Abstract
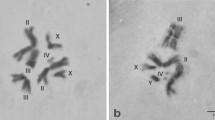
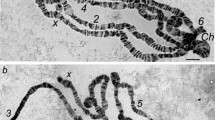
We hybridized cloned DNA segments to salivary gland polytene chromosomes of the medfly,Ceratitis capitata, and thus established molecular markers for 24 sites on 6 out of 10 autosomal arms. An additional marker identified a medfly repetitive element that hybridizes to approximately 100 autosomal sites as well as a granular network that is thought to represent theX chromosome. Some of the markers correspond to 9 characterized transcription units, while 17 remain anonymous; at least 3 of the latter are restriction fragment length polymorphism (RFLP) markers. The characterized transcription units document that chromosomal arm 5L ofC. capitata is homologous to theDrosophila melanogaster X chromosome, in agreement with previous inferences based on the extensive conservation of linkage groups in Diptera.
Similar content being viewed by others
References
Anonymous (1987) Medfly genetics. Informational Circular, Joint FA/IAEA Division, Vienna, Austria
Bedo DG (1982) Differential sex chromosome replication and dosage compensation in polytene trichogen cells ofLucillia cuprina (Diptera: Calliphoridae). Chromosoma 87: 21–32
BedoDG (1986) Polytene and mitotic chromosome analysis inCeratitis capitata (Diptera: Tephritidae). Can J Gen Cytol 28: 180–188
BedoDG (1987) Polytene chromosome mapping inCeratitis capitata (Diptera: Tephritidae). Genome 29: 598–611
BedoDG, ZacharopoulouA (1988) Inter-tissue variability of the polytene chromosome banding. Trends Genet 4: 90–91
BeverlySM, WilsonAC (1984) Molecular evolution inDrosophila and the higher Diptera. II. A time scale for fly evolution. J Mol Evol 21: 1–13
BrownNH, KingDL, WilcoxM, KafatosFC (1989) Alternative splicing ofDrosophila integrinPS2α transcripts. Cell 59: 185–195
ChurchGM, GilbertW (1984) Genomic sequencing. Proc Natl Acad Sci USA 81: 1991–1995
FeinbergAP, VogelsteinB (1983) A technique for radiolabelling DNA restriction endonuclease fragments to high specific activity. Anal Biochem 132: 6–13
FeinbergAP, VogelsteinB (1984) A technique for radiolabelling DNA restriction endonuclease fragments to high specific activity, addendum. Anal Biochem 137: 266–267
FosterGG, WhittenMJ, KonovalovC, ArnoldSTA, MaffiG (1981) Autosomal genetic maps of the Australian Sheep Blowfly,Lucillia cuprina dorsalis R-D (Dipteral, Calliphoridae) and possible correlations with the linkage maps ofMusca domestica L, andDrosophila melanogaster (Mg). Genet Res 37: 55–69
GarabedianMJ, ShirrasAD, BownesM, WensinkPC (1987) The nucleotide sequence of the gene coding forDrosophila melanogaster yolk protein 3. Gene 55: 1–8
Gasperi G, Guglielmino CR, Malacrida AR, Milani R (1991) Genetic variability and gene flow in geographic populations of the medfly,Ceratitis capitata. Heredity 67 (in press)
HarrisEJ (1989) Pest status of fruit flies. In: RobinsonAS, HooperGH (eds) Fruit flies, their biology, natural enemies and control. Elsevier, Amsterdam, pp 73–81
HatzopoulosP, KambysellisMP (1987) Isolation and structural analysis ofDrosophila grimshawi vitellogenin genes. Mol Gen Genet 206: 475–484
HaymerDS, AntleinerJE, HeM, ThanaphumS, SaulSH, IvyJ, HoutchensK, ArcangeliL (1990) Actin genes in the Mediterranean fruit fly,Ceratitis capitata. Genetics 125: 155–160
HungM-C, WensinkPC (1983) Sequence and structure conservation in yolk proteins and their genes. J Mol Biol 164: 481–492
KerremansPh, BourtzisK, ZacharopoulouA (1990) Cytogenetic analysis of three genetic sexing strains ofCeratitis capitata. Theor Appl Genet 80: 177–182
KonsolakiM, KomitopoulouK, ToliasPP, KingDL, SwimmerC, KafatosFC (1990) The chorion genes of the medfly,Ceratitis capitata. I. Structural and regulatory conservation of thes36 gene relative to twoDrosophila species. Nucleic Acids Res 18: 1731–1737
LoukasM, KafatosFC (1986) The actin loci in the genusDrosophila: establishment of chromosomal homologies among distantly related species by in situ hybridization. Chromosoma 94: 297–308
LoukasM, KafatosFC (1988) Chromosomal locations of actin genes are conserved between themelanogaster andobscura groups ofDrosophila. Genetica 76: 33–41
