delivery rate on disruption by extinction and naltrexone. Behav Pharmacol 18:161–169
Johnson BA (2008) Update on neuropharmacological treatment for alcoholism: scientific basis and clinical findings. Biochem Pharmacol 75:34–56
Johnson BA (2010) Medication treatment of different types of alcoholism. Am J Psychiatry 167:630–639
Johnson BA, Swift RM, Ait-Daoud N, DiClemente CC, Javors MA, Malcolm RJ Jr (2004) Development of novel pharmacotherapies for the treatment of alcohol dependence: focus on antiepileptics. Alcohol Clin Exp Res 28:295–301
Kaminski BJ, Goodwin AK, Wand G, Weerts EM (2008) Dissociation of alcohol-seeking and consumption under a chained schedule of oral alcohol reinforcement. Alcohol Clin Exp Res 32:1–9
Katner SN, Magalong JG, Weiss F (1999) Reinstatement of alcohol-seeking behavior by drug-associated discriminative stimuli after prolonged extinction in the rat. Neuropsychopharmacology 20:471–479
Killeen TK, Brady KT, Gold PB, Simpson KN, Faldowski RA, Tyson C, Anton RF (2004) Effectiveness of naltrexone in a community treatment program. Alcohol Clin Exp Res 28:1710–1717
King AC, Volpicelli JR, Frazer A, O’Brien CP (1997) Effect of naltrexone on subjective alcohol response in subjects at high and low risk for future alcohol dependence. Psychopharmacology (Berl) 129:15–22
Krank MD (2003) Pavlovian conditioning with ethanol: sign-tracking (autoshaping), conditioned incentive, and ethanol self-administration. Alcohol Clin Exp Res 27:1592–1598
Lê AD, Poulos CX, Quan B, Chow S (1993) The effects of selective blockade of delta and mu opiate receptors on ethanol consumption by C57BL/6 mice in a restricted access paradigm. Brain Res 630:330–332
Leeman RF, Heilig M, Cunningham CL, Stephens DN, Duka T, O’Malley SS (2010) Ethanol consumption: how should we measure it? Achieving consilience between human and animal phenotypes. Addict Biol 15:109–124
Liu X, Weiss F (2002) Additive effect of stress and drug cues on reinstatement of ethanol seeking: exacerbation by history of dependence and role of concurrent activation of corticotropin-releasing factor and opioid mechanisms. J Neurosci 22:7856–7861
Macenski MJ, Meisch RA (1992) Ethanol-reinforced responding of naïve rhesus monkeys: acquisition without induction procedures. Alcohol 9:547–554
Markou A, Weiss F, Gold LH, Caine SB, Schulteis G, Koob GF (1993) Animal models of drug craving. Psychopharmacology (Berl) 112:163–182
McCaul ME, Wand GS, Eissenberg T, Rohde CA, Cheskin LJ (2000) Naltrexone alters subjective and psychomotor responses to alcohol in heavy drinking subjects. Neuropsychopharmacology 22:480–492
Monterosso JR, Flannery BA, Pettinati HM, Oslin DW, Rukstalis M, O’Brien CP, Volpicelli JR (2001) Predicting treatment response to naltrexone: the influence of craving and family history. Am J Addict 10:258–268
Mordenti J, Chappell W (1989) The use of interspecies scaling in toxicokenetics. In: Yacobi A, Kelly J, Batra V (eds) Toxicokenetics and new drug development. Pergamon Press, New York, pp 42–96
Olszewski PK, Alsio J, Schioth HB, Levine AS (2011) Opioids as facilitators of feeding: can any food be rewarding? Physiol Behav 104:105–110
O’Malley SS, Froehlich JC (2003) Advances in the use of naltrexone: an integration of preclinical and clinical findings. Recent Dev Alcohol 16:217–245
Pettinati HM, O’Brien CP, Rabinowitz AR, Wortman SP, Oslin DW, Kampman KM, Dackis CA (2006) The status of naltrexone in the treatment of alcohol dependence: specific effects on heavy drinking. J Clin Psychopharmacol 26:610–625
Ray LA, Krull JL, Leggio L (2010) The effects of naltrexone among alcohol non-abstainers: results from the COMBINE Study. Front Psychiatr 1:1–6
