Abstract
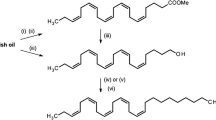
The major component of the sex pheromone of femaleSpodoptera littoralis, (Z,E)-9,11-tetradecadienyl acetate (1), elicited all steps of the male behavioral sequence, i.e., wing fanning and taking flight, oriented upwind flight and arrival to the middle of the tunnel, close approach and contact with the source. The activity was equivalent to that elicited by virgin females. In the range of doses tested, the dosage of1 had no significant effect on the number of source contacts. Male response was significantly affected by light intensity, being optimum at 3 lux. Activity of the minor components (Z)-9-tetradecenyl acetate (2), (E)-11-tetradecenyl acetate (3), tetradecyl acetate (4), (Z)-11-tetradecenyl acetate (5), and (Z,E)-9,12-tetradecadienyl acetate (6) was significantly lower than that of the major component when assayed individually. In multicomponent blends compound4 appeared to strongly decrease the number of males arrested at the source, the effect being particularly important when compound5 was present in the blend. Results of single sensillum experiments confirmed the existence of two main physiologically distinct sensillar types. The most common type of sensilla contained a neuron that responded specifically to compound1. A second type of sensilla, located laterally on the ventral sensory surface, contained two receptor neurons responding to compound6 and to (Z)-9-tetradecenol. Among short sensilla, one hair responded to compound4 and could represent a minor sensillar type. No sensory neuron was found to detect the other minor pheromone compounds2, 3, and5.
Similar content being viewed by others
References
Baker, T. C. andCardé, R. T. 1984. Techniques for behavioral bioassays, pp. 45–73,in H. E. Hummel and T. A. Miller (eds.). Techniques in Pheromone Research. Springer-Verlag, New York.
Campion, D. G., Hunter-Jones, P., McVeigh, L. J., Hall, D. R., Lester, R., andNesbitt, B. F. 1980. Modification of the attractiveness of the primary pheromone component of the Egyptian cotton leafwormSpodoptera littoralis (Boisduval) (Lepidoptera, Noctuidae) by secondary pheromone components and related chemicals.Bull. Entomol. Res. 70:417–434.
Cardé, R. T. 1984. Chemo-orientation in flying insects, pp. 111–124,in W. J. Bell and R. T. Cardé (eds.). Chemical Ecology of Insects. Chapman and Hall, London.
Charlton, R. E., Kanno, H., Collins, R. D., andCardé, R. T. 1993. Influence of pheromone concentration and ambient temperature on flight of the gypsy mothLymantria dispar (l.) in a sustained-flight wind tunnel.Physiol. Entomol. 18:349–362.
David, C. T. 1982. Competition between fixed and moving stripes in the control of orientation by flyingDrosophila.Physiol. Entomol. 7:151–156.
Dunkelblum, E., Kehat, M., Gothilf, S., Greenberg, S., andSklarsz, B. 1982. Optimized mixture of sex pheromonal components for trapping of maleSpodoptera littoralis in Israel.Phytoparasitica 10:21–26.
Dunkelblum, E., Kehat, H., Harel, M., andGordon, D. 1987. Sexual behaviour and pheromone titre of theSpodoptera littoralis female moth.Entomol. Exp. Appl. 44:241–247.
Frérot, B., Lucas, P., andRochat, D. 1989. Responses ofMamestra suasa male moths to synthetic pheromone compounds in a wind tunnel.Entomol. Exp. Appl. 53:81–87.
Haines, L. C. 1983. Wind tunnel studies on the effects of secondary sex pheromone components on the behaviour of male Egyptian cotton leafwormSpodoptera littoralis.Physiol. Entomol. 8:29–40.
Kaissling, K. E. 1979. Recognition of pheromones by moths, especially in saturniids andBombyx mori, pp. 43–56,in F. J. Ritter (ed.). Chemical Ecology: Odour Communication in Animals. Elsevier/North Holland, Amsterdam.
Kaissling, K. E., andThorson, J. 1980. Insect olfactory sensilla: structural, chemical and electrical aspects of the functional organization, pp. 261–282,in D. B. Satelle, L. M. Hall, and J. G. Hildebrand (eds.). Receptors for Neurotransmitters, Hormones and Pheromones in Insects. Elsevier/North Holland, Amsterdam.
Kawasaki, K. 1986. Activity rhythms and behavior of adultSpodoptera litura F. (Lepidoptera, Noctuidae) at night: Factors determining male attraction time by females.Appl. Entomol. Zool. 21:493–499.