MalacridaA, GasperiG, BiscaldiGF, MilaniR (1984) Functional significance of gene clusters in the housefly,Musca domestica and in other Diptera. Atti 2nd Congr Nat Soc Ital Ecol SItE 5: 307–310
MalacridaA, GasperiG, BiscaldiGF, MilaniR (1986) Persistence of linkage relationships among enzyme loci in some Dipteran species. Atti Assoc Genet Ital 31: 121–122
ManiatisT, FritschEF, SambrookJ (1982) Molecular cloning: A laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Martínez-CruzadoJ-C, SwimmerC, FenerjianMG, KafatosFC (1988) Evolution of the autosomal chorion locus inDrosophila I: General organization of the locus and sequence comparisons of geness15 ands19 in evolutionarily distant species. Genetics 119: 663–667
MiklosGLG, YamamotoMT, DaviesJ, PirrottaV (1988) Micro-cloning reveals a high frequency of repetitive sequences characteristic of chromosome 4 and the β-heterochromatin ofDrosophila melanogaster. Proc Natl Acad Sci USA 85: 2051–2055
RinaM, SavakisC (1991) A cluster of vitellogenin genes in the Mediterranean fruit flyCeratitis capitata: sequence and structural conservation in Dipteran yolk proteins and their genes. Genetics 127: 769–778
RobinsonAS, VanHeemertC (1982)Ceratitis capitata — a suitable case for genetic sexing. Genetica 58: 224–232
RösslerY (1979) Automated sexing ofCeratitis capitata: the development of strains with inherited, sex-limited pupal colour dimorphism. Entomophaga 24: 411–416
SaulSH (1985) Diagnosing the heterogametic sex in the Mediterranean fruitfly (Diptera: Tephritidae): The first sex-linked gene. Ann Entomol Soc Am 78: 198–200
SaulSH, RösslerY (1984) Genetic markers of the autosomal linkage groups of the Mediterranean fruitflyCeratitis capitata (Diptera: Tephritidae). Ann Entomol Soc Am 77: 323–327
SaundersRDC, GloverDM, AshburnerM, Sidén-KiamosI, LouisC, MonastiriotouM, SavakisC, KafatosFC (1989) PCR amplification of DNA microdissected from a single polytene chromosome band: a comparison with conventional microcloning. Nucleic Acids Res 17: 9027–9029
SpradlingAC, MahowaldAP (1980) Amplification of genes for chorion proteins during oogenesis inDrosophila melanogaster. Proc Natl Acad Sci USA 77: 1096–1100
SteinemannM (1982) Analysis of chromosomal homologies between two species of the subgenusSophophora: D. miranda andD. melanogaster using cloned DNA segments. Chromosoma 87: 77–88
SteinemannM, PinskerW, SperlichD (1984) Chromosomal homologies within theDrosophila obscura group probed by in situ hybridization. Chromosoma 91: 46–53
SturtevantAH, NovitskiE (1941) The homologies of the chromosome elements in the genusDrosophila. Genetics 26: 517–541
ToliasPP, KonsolakiM, KomitopoulouK, KafatosFC (1990) The chorion genes of the medfly,Ceratitis capitata. II. Characterization of three novel cDNA clones obtained by differential screening of an ovarian library. Dev Biol 140: 105–112
WhitingJHJr, PlileyMD, FarmerJL, JefferyDE (1989) In situ hybridization analysis of chromosomal homologies inDrosophila melanogaster andDrosophila virilis. Genetics 122: 99–109
YanYL, KunertCJ, PostlethwaitJH (1987) Sequence homologies among the three yolk polypeptide (Yp) genes inDrosophila melanogaster. Nucleic Acids Res 15: 67–85
ZacharopoulouA (1987) Cytogenetic analysis of mitotic and salivary gland chromosomes in the medflyCeratitis capitata. Genome 29: 67–71
ZacharopoulouA (1990) Polytene chromosome maps in the medflyCeratitis capitata. Genome 33: 184–197
ZacharopoulouA, BourtzisK, KerremansPh (1991 a) A comparison of polytene chromosomes in salivary glands and orbital bristle trichogen cells inCeratitis capitata. Genome 34: 215–219
Zacharopoulou A, Riva ME, Malacrida A, Gasperi G (1991 b) Cytogenetic characterization of a genetic sexing strain inCeratitis capitata. Genome 34 (in press)
Author information
Authors and Affiliations
Additional information
by P.B. Moens
Rights and permissions
About this article
Cite this article
Zacharopoulou, A., Frisardi, M., Savakis, C. et al. The genome of the Mediterranean fruitflyceratitis capitata: Localization of molecular markers by in situ hybridization to salivary gland polytene chromosomes. Chromosoma 101, 448–455 (1992). https://doi.org/10.1007/BF00582839
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00582839