Abstract
The discovery of a new maize (Zea mays L.) transposon system, Mutator, and the cloning of the 1.4 kilobase transposon, Mul, have made feasible the isolation of nuclear photosynthetic genes which are recognized only by their mutant phenotype. Mutant maize plants which express a high chlorophyll fluorescent (hcf) phenotype due to a defect in the electron transport or photophosphorylation apparatus have been isolated following mutagenesis with an active Mutator stock. The affected genes and their products in these mutants are inaccessible to classical methods of analysis. However, mutagenesis with the Mutator transposon makes it possible to isolate these genes.
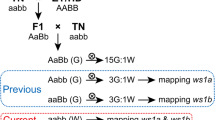
Although the PSII-deficient mutant hcf3 has been thoroughly studied by classical photo-biological methods, the nature of the lesion which results in the observed phenotype has not been established. A Mutator-induced allele of hcf3 has been isolated. A fragment of genomic DNA has been identified which is homologous to Mul and co-segregates with the mutant phenotype. This fragment is expected to contain a portion of the hcf3 locus which will be used to clone the normal gene. Direct study of the gene can provide insight into the nature and function of its polypeptide product.
This approach can be used to study any photosynthetic gene which has been interrupted by a transposon. The isolation of more than 100 different chemically-induced hcf mutants, most of which can not be fully characterized using classical means, indicates the wealth of information which can be obtained using a transposon tagging technique.
Similar content being viewed by others

References
Alleman M and Freeding M (1986) The Mu transposable elements of maize: Evidence for transposition and copy number regulation during development. Genetics 112: 107–119
Baker B, Schell J, Lorz H and Federoff NV (1986) Transposition of the maize controlling element ‘Activator’ in tobacco. Proc Natl Acad Sci USA 83: 4844–4848
Barkan A, Miles D and Taylor WC (1986) Chloroplast gene expression in nuclear, photosynthetic mutants in maize. EMBO J 5: 1421–1427
Barker RF, Thompson DV, Talbot DR, Swanson J and Bennetzen JL (1984) Nucleotide sequence of the maize transposable element Mul. Nucleic Acids Res 12: 5955–5967
Beckett JB (1978) B-A translocation in maize. I. Use in locating genes by chromosome arms. J Heredity 69: 27–36.
Bennetzen JL, Swanson J, Taylor WC and Freeling WC (1984) DNA insertion in the first intron of maize Adhl affects message levels: Cloning of progenitor and mutant alleles. Proc Natl Acad Sci USA 81: 4125–4128
Bennetzen JL (1987) Covalent DNA modification and the regulation of Mutator element transposition in maize. Mol Gen Genet 208: 45–51
Bennetzen JL, Fracasso RP, Morris DW, Robertson DS and Skogen-Hagenson MJ (1987) Concomitant regulation of Mu1 transposition and Mutator activity in maize. Mol Gen Genet 208: 57–62
Bonas U, Sommer H, Harrison BJ and Saedler H (1984) The transposable element Taml of Antirrhinum majus is 17 kb long. Mol Gen Genet 194: 138–143
Calos MP and Miller JH (1980) Transposable elements. Cell 20: 579–595
Campbell A (1981) Evolutionary significance of accessory DNA elements in bacteria. Ann Rev Microbiol 35: 55–83
Carpenter R, Martin C and Coen ES (1987) Comparison of genetic behavior of the transposable element Tam3 at two unliked pigment loci in Antirrhinum majus. Mol Gen Genet 207: 82–89
Chandler VL and Walbot V (1986) DNA modification of a maize transposable element correlates with loss of activity. Proc Natl Acad Sci USA 83: 1767–1771
Chandler VL, Rivin C and Walbot V (1986) Stable non-Mutator stocks of maize have sequences homologous to the Mu1 transposable element. Genetics 114: 1007–1021
Chen J (1986) Plant genomic DNA preparation. In: Plant Gene Cloning Manual. Cold Spring Harbor: Maize molecular genetics group, Cold Spring Harbor Laboratory
Cone KC, Burr FA and Burr B (1986) Molecular analysis of the maize anthocyanin regulatory locus C1. Proc Natl Acad Sci USA 83: 9631–9635
Cook B and Miles D (1987) Mutator-induced PSII photosynthesis mutant is allelic to hcf3. Maize Genet Coop Newslett 61: 44
Cook B, Hunt M and Miles D (1987) Mutator-induced mutation on 8L affects the chloroplast cytochrome b6/f complex. Maize Genet Coop Newslett 61: 44
Federoff NV (1983) Controlling elements in maize. In: Shapiro JA (ed) Mobile Genetic Elements, pp 1–65. New York: Academic Press/Harcourt Brace Jovanovich
Federoff NV, Wessler S and Shure M (1983) Isolation of the transposable maize controlling elements Ac and Ds, Cell 35: 235–242
