Abstract.
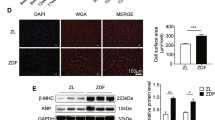
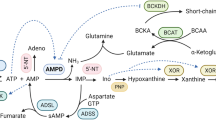
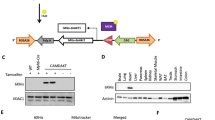
Mitochondrial dysfunction and protein kinase C (PKC) activation are consistently found in diabetic cardiomyopathy but their relationship remains unclear. This study identified mitochondrial aconitase as a downstream target of PKC activation using immunoblotting and mass spectrometry, and then characterized phosphorylation-induced changes in its activity in hearts from type 1 diabetic rats. PKCβ2 co-immunoprecipitated with phosphorylated aconitase from mitochondria isolated from diabetic hearts. Augmented phosphorylation of mitochondrial aconitase in diabetic hearts was found to be associated with an increase in its reverse activity (isocitrate to aconitate), while the rate of the forward activity was unchanged. Similar results were obtained on phosphorylation of mitochondrial aconitase by PKCβ2 in vitro. These results demonstrate the regulation of mitochondrial aconitase activity by PKC-dependent phosphorylation. This may influence the activity of the tricarboxylic acid cycle, and contribute to impaired mitochondrial function and energy metabolism in diabetic hearts.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Additional information
Received 31 October 2008; received after revision 17 December 2008; accepted 2 January 2009
Rights and permissions
About this article
Cite this article
Lin, G., Brownsey, R.W. & MacLeod, K.M. Regulation of mitochondrial aconitase by phosphorylation in diabetic rat heart. Cell. Mol. Life Sci. 66, 919–932 (2009). https://doi.org/10.1007/s00018-009-8696-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-009-8696-3