Kehat, M., Greenberg, S., andTamaki, Y. 1976. Field evaluation of the synthetic sex pheromone, as an attractant for males of the cottom leafwormSpodoptera littoralis (Boisd.) in Israel.Appl. Entomol. Zool. 11:45–52.
Linn, C. E., andGaston, L. K. 1981. Behavioural responses of maleTrichoplusia ni in a sustained-flight tunnel to the two sex pheromone components.Environ. Entomol. 10:379–385.
Linn, C. E., andRoelofs, W. L. 1983. Effect of varying proportions of the alcohol component on sex pheromone blend discrimination in male Oriental fruit moths.Physiol. Entomol. 8:291–306.
Linn, C. E. J., Campbell, M. G., andRoelofs, W. L. 1987. Pheromone components and active spaces: What do moths smell and where do they smell it?Science 237:650–652.
Linn, C. E., Campbell, M. G., andRoelofs, W. L. 1988. Temperature modulation of behavioural thresholds controlling male moth sex pheromone response specificity.Physiol. Entomol. 13:59–67.
Liungberg, H., Anderson, P., andHansson, B. S. 1993. Physiology and morphology of pheromone-specific sensilla on the antennae of male and femaleSpodoptera littoralis.J. Insect Physiol. 39:253–260.
Löfstedt, C., Van der Pers, J. N. C., Lofqvist, J., Lanne, B. S., Appelgren, M., Bergström, G., andThelin, B. 1982. Sex pheromone components of the turnip mothAgrotis segetum. Chemical identification, electrophysiological evaluation and behavioral activity.J. Chem. Ecol. 8:1305–1321.
Löfstedt, C., Linn, C. E. J., andLöfqvist, J. 1985. Behavioral responses of male turnip mothAgrotis segetum to sex pheromone in a flight tunnel and in the field.J. Chem. Ecol. 11:1209–1221.
Mafra-Neto, A., andCardé, R. T. 1994. Fine-scale structure of pheromone plumes modulates upwind orientation of flying moths.Nature 369:142–144.
Martinez, T., Fabriás, G., andCamps, F. 1990. Sex pheromone biosynthetic pathway inSpodoptera littoralis and its activation by a neurohormone.J. Biol. Chem. 265:1381–1387.
Miller, J. R., andRoelofs, W. L. 1978. Sustained-flight tunnel for measuring insect responses to windborne sex pheromones.J. Chem. Ecol. 4:187.
Nesbitt, B. F., Beevor, P. S., Hall, D. R., Lester, R., andPoppi, R. G. 1973. Sex pheromones of two noctuid moths.Nature (London) New Biol. 244:208–209.
Poitout, S., Bues, R., andLe Rumeur, C. 1972. Élevage sur milieu artificiel simple de deux noctuelles parasites du cotonEarias insulana etSpodoptera littoralis.Entomol. Exp. Appl. 15:341–350.
Preiss, R., andPriesner, E. 1988. Responses of male codling moths (Laspeyresia pomonella) to codlemone and other alcohols in a wind tunnel.J. Chem. Ecol. 14:797–813.
Preisner, E. 1979. Specificity studies on pheromone receptors in noctuid and tortricid Lepidoptera, pp. 57–71,in F. J. Ritter (ed.). Chemical Ecology: Odour Communication in Animals, Elsevier/North Holland, Amsterdam.
Ridgway, R. L., Silverstein, R. M., andInscoe, M. N. (eds.). 1990. Behavior-Modifying Chemicals for Insect Management. Marcel Dekker, New York.
Sanders, C. J. 1982. Disruption of male spruce budworm orientation to calling females in a wind tunnel by synthetic pheromone.J. Chem. Ecol. 8:493–506.
Sokal, R. R., andRohlf, F. G. (eds.). 1969. Biometry. W. H. Freeman & Co., San Francisco.
Tamaki, Y., andYushima, T. 1974. Sex pheromone of the cotton leafwormSpodoptera littoralis.J. Insect Physiol. 20:1005–1014.
Author information
Authors and Affiliations
Additional information
Dedicated to the memory of the late Prof. Félix Serratosa (1925–1995).
Rights and permissions
About this article
Cite this article
Quero, C., Lucas, P., Renou, M. et al. Behavioral responses ofSpodoptera littoralis males to sex pheromone components and virgin females in wind tunnel. J Chem Ecol 22, 1087–1102 (1996). https://doi.org/10.1007/BF02027947
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02027947