Summary
Previous attempts to produce nonenzymatic template-directed oligomerizations of activated pyrimidines on polypurine templates have been unsuccessful. The only efficient reactions are those where the template is composed primarily of pyrimidines, especially cytosine. Because molecular evolution requires that a synthesized daughter polynucleotide be capable of acting as a template for the synthesis of the original polynucleotide, the one-way replication achieved thus far is inadequate to initiate an evolving system.
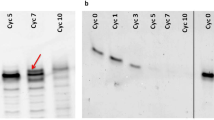
Several uracil analogs were used in this investigation in order to search for possible replacements for uracil. The monomers used in this investigation were the imidazolides of UMP, xanthosine 5′-monophosphate, the bis-monophosphates of the acyclic nucleosides of uracil, and 2,4-quinazolinedione. The concentrations of various salts, buffers, pH, and temperature were among the different variables investigated in attempts to find conditions that would permit template-directed oligomerizations. Although the different monomers in this study demonstrated varying abilities to form very short oligomers, we were unable to detect any enhancement of this oligomerization that could be attributed to the poly(A) template.
Although special conditions might be found that would allow purine-rich templates to work, these reactions cannot be considered robust. The results of our experiments suggest that pyrimidines were not part of the original replicating system on the primitive Earth. It has already been shown that ribose is an unlikely component of the first replicating systems, and we now suggest that phosphate was absent as well. This is due to the low solubility of phosphate in the present ocean (3×10−6 M), as well as the difficulty of prebiotic activation of phosphates.
Similar content being viewed by others
Abbreviations
- pN (N=A, C, U, X, or G):
-
the nucleoside 5′-phosphates of adenine, cytosine, uracil, xanthine, or guanine
- ImpN:
-
the 5′-phosphoimidazolide of N
- 2MeImpN:
-
the 5′-phospho-2-methylimidazolide of N
- \(\tilde U\),\(\tilde Q\) :
-
the acyclic nucleosides of U and Q {1-[1, 3-dihydroxy-2-propoxy)methyl]uracil}
- Q:
-
quinazoline-2,4-dione
- ImpÑpIm:
-
the bis-monophosphoimidazoides of Ñ
- poly(C):
-
polycytidylic acid
- poly(A):
-
polyadenylic acid
References
Cairns-Smith AG (1982) Genetic takeover and the mineral origins of life. Cambridge University Press, New York
Cheikh AB, Orgel LE (1990) Polymerization of amino acids containing nucleotide bases. J Mol Evol 30:315–321
Crick FHC (1968) Origin of the genetic code. J Mol Biol 38: 367–379
Ferris FP, Sanchez RA, Orgel LE (1968) Studies in prebiotic synthesis. III. Synthesis of pyrimidines from cyanoacetylene and cyanate. J Mol Biol 33:693–704
Hill AR Jr, Kumar S, Leonard NJ, Orgel LE (1988a) Templatedirected oligomerization of 3-isoadenosine 5′-phosphate. J Mol Evol 27:91–95
Hill AR Jr, Nord LD, Orgel LE, Robins RK (1988b) Cyclization of nucleotide analogues as an obstacle to polymerization. J Mol Evol 28:170–171
Inoue T, Orgel LE (1981) Substituent control of the poly(C)-directed oligomerization of guanosine 5′-phosphoroimidazolide. J Am Chem Soc 103:7666–7667
Inoue T, Orgel LE (1982) Oligomerization of (guanosine 5′-phosphor-)2-methylimidazolide on poly(C): an RNA polymerase model. J Mol Biol 162:201–217
Inoue T, Orgel LE (1983) A nonenzymatic RNA polymerase model. Science 219:859–862
Joyce GF, Orgel LE (1986) Non-enzymic template-directed synthesis on RNA random copolymers: poly(C,G) templates. J Mol Biol 188:433–441
Joyce GF, Orgel LE (1988) Non-enzymatic template-directed synthesis on RNA random copolymers: poly(C,A) templates. J Mol Biol 202:677–681
Joyce GF, Inoue T, Orgel LE (1984a) Non-enzymatic template-directed synthesis on RNA random copolymers: poly(C,U) templates. J Mol Biol 176:279–306
Joyce GF, Visser GM, van Boeckel CAA, van Boom JH, Orgel LE, van Westrenen J (1984b) Chiral selection in poly(C)-directed synthesis of oligo(G). Nature 310:602–604
Joyce GF, Schwartz AW, Miller SL, Orgel LE (1987) The case for an ancestral genetic system involving simple analogues of the nucleotides. Proc Natl Acad Sci USA 84:4398–4402
Lohrmann R, Orgel LE (1978) Preferential formation of (2′–5′)-linked internucleotide bonds in non-enzymatic reactions. Tetrahedron 34:853–855 [See also errata, 35:566 (1979].
