Summary
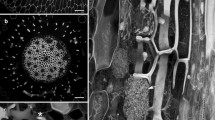
Following a 5 hours ethylene treatment, cortical cells of Pea (Pisum sativum L. var Alaska) epicotyl third internode showed a change in the orientation of both microtubules near the plasma membrane and recently deposited cellulose microfibrils. Control cortical cells had mostly transverse microtubules. The ratio of the average frequency of transverse to longitudinal microtubules was 6.0. After 5 hours of ethylene treatment, cortical cells had mostly longitudinal microtubules, with the ratio of transverse to longitudinal microtubules equal to 0.1. Epidermal cells were more variable than cortical cells with regard to the frequency of longitudinal and transverse microtubules. Observation of cortical cell walls in conventionally stained thin sections revealed that recent deposition of microfibrils had been primarily transverse in almost all of the control cortical cells sampled. In contrast, more than half of the ethylene-treated cortical cells had recent deposition oriented primarily longitudinally. This change in microtubule and microfibril orientation may be early enough to constitute the primary effect of ethylene leading to radial cell expansion.
Similar content being viewed by others
References
Apelbaum, A., Burg, S. P., 1971: Altered cell microfibrillar orientation in ethylene-treatedPisum sativum stems. Plant Physiol.48, 648–652.
Burg, S. P., Burg, E. A., 1966: The interaction between auxin and ethylene and its role in plant growth. Proc. Nat. Acad. Sci. (USA)55, 262–296.
— —, 1968: Ethylene formation in pea seedlings: its relation to the inhibition of bud growth caused by indole-3-acetic acid. Plant Physiol.43, 1069–1074.
Chafe, S. C., Wardrop, A. B., 1972: Fine structural observations on the epidermis. I. The epidermal cell wall. Planta107, 269–278.
Eisinger, W. R., Burg, S. P., 1972: Ethylene-induced pea internode swelling: its relation to ribonucleic acid metabolism, wall protein synthesis and cell wall structure. Plant Physiol.50, 510–517.
Frey-Wyssling, A., 1976: The plant cell wall. Berlin: Borntraeger.
Green, P. B., 1963: On mechanisms of elongation. In: Cytodifferentiation and macromolecular synthesis (Locke, M., ed.). New York: Academic Press.
Hardham, A. R., Green, P. B., Lang, J. M., 1980: Reorganization of cortical microtubules and cellulose deposition during leaf formation inGraptopetalum paraguayense. Planta149, 181–195.
Henry, E. W., 1978: An ultrastructural study of ethylene-treated stem segments of Alaska pea (Pisum sativum). Cytologia43, 423–432.
Hepler, P. K., Palevitz, B. A., 1972: Microtubules and microfilaments. Ann. Rev. Plant Physiol.25, 309–362.
Hogetsu, T., Shibaoka, H., 1978: Effects of colchicine on cell shape and on microfibril arrangement in the cell wall ofClosterium acerosum. Planta140, 15–18.
Nee, M., Chiu, L., Eisinger, W., 1978: Induction of swelling in pea internode tissue by ethylene. Plant Physiol.62, 902–906.
Newcomb, E. H., Bonnet, H. T., 1965: Cytoplasmic microtubule and wall microfibril orientation in root hairs of radish. J. Cell. Biol.27, 575–589.
Palevitz, B. A., Hepler, P. K., 1976: Cellulose microfibril orientation and cell shaping in developing guard cells ofAllium: The role of microtubules and ion accumulation. Planta132, 71–93.
Prat, R., Roland, J.-C., 1980: Croissance différentielle des tissues et texture des parois. Physiol. Veg.18, 241–257.
Preston, R. D., 1974: The physical biology of plant cell walls. London: Chapman and Hall.
—,Astbury, W. T., 1937: The structure of the wall of the green algaValonia ventricosa. Proc. Roy. Soc., Ser. B.122, 76–97.
—,Ripley, G. W., 1954: Electron diffraction diagrams of cellulose microfibrils inValonia. Nature174, 76–77.
Ray, P. M., 1967: Radioautographic study of cell wall deposition in growing plant cells. J. Cell. Biol.35, 659–674.
Richmond, P. A., 1977: Control of plant cell morphogenesis by the cell wall: analysis inNitella. Ph.D. thesis. University of Pennsylvania, Philadelphia.
—,Metraux, J.-P., Taiz, L., 1980: Cell expansion patterns and directionality of wall mechanical properties inNitella. Plant Physiol.65, 211–217.
Ridge, I., 1973: The control of cell shape and rate of cell expansion by ethylene: effects on microfibril orientation and cell wall extensibility in etiolated peas. Acta Bot. Neerl.22, 144–158.
Robards, A. W., Kidwai, P., 1972: Microtubules and microfibrils in xylem fibres during secondary cell wall formation. Cytobiologie6, 1–21.
Roelofson, P. A., 1959: The plant cell wall. Berlin: Borntraeger.
Roland, J.-C., Vian, B., 1979: The wall of the growing plant cell: its three dimensional organization. Int. Rev. Cytol.61, 129–166.
Sawhney, V. K., Srivastava, L. M., 1975: Wall fibrils and microtubules in normal and gibberellic-acid-induced growth of lettuce hypocotyl cells. Can. J. Bot.53, 824–835.
Shibaoka, H., 1974: Involvement of wall microtubules in gibberellin promotion and kinetin inhibition of stem elongation. Plant Cell Physiol.15, 255–263.
Sokal, R. R., Rohlf, F. J., 1969: Biometry; the principles and practice of statistics in biological research. San Francisco: W. H. Freeman.
Steen, D. A., Chadwick, A. V., 1981: Ethylene effects in pea stem tissue, evidence of microtubule mediation. Plant Physiol.67, 460–466.
Takeda, K., Shibaoka, H., 1981a: Changes in microfibril arrangement on the inner surface of the epidermal cell walls in the epicotyl ofVigna angularis Ohwi et Ohashi during cell growth. Planta151, 385–392.
— —, 1981b: Effects of gibberellin and colchicine on microfibril arrangement in epidermal cell walls ofVigna angularis Ohwi et Ohashi epicotyls. Planta151, 393–398.
Went, F. W., 1934: On the pea test method for auxin, the plant growth hormone. K. Akad. Wetenschap. Amsterdam. Proc. Sect. Sci.37, 547–555.
Author information
Authors and Affiliations
Additional information
Research supported by NSF grant PCM 78-03244, A1, 2 to PBG and by a Research Corporation grant to WRE.
Rights and permissions
About this article
Cite this article
Lang, J.M., Eisinger, W.R. & Green, P.B. Effects of ethylene on the orientation of microtubules and cellulose microfibrils of pea epicotyl cells with polylamellate cell walls. Protoplasma 110, 5–14 (1982). https://doi.org/10.1007/BF01314675
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01314675