Summary
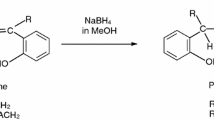
Partial displacement of the symmetrical bis-Schiff bases (1)–(4) of ethylenediamine and salicyladehyde, o-hydroxyacetophenone, acetylacetone or benzoylacetone by dehydroacetic acid have led to the formation and isolation of the unsymmetrical bis-Schiff base ligands (6)–(9). Attempted partial displacements of the symmetrical bis-Schiff base (5) of dehydroacetic acid or (1)–(4) by each of the other four carbonyl compounds have been unsuccessful. The Ni(II) and Cu(II) complexes of (6)–(9) have been prepared and characterized.
Similar content being viewed by others
References
K. Ueno and A. E. Martell,J. Phys. Chem.,59, 998 (1955); ibid J. Phys. Chem.,60, 1270 (1956).
P. J. McCarthy, R. J. Hovey, K. Ueno and A. E. Martell,J. Am. Chem. Soc.,77, 5820 (1955).
R. H. Bailes and M. Calvin,J. Am. Chem. Soc.,69, 1886 (1947).
M. J. Carter, D. Paul Rillema and F. Basolo,J. Am. Chem. Soc.,96, 392 (1974).
S. F. Tan, K.-P. Ang and H. Jayachandran,Transition Met. Chem.,9, 390 (1984).
V. Doraswamy and P. K. Bhattacharya,Indian J. Chem.,15A, 129 (1977); V. B. Mohan Kumar and P. K. Bhattacharya,ibid Indian J. Chem.,15A, 132 (1977).
R. P. Sharma and R. N. Prasad,Synth. React. Inorg. Met-Org. Chem.,12, 347 (1982).
P. J. Burke and D. R. McMillin,J. Chem. Soc. Dalton Trans., 1794 (1980).
W. P. Jencks, in S. G. Cohen, A. Stireitweiser & R. W. Taft, (Ed.)in Physical Organic Chemistry, vol. 2, Interscience Publishers, New York, 1964, pp. 63–128.
E. H. Cordes and W. P. Jencks,J. Am. Chem. Soc.,84, 826 (1962).
B. Witkop and T. W. Beiler,J. Am. Chem. Soc.,76, 5589 (1954).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tan, SF., Ang, KP. Unsymmetrical bis-Schiff bases derived from ethylenediamine, dehydroacetic acid and another aldehyde/ketone and the preparation and characterisation of their copper(II) and nickel(II) complexes. Transition Met Chem 13, 64–68 (1988). https://doi.org/10.1007/BF01041502
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01041502