Federoff NV, Furtek DB and Nelson OE (1984) Cloning of the bronze locus in maize by a simple and generalizable procedure using the transposable controlling element Activator (Ac). Proc Natl Acad Sci USA 81: 3825–3829
Feinberg AP and Vogelstein B (1983) A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Bioch 132: 6–13 and Addendum Anal Bioch 137: 266–267
Freeling M (1984) Plant transposable elements and insertion sequences. Ann Rev Plant Physiol 35: 277–298
Goldberg RB, Hoschek G and Vodkin LO (1983) An insertion sequence blocks the expression of a soybean lectin gene. Cell 33: 465–475
Grindley NDF and Reed RR (1985) Transpositional recombination in prokaryotes. Ann Rev Biochem 54: 863–896
Guikema JA and Sherman LA (1980) Electrophoretic profiles of cyanobacterial membrane polypeptides showing heme-dependent peroxidase activity. Biochem Biophys Acta 637: 189–201
Heffron F (1983) Tn3 and its relatives. In: Shapiro JA (ed) Mobile Genetic Elements, pp 223–260. New York: Academic Press/Harcourt Brace Jovanovich
Hehl R, Sommer H and Saedler H (1987) Interaction between Tam1 and Tam2 transposable elements of Antirrhinum majus. Mol Gen Genet 207: 47–53
Helentjaris T, King G, Slocum M, Siedenstrang C and Wegman S (1985) Restriction fragment polymorphisms as probes for plant diversity and their development as tools for applied plant breeding. Plant Mol Biol 5: 109–118
Hudson A, Carpenter R and Coen ES (1987) De novo activation of the transposable element Tam2 of Antirrhinum majus. Mol Gen Genet 207: 54–59
Iida S, Meyer J and Arber W (1983) Prokaryotic IS elements. In: Shapiro JA (ed) Mobile Genetic Elements, pp 159–222. New York: Academic Press/Harcourt Brace Jovanovich
Kleckner N (1981) Transposable elements in prokaryotes. Ann Rev Genet 15: 341–404
Krueger RW and Miles D (1981) Photosynthesis in tall fescue. I. High rates of electron transport and photophosphorylation in chloroplasts of hexaploid plants. Plant Physiol 67: 763–767
Leto KJ and Miles D (1980) Characterization of three Photosystem II mutants in Zea mays L. lacking a 32,000 Dalton lamellar polypeptide. Plant Physiol 66: 18–24
Lillis M and Freeling M (1986) Mu transposons in maize. Trends Genet 2: 183–188
Martienssen RA, Barkan A, Scriven A and Taylor WC (1987) Identification of a nuclear gene involved in thylakoid structure. In: Leaver C and Sze H (Eds) Plant Membranes: Structures, Function and biogenesis (UCLA Symposia on Molecular and Cellular Biology 63: 118–194). New York: Alan R Liss
McClintock B (1984) the significance of responses of the genome to challenge. Science 226: 792–801
Metz JG and Miles D (1982) Use of a nuclear mutant of maize to identify components of photosystem II. Biochem Biophys Acta 681: 95–102
Metz JG, Miles D and Rutherford AW (1983) Characterization of nuclear mutants of maize which lack the cytochrome f/b-563 complex. Plant Physiol 73: 452–459
Miles D (1980) Mutants of higher plants: Maize. In: San Pietro A (ed) Photosynthesis, Part C, Methods in Enzymology 69: 3–22. New York: Academic Press
Miles D (1982) the use of mutations to probe photosynthesis in higher plants. In: Edelman M, Hallick RB and Chua N-H (eds) Methods in Chloroplast Molecular Biology, pp 75–107. Amsterdam: Elsevier Biomedical Press
Miles D and Randall D (1983) Nuclear mutants of maize altering the large subunit of ribulose-1,5-bisphosphate carboxylase. In: Randall DD, Blevins DG and Larson R (eds) Current Topics in Plant Biochemistry and Physiology 1: 231. Columbia: Interdisciplinary Plant Biochemistry and Physiology Program
Miles D, Leto KJ, Neuffer MG, Polacco M, Hanks JF and Hunt MA (1985) Chromosome arm location of photosynthesis mutants in Zea mays L. using B-A translocations. In: Steinbeck KE, Arntzen CJ and Bogorad J (Eds) Molecular biology of the photosynthetic apparatus, pp 361–365. Cold spring Harbor: Cold Spring Harbor Laboratory
Nevers P, Shepherd NS and Saedler H (1986) Plant transposable elements. Adv Bot Res 12: 103–203
O'Reilly C, Shepherd NS, Pereira A, Schwarz-Sommer Zs, Bertram I, Robertson DS, Peterson PA and Saedler H. (1985) Molecular cloning of the a1 locus of Zea mays using the transposable elements En and Mu1. EMBO J 4: 877–882
Paz-Ares J, Wienand U, Peterson PA and Saedler H (1986) Molecular cloning of the c locus of Zea mays: a locus regulating the anthocyanin pathway. EMBO J 5: 829–833
Pereira A, Schwarz-Sommer Zs, Gierl A, Bertram I, Peterson PA and Saedler H (1985) Genetic and molecular analysis of the Enhancer (En) transposable element system of Zea maus. EMBO J 4: 17–23
Pohlman RF, Federoff NV and Mesing J (1984) the nucleotide sequence of the maize controlling element Activator. Cell 37: 635–643
Robertson GS (1978) Characterization of a mutator system in maize. Mutation Res 51: 21–28
Roeder GS and Fink GR (