Abstract
Summary
This study shows that teriparatide promotes the circulating osteoblast (OB) precursor degree of maturation in patients affected by postmenopausal osteoporosis.
Introduction
Anabolic treatment with teriparatide has proven effective for the therapy of postmenopausal osteoporosis and significantly reduces the risk of non-vertebral fragility fractures. The aim of this study was to investigate the effect of teriparatide on circulating OB precursors.
Methods
We evaluated by flow cytometry and real-time PCR the expression of OBs typical markers in peripheral blood mononuclear cells during treatment with teriparatide plus calcium and vitamin D, raloxifene plus calcium and vitamin D or calcium and vitamin D alone at various time points. Serum bone alkaline phosphatase and osteocalcin (OC) were measured as markers of bone turnover.
Results
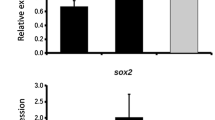
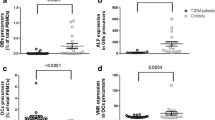
Our results show that circulating OB precursors are more numerous and more immature in patients affected by fragility fractures than in osteoporotic patients without fractures. We also show that teriparatide treatment increases the expression of alkaline phosphatase and of OC in OB precursors; thus, it increases their degree of maturation.
Conclusions
We suggest that teriparatide acts as anabolic agents also by promoting the maturation of OB precursors.






Similar content being viewed by others
References
Eghbali-Fatourechi GZ, Lamsam J, Fraser D, Nagel D, Riggs BL, Khosla S (2005) Circulating osteoblast-lineage cells in humans. N Engl J Med 352:1959–1966
Eghbali-Fatourechi GZ, Modder UI, Charatcharoenwitthaya N, Sanyal A, Undale AH, Clowes JA, Tarara JE, Khosla S (2007) Characterization of circulating osteoblast lineage cells in humans. Bone 40:1370–1377
D’Amelio P, Cristofaro MA, Grimaldi A, Ravazzoli M, Pluviano F, Grosso E, Pescarmona GP, Isaia GC (2010) The role of circulating bone cell precursors in fracture healing. Calcif Tissue Int 86:463–469
Kumagai K, Vasanji A, Drazba JA, Butler RS, Muschler GF (2008) Circulating cells with osteogenic potential are physiologically mobilized into the fracture healing site in the parabiotic mice model. J Orthop Res 26:165–175
Pirro M, Leli C, Fabbriciani G, Manfredelli MR, Callarelli L, Bagaglia F, Scarponi AM, Mannarino E (2010) Association between circulating osteoprogenitor cell numbers and bone mineral density in postmenopausal osteoporosis. Osteoporos Int 21:297–306
D’Amelio P, Roato I, D’Amico L, Veneziano L, Suman E, Sassi F, Bisignano G, Ferracini R, Gargiulo G, Castoldi F, Pescarmona GP, Isaia GC (2011) Bone and bone marrow pro-osteoclastogenic cytokines are up-regulated in osteoporosis fragility fractures. Osteoporos Int (in press)
Velasco J, Zarrabeitia MT, Prieto JR, Perez-Castrillon JL, Perez-Aguilar MD, Perez-Nunez MI, Sanudo C, Hernandez-Elena J, Calvo I, Ortiz F, Gonzalez-Macias J, Riancho JA (2010) Wnt pathway genes in osteoporosis and osteoarthritis: differential expression and genetic association study. Osteoporos Int 21:109–118
Greenspan SL, Bone HG, Ettinger MP, Hanley DA, Lindsay R, Zanchetta JR, Blosch CM, Mathisen AL, Morris SA, Marriott TB (2007) Effect of recombinant human parathyroid hormone (1–84) on vertebral fracture and bone mineral density in postmenopausal women with osteoporosis: a randomized trial. Ann Intern Med 146:326–339
Neer RM, Arnaud CD, Zanchetta JR, Prince R, Gaich GA, Reginster JY, Hodsman AB, Eriksen EF, Ish-Shalom S, Genant HK, Wang O, Mitlak BH (2001) Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med 344:1434–1441
Jilka RL, Weinstein RS, Bellido T, Roberson P, Parfitt AM, Manolagas SC (1999) Increased bone formation by prevention of osteoblast apoptosis with parathyroid hormone. J Clin Invest 104:439–446
