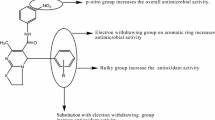
Abstract
Recently there has been an increased interest in the synthesis of pyrimidinone and their applications in medicinal chemistry. Pyrimidinone derivatives represent one of the active classes of the compounds possessing a wide spectrum of biological activities such as anti-tumour, anti-fungal, anti-inflammatory, anti-bacterial, etc. This study utilises chalcones as starting compounds that are obtained by Claisen-Schmidt condensation. These chalcones underwent cyclisation upon treatment with urea and thiourea in the presence of sodium hydroxide to obtain the corresponding pyrimidinones and pyrimidinethiones. The structures of the synthesized compounds were investigated by means of UV, IR, 1H NMR and Mass spectral analysis. The anti-oxidant activity of the compounds was measured by standard free radical scavenging assays (DPPH, Nitric oxide radicals). The compounds with significant anti-oxidant activity were then screened for the in vivo anti-inflammatory activity and it showed that the compounds ETT and DMT possess good activity as comparable to the standard. All the synthesized compounds were evaluated for their in vitro anti-cancer activity by MTT assay among which compounds CLU and ETU showed significant activity against HeLa and HepG2 cell lines, respectively.





Similar content being viewed by others
References
Azam MA, Kumar BRP, Shalini S, Suresh B, Reddy TK, Reddy CD (2008) Synthesis and biological screening of 5-{[(4,6-disubstituted pyrimidine-2-yl)thio]methyl}-N-phenyl-1,3,4-thiadiazol-2-amines. Indian J Pharm Sci 70(5):672–677
Baddiley J, Lythgoe B, Todd AR (1944) Experiments on the synthesis of purine nucleosides. Part VI. The synthesis of 9-d-xylosido-2-methyladenine and 6-d-xylosidamino-2-methylpurine. J Chem Soc 318–322. doi:10.1039/JR9440000318
Batovska D, Slavova A, Ninova M (2007) Study on the substituents’ effects of a series of synthetic chalcones against the yeast Candida albicans. Eur J Med Chem 42:87–92
Chetana BP, Mahajan SK, Suvarna AK (2009) Chalcone: a versatile molecule. J Pharm Sci Res 1(3):11–22
Dipti RP, Sonali MS, Madhukar BD, Prashant VA (2010) One step synthesis of 6-amino-5-cyano-4-phenyl-2-mercaptopyrimidine using phosphorous pentoxide. The Open Catal J 3:83–86
Ducki S, Forrest R, Hadfield JA, Kendall A, Lawrence NJ, Rennison D (1998) Potent antimitotic and cell growth inhibitory property of substituted chalcones. Bioorg Med Chem Lett 8:1051–1056
Fujiwara N, Nakajima T, Ueda Y, Fujita H, Kawakami H (2008) Novel piperidinylpyrimidine derivatives as inhibitors of HIV-1 LTR activation. Bioorg Med Chem 16:9804–9816
Hugo PA, de Elza FAS, Franco DM, Artur SJ (2008) Structure-activity relationship of antibacterial chalcones. Bioorg Med Chem Lett 16:9790–9794
Kenner GW, Lythgoe B, Todd AR, Topham A (1943) Experiments on the synthesis of purine nucleosides. experiments on the synthesis of purine nucleosides, Part IV. 4,6-diaminopyrimidine. A new synthesis of pyrimidine derivatives. J Chem Soc 574–575. doi:10.1039/JR9430000574
Lang Jr, Cohen E (1975) 4-amino-5-arylpyrimidines as antiinflammatory agents. J Med Chem 18(6):623–625
Maria CP, Claudia M, Michela LR, Federico C, Maurizio B (2004) Solid phase synthesis of 5,6-disubstituted pyrimidindione derivatives. Arkivoc 5:349–363
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Mudaliar VR, Joshi V (1995) Synthesis and insecticidal activity of new substituted phenoxychalcones. Indian J Chem 34B:456–458
Neeraj K, Jain JS, Sinha R, Garg VK, Bansal SK (2009) Evaluation of some novel chalcone derivatives for antimicrobial and anti-inflammatory activity. Der Pharmacia Lettre 1(1):169–176
Nicholas JL, Richard PP, Darren C (2006) Effects of α-substitutions on structure and biological activity of anti-cancer chalcones. Bioorg Med Chem Lett 16:5844–5848
Nowakowska Z (2007) A review of anti-infective and anti-inflammatory chalcones. Eur J Med Chem 42:125–137
Rajesh SB, Sidhanath VB, Sheshanath VB, Tianyu W, Zubaidha PK (2004) An efficient, high yield protocol for the one-pot synthesis of dihydropyrimidin-2(1H)-ones catalysed by iodine. Tetrahedron Lett 4:9111–9113
Shivarema Holla B, Sooryanarayana Rao B, Sarojini BK (2004) One pot synthesis of thiazolodihydropyrimidinones and evaluation of their anticancer activity. Eur J Med Chem 39:777–783
Singh P, Kaur P, Luxami V, Kumar S (2007) Synthesis and anti-cancer activities of 2-[1-(indol-3-yl-/pyrimidin-5-yl-/quinolin-2-yl)-but-3-enylamino]-2-phenyl-ethanols. Bioorg Med Chem 15(6):2386–2395
Singh OM, Singh SJ, Devi MB, Devi LN, Lee S-G (2008) Synthesis and in vitro evaluation of the antifungal activities of dihydropyrimidinones. Bioorg Med Chem Lett 18:6462–6467
Sun W, Halller D (2001) UFT in the treatment of colorectal and breast cancer. Oncology (Williston Park the Huntington) 15:49–50
Wageeh SE, Kamelia MA, Hanaa AT, Mausa EM (2011) Synthesis and antimicrobial activity of new 3,4-dihydropyrimidinones. Int J Pharm Sci Res 2(4):1054–1062
Winter CA, Risley EA, Nuss GW (1962) Carrageenan induced oedema in hind paw of the rat as an assay for anti-inflammatory drugs. Proc Soc Exp Biol Med 111:544–547
Yang W, Ruan Z, Dickson JK (2009) Discovery and structure-activity relationship of trisubstituted pyrimidines/pyridines as novel calcium-sensing receptor antagonists. J Med Chem 52(4):1204–1208
Acknowledgments
The authors (Vachala SD and Divya J) are thankful to AICTE and DST for providing the instrumental facilities (FTIR and UV). The authors are also thankful to Manipal University, Manipal, and Dr. N. Udupa, Principal, Manipal College of Pharmaceutical Sciences, for providing all necessary facilities to carry out this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dinakaran, V.S., Jacob, D. & Mathew, J.E. Synthesis and biological evaluation of novel pyrimidine-2(1H)-ones/thiones as potent anti-inflammatory and anticancer agents. Med Chem Res 21, 3598–3606 (2012). https://doi.org/10.1007/s00044-011-9909-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-011-9909-5