Richardson K, Baillie A, Reid S, Morley K, Teesson M, Sannibale C, Weltman M, Haber P (2008) Do acamprosate or naltrexone have an effect on daily drinking by reducing craving for alcohol? Addiction 103:953–959
Rodefer JS, Campbell UC, Cosgrove KP, Carroll ME (1999) Naltrexone pretreatment decreases the reinforcing effectiveness of ethanol and saccharin but not PCP or food under concurrent progressive-ratio schedules in rhesus monkeys. Psychopharmacology 141:436–446
Rosner S, Hackl-Herrwerth A, Leucht S, Vecchi S, Srisurapanont M, Soyka M (2010) Opioid antagonists for alcohol dependence. Cochrane Database Syst Rev 12
Samson HH, Czachowski CL, Slawecki CJ (2000) A new assessment of the ability of oral ethanol to function as a reinforcing stimulus. Alcohol Clin Exp Res 24:766–773
Setiawan E, Pihl RO, Cox SML, Gianoulakis C, Palmour RM, Benkelfat C, Leyton M (2011) The effect of naltrexone on alcohol’s stimulant properties and self-administration behavior in social drinkers: influence of gender and genotype. Alcohol Clin Exp Res 35:1134–1141
Shahan TA, Burke KA (2004) Ethanol-maintained responding of rats is more resistant to change in a context with alternative non-drug reinforcement. Behav Pharmacol 15:279–285
Shelton KL, Grant KA (2001) Effects of naltrexone and Ro15-4513 on a multiple schedule of ethanol and Tang self-administration. Alcohol Clin Exp Res 25:1576–1585
Ulm RR, Volpicelli JR, Volpicelli LA (1995) Opiates and alcohol self-administration in animals. J Clin Psychiatr 56(Supple7):5–14
Unterwald EM (2008) Naltrexone in the treatment of alcohol dependence. J Addict Med 2:121–127
Vivian JA, Green HL, Young JE et al (2001) Induction and maintenance of ethanol self-administration in cynomolgus monkeys (Macaca fascicularis): long-term characterization of sex and individual differences. Alcohol Clin Exp Res 25:1087–1097
Weerts EM, Goodwin AK, Kaminski BJ, Hienz RD (2006) Environmental cues, alcohol seeking, and consumption in baboons: effects of response requirement and duration of alcohol abstinence. Alcohol Clin Exp Res 30:2026–2036
Weiss F (2010) Advances in animal models of relapse for addiction research. In: Kuhn CM, Koob GF (eds) Advances in the neuroscience of addiction, 2nd edn. CRC Press, Boca Rotan
Williams KL, Woods JH (1999) Naltrexone reduces ethanol- and/or water-reinforced responding in rhesus monkeys: effect depends upon ethanol concentration. Alcohol Clin Exp Res 23:1462–1467
Williams KL, Winger G, Pakarinen ED, Woods JH (1998) Naltrexone reduces ethanol- and sucrose-reinforced responding in rhesus monkeys. Psychopharmacology 139:53–61
Yoon G, Kim SW, Thuras P, Westermeyer J (2011) Safety, tolerability, and feasibility of high-dose naltrexone in alcohol dependence: an open-label study. Hum Psychopharmacol 26:125–132
Zarcone TJ, Ator NA (2000) Drug discrimination: stimulus control during repeated testing in extinction. J Exp Anal Behav 74:283–294
Disclosure/conflict of interest
This research was supported by NIH/NIAAA R01AA15971. Dr. Weerts was a co-investigator on a contract in human subjects (A Phase 2 Study of LY2196044 Compared with Naltrexone and Placebo in the Treatment of Alcohol Dependence) funded by Lilly Research Laboratories. Drs. Duke and Kaminski have no conflicts to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaminski, B.J., Duke, A.N. & Weerts, E.M. Effects of naltrexone on alcohol drinking patterns and extinction of alcohol seeking in baboons. Psychopharmacology 223, 55–66 (2012). https://doi.org/10.1007/s00213-012-2688-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-012-2688-y