Juppner H, Abou-Samra AB, Freeman M, Kong XF, Schipani E, Richards J, Kolakowski LF Jr, Hock J, Potts JT Jr, Kronenberg HM et al (1991) A G protein-linked receptor for parathyroid hormone and parathyroid hormone-related peptide. Science 254:1024–1026
Abou-Samra AB, Juppner H, Force T, Freeman MW, Kong XF, Schipani E, Urena P, Richards J, Bonventre JV, Potts JT Jr et al (1992) Expression cloning of a common receptor for parathyroid hormone and parathyroid hormone-related peptide from rat osteoblast-like cells: a single receptor stimulates intracellular accumulation of both cAMP and inositol trisphosphates and increases intracellular free calcium. Proc Natl Acad Sci U S A 89:2732–2736
Bringhurst FR, Juppner H, Guo J, Urena P, Potts JT Jr, Kronenberg HM, Abou-Samra AB, Segre GV (1993) Cloned, stably expressed parathyroid hormone (PTH)/PTH-related peptide receptors activate multiple messenger signals and biological responses in LLC-PK1 kidney cells. Endocrinology 132:2090–2098
Schwindinger WF, Fredericks J, Watkins L, Robinson H, Bathon JM, Pines M, Suva LJ, Levine MA (1998) Coupling of the PTH/PTHrP receptor to multiple G-proteins. Direct demonstration of receptor activation of Gs, Gq/11, and Gi(1) by [alpha-32P]GTP-gamma-azidoanilide photoaffinity labeling. Endocr 8:201–209
Shinoda Y, Kawaguchi H, Higashikawa A, Hirata M, Miura T, Saito T, Nakamura K, Chung UI,Ogata N Mechanisms underlying catabolic and anabolic functions of parathyroid hormone on bone by combination of culture systems of mouse cells. J Cell Biochem 109:755–763
Undale A, Srinivasan B, Drake M, McCready L, Atkinson E, Peterson J, Riggs BL, Amin S, Modder UI, Khosla S (2010) Circulating osteogenic cells: characterization and relationship to rates of bone loss in postmenopausal women. Bone 47:83–92
Kream BE, LaFrancis D, Petersen DN, Woody C, Clark S, Rowe DW, Lichtler A (1993) Parathyroid hormone represses alpha 1(I) collagen promoter activity in cultured calvariae from neonatal transgenic mice. Mol Endocrinol 7:399–408
Clohisy JC, Scott DK, Brakenhoff KD, Quinn CO, Partridge NC (1992) Parathyroid hormone induces c-fos and c-jun messenger RNA in rat osteoblastic cells. Mol Endocrinol 6:1834–1842
Civitelli R, Hruska KA, Jeffrey JJ, Kahn AJ, Avioli LV, Partridge NC (1989) Second messenger signaling in the regulation of collagenase production by osteogenic sarcoma cells. Endocrinology 124:2928–2934
Greenfield EM, Shaw SM, Gornik SA, Banks MA (1995) Adenyl cyclase and interleukin 6 are downstream effectors of parathyroid hormone resulting in stimulation of bone resorption. J Clin Invest 96:1238–1244
Miki Y, Suzuki T, Nagasaki S, Hata S, Akahira J, Sasano H (2009) Comparative effects of raloxifene, tamoxifen and estradiol on human osteoblasts in vitro: estrogen receptor dependent or independent pathways of raloxifene. J Steroid Biochem Mol Biol 113:281–289
Matsumori H, Hattori K, Ohgushi H, Dohi Y, Ueda Y, Shigematsu H, Satoh N, Yajima H, Takakura Y (2009) Raloxifene: its ossification-promoting effect on female mesenchymal stem cells. J Orthop Sci 14:640–645
D’Amelio P, Muratore M, Tinelli F, Tamone C, Cosentino L, Quarta E, Calcagnile F, Carlo Isaia G (2003) Effect of raloxifene and clodronate on bone density in postmenopausal osteoporotic women. Int J Tissue React 25:73–78
D’Amelio P, Grimaldi A, Cristofaro MA, Ravazzoli M, Molinatti PA, Pescarmona GP, Isaia GC (2010) Alendronate reduces osteoclast precursors in osteoporosis. Osteoporos Int 21:1741–1750
D’Amelio P, Grimaldi A, Di Bella S, Tamone C, Brianza SZ, Ravazzoli MG, Bernabei P, Cristofaro MA, Pescarmona GP, Isaia G (2008) Risedronate reduces osteoclast precursors and cytokine production in postmenopausal osteoporotic women. J Bone Miner Res 23:373–379
D’Amelio P, Grimaldi A, Pescarmona GP, Tamone C, Roato I, Isaia G (2005) Spontaneous osteoclast formation from peripheral blood mononuclear cells in postmenopausal osteoporosis. FASEB J 19:410–412