Martens CS, Harriss RC (1970) Inhibition of apatite precipitation in the marine environment by magnesium ions. Geochim Cosmochim Acta 34:621–625
Miller ML, Orgel LE (1974) The origins of life on the earth. Prentice-Hall, Englewood Cliffs NJ
Ninio J, Orgel LE (1978) Heteropolynucleotides as templates for non-enzymatic polymerizations. J Mol Evol 12:91–99
Ogilvie KK, Hamilton RG, Gillen MF, Radatus BK, Smith KO, Galloway KS (1984) Uracil analogues of the acyclonucleoside 9-[[2-hydroxy-1-(hydroxymethyl)ethoxy]-methyl]guanine (BIOLF-62). Can J Chem 62:16–21
Orgel LE (1986) RNA catalysis and the origins of life. J Theor Biol 123:127–149
Piccirilli JA, Krauch T, Moroney SE, Benner SA (1990) Enzymatic incorporation of a new base pair into DNA and RNA extends the genetic alphabet. Nature 343:33–37
Pitha J (1977) Vinyl polymer analogues of nucleic acids. Polymer 18:425–429
Sawai H (1988a) Efficient oligoadenylate synthesis catalyzed by uranyl ion complex in aqueous solution. Nucleic Acids Symp Ser 19:5–7
Sawai H (1988b) Oligonucleotide formation catalyzed by divalent metal ions. The uniqueness of the ribosyl system. J Mol Evol 27:181–186
Schneider KC, Benner SA (1990) Oligonucleotides containing flexible nucleoside analogues. J Am Chem Soc 11:453–455
Schwartz AW, Orgel LE (1985) Template-directed synthesis of novel nucleic acid-like structures. Science 228:585–587
Shapiro R (1986) Origins—a skeptic's guide to the creation of life on earth. Summit Books, New York
Shapiro R (1988) Prebiotic ribose synthesis: a critical analysis. Origins Life Evol Biosphere 18:71–85
Shapiro R, Klein RS (1966) The deamination of cytidine and cytosine by acidic buffer solutions. Mutagenic implications. Biochemistry 5:2358–2362
Stribling R, Miller SL (1991) Template-directed synthesis of oligonucleotides under eutectic conditions. J Mol Evol 32: 289–295
Sulston J, Lohrmann R, Orgel LE, Miles HT (1968a) Nonenzymatic synthesis of oligoadenylates on a polyuridylic acid template. Proc Natl Acad Sci USA 59:726–733
Sulston J, Lohrmann R, Orgel LE, Miles HT (1968b) Specificity of oligonucleotide synthesis directed by polyuridylic acid. Proc Natl Acad Sci USA 60:409–415
Sulston J, Lohrmann R, Orgel LE, Schneider-Bernloehr H, Weimann BJ, Miles HT (1969) Non-enzymic oligonucleotide synthesis on a polycytidylate template. J Mol Biol 40:227–234
Ts'o POP (1974) Basic principles in nucleic acid chemistry, vols I and II. Academic Press, New York
Visscher J, Schwartz AW (1988) Template-directed synthesis of acyclic oliogonucleotide analogues. J Mol Evol 28:3–6
Visscher J, Schwartz AW (1990) Oligomerization of cytosine-containing nucleotide analogs in aqueous solution. J Mol Evol 30:3–6
Wächtershäuser G (1988) An all-purine precursor of nucleic acids. Proc Natl Acad Sci USA 85:1134–1135
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stribling, R., Miller, S.L. Attempted nonenzymatic template-directed oligomerizations on a polyadenylic acid template: Implications for the nature of the first genetic material. J Mol Evol 32, 282–288 (1991). https://doi.org/10.1007/BF02102185
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02102185