Summary
-
1.
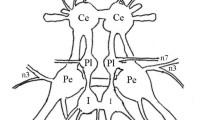
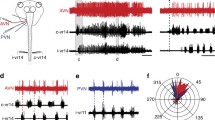
Intracellular stimulation of each of three different types of mechanoreceptors, the T, P and N cells, evokes swimming behavior in leech preparations. Stimulation of an individual N cell or P cell evoked swimming in 75% and 53% respectively, of the preparations tested. Stimulation of an individual T cell was ineffective in eliciting swimming; however, simultaneous stimulation of two T cells evoked swimming in 59% of our preparations.
-
2.
Stimulation of mechanosensory neurons elicited swimming activity for a limited number of trials; i.e. the response habituated. The number of swim episodes evoked before habituation to criterion did not differ significantly for the different types of mechanoreceptors.
-
3.
The duration of swim episodes declined significantly over the course of N cell stimulation. The tendency for swim length to decline with repeated stimulation was present as well for swim episodes elicited by P or T cell stimulation.
-
4.
Swim initiation recovered spontaneously following habituation resulting from T cell stimulation. Spontaneous recovery following N cell stimulation was not demonstrated. However, N cell stimulation evoked swimming again after DP nerve shock or to a limited extent, after cell 204 stimulation. Spontaneous recovery of swim initiation to P cell stimulation was not investigated.
-
5.
A previous study detailed habituation of swimming activity to mechanical stimulation of the body wall (Debski and Friesen 1985). Only the T cells are activated significantly by this stimulus. Stimulation of sensory receptors other than mechanoreceptors was not effective in eliciting swimming in our preparation. We conclude that T cells mediate swim initiation elicited by stroking of the body wall and that the cessation of swimming to this stimulus is not due to sensory adaptation.
Similar content being viewed by others
References
Baylor DA, Nicholls JG (1969) Chemical and electrical synaptic connexions between cutaneous mechanoreceptor neurones in the central nervous system of the leech. J Physiol 203:591–609
Blackshaw SE, Nicholls JG, Parnas I (1982) Physiological responses, receptive fields and terminal arborizations of nociceptive cells in the leech. J Physiol 326:251–260
Brodfuehrer PD, Friesen WO (1984) A sensory system initiating swimming activity in the medicinal leech. J Exp Biol 108:341–355
Debski EA, Friesen WO (1985) Habituation of swimming activity in the medicinal leech. J Exp Biol 116:169–188
Debski EA, Friesen WO (1986) Role of central interneurons in habituation of swimming activity in the medicinal leech. J Neurophysiol 55:977–994
Friesen WO (1981) Physiology of water motion detection in the medicinal leech. J Exp Biol 92:255–275
Friesen WO (1985) Neuronal control of leech swimming movements: interactions between cell 60 and previously described oscillator neurons. J Comp Physiol A 156:231–242
Friesen WO, Poon M, Stent GS (1978) Neuronal control of swimming in the medicinal leech. IV. Identification of a network of oscillatory interneurones. J Exp Biol 75:25–43
Gillon JW, Wallace BG (1984) Segmental variation in the arborization of identified neurons in the leech central nervous system. J Comp Neurol 228:142–148
Johansen J, Hockfield S, McKay RDG (1984) Distribution and morphology of nociceptive cells in the CNS of three species of leeches. J Comp Neurol 226:263–273
Johansen J, Yang J, Kleinhaus AL (1984) Actions of procaine on specific nociceptive cells in leech central nervous system. J Neurosci 4:1253–1261
Kretz JR, Stent GS, Kristan WB Jr (1976) Photosensory input pathways in the medicinal leech. J Comp Physiol 106:1–37
Kristan WB Jr, Calabrese RL (1976) Rhythmic swimming activity in neurones of the isolated nerve cord of the leech. J Exp Biol 65:643–668
Kristan WB Jr, Stent GS (1976) Peripheral feedback in the leech swimming rhythm. Cold Spring Harbor Symp Quant Biol 40:663–674
Kristan WB Jr, McGirr SJ, Simpson GV (1982) Behavioral and mechanosensory neurone response to skin stimulation in leeches. J Exp Biol 96:143–160
Mann KM (1962) Leeches (Hirudinea). Their structure, physiology, ecology and embryology. Pergamon Press, New York, pp 79–100
McKay R, Hockfield S, Johansen J, Kleina L, Thompson I (1982) Monoclonal antibodies as probes of the leech nervous system. Soc Neurosci Abstr 8:714
Nicholls JG, Baylor DA (1968) Specific modalities and receptive fields of sensory neurons in the CNS of the leech. J Neurophysiol 31:740–756
Nusbaum MP, Kristan WB Jr (1986) Swim initiation in the leech by serotonin-containing interneurones, cells 21 and 61. J Exp Biol 122:277–302
Ort CA, Kristan WB Jr, Stent GS (1974) Neuronal control of swimming in the medicinal leech. II. Identification and connections of motor neurons. J Comp Physiol 94:121–154
Phillips CE, Friesen WO (1982) Ultrastructure of the watermovement-sensitive sensilla in the medicinal leech. J Neurobiol 13:473–486
Stent GS, Kristan WB Jr, Friesen WO, Ort CA, Poon M, Calabrese RL (1978) Neuronal generation of the leech swimming movement. Science 200:1348–1357
Weeks JC (1982) Synaptic basis of swim initiation in the leech. II. A pattern-generating neuron (cell 208) which mediates motor effects of swim-initiation neurons. J Comp Physiol 148:265–279
Weeks JC, Kristan WB Jr (1978) Initiation, maintenance and modulation of swimming in the medicinal leech by the activity of a single neurone. J Exp Biol 77:71–88
Willard A (1981) Effects of serotonin on the generation of the motor program for swimming by the medicinal leech. J Neurosci 1:936–944
Yau K-W (1976) Receptive fields, geometry and conduction block of sensory neurones in the central nervous system of the leech. J Physiol (Lond) 263:489–512
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Debski, E.A., Friesen, W.O. Intracellular stimulation of sensory cells elicits swimming activity in the medicinal leech. J. Comp. Physiol. 160, 447–457 (1987). https://doi.org/10.1007/BF00615078
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00615078