Ballen K (2007) Targeting the stem cell niche: squeezing blood from bones. Bone Marrow Transplant 39:655–660
Gossl M, Modder UI, Atkinson EJ, Lerman A, Khosla S (2008) Osteocalcin expression by circulating endothelial progenitor cells in patients with coronary atherosclerosis. J Am Coll Cardiol 52:1314–1325
Chen P, Miller PD, Recker R, Resch H, Rana A, Pavo I, Sipos AA (2007) Increases in BMD correlate with improvements in bone microarchitecture with teriparatide treatment in postmenopausal women with osteoporosis. J Bone Miner Res 22:1173–1180
Dobnig H, Turner RT (1995) Evidence that intermittent treatment with parathyroid hormone increases bone formation in adult rats by activation of bone lining cells. Endocrinology 136:3632–3638
Nishida S, Yamaguchi A, Tanizawa T, Endo N, Mashiba T, Uchiyama Y, Suda T, Yoshiki S, Takahashi HE (1994) Increased bone formation by intermittent parathyroid hormone administration is due to the stimulation of proliferation and differentiation of osteoprogenitor cells in bone marrow. Bone 15:717–723
Pettway GJ, Meganck JA, Koh AJ, Keller ET, Goldstein SA, McCauley LK (2008) Parathyroid hormone mediates bone growth through the regulation of osteoblast proliferation and differentiation. Bone 42:806–818
Schmidt IU, Dobnig H, Turner RT (1995) Intermittent parathyroid hormone treatment increases osteoblast number, steady state messenger ribonucleic acid levels for osteocalcin, and bone formation in tibial metaphysis of hypophysectomized female rats. Endocrinology 136:5127–5134
Almeida M, Han L, Bellido T, Manolagas SC, Kousteni S (2005) Wnt proteins prevent apoptosis of both uncommitted osteoblast progenitors and differentiated osteoblasts by beta-catenin-dependent and -independent signaling cascades involving Src/ERK and phosphatidylinositol 3-kinase/AKT. J Biol Chem 280:41342–41351
Bellido T, Ali AA, Gubrij I, Plotkin LI, Fu Q, O’Brien CA, Manolagas SC, Jilka RL (2005) Chronic elevation of parathyroid hormone in mice reduces expression of sclerostin by osteocytes: a novel mechanism for hormonal control of osteoblastogenesis. Endocrinology 146:4577–4583
Rubin MR, Manavalan JS, Dempster DW, Shah J, Cremers S, Kousteni S, Zhou H, McMahon DJ, Kode A, Sliney J, Shane E, Silverberg SJ, Bilezikian JP (2011) Parathyroid hormone stimulates circulating osteogenic cells in hypoparathyroidism. J Clin Endocrinol Metab 96:176–186
Acknowledgments
This work was supported by an unrestricted grant from Ely Lilly and the Fondazione Internazionale Ricerche Medicina Sperimentale (FIRMS) Compagnia San Paolo. SP was supported by a fellowship from the Italian Association for Cancer Research (AIRC); FS was supported by a fellowship of the Regione Piemonte. Teriparatide and Raloxifene were kindly provided by Ely Lilly; calcium and vitamin D supplements were kindly provided by Italfarmaco SpA.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
supplemental Fig. 1
Flow cytometry analyses of OB precursors. a Dot plot represents PBMCs, monocytes are indicated (R1). b Dot plot represents CD 15 positive cells (R2) analyzed on the whole PBMCs population. c Dot plot represents monocytes positive for AP and OC, CD15-positive cells (R2) were excluded by the analyses. d Histogram represents the mean fluorescence intensity (MFI) of OC+ cells (gated on monocytes after exclusion of CD 15+ cells). Broken line is the isotype control; unbroken line is the stained sample. (PPT 271 kb)
supplemental Table 1
Gene primer sequences. F forward, R reverse. All the genes were quantified by considering signals under 33 Ct. (DOC 26 kb)
Rights and permissions
About this article
Cite this article
D’Amelio, P., Tamone, C., Sassi, F. et al. Teriparatide increases the maturation of circulating osteoblast precursors. Osteoporos Int 23, 1245–1253 (2012). https://doi.org/10.1007/s00198-011-1666-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-011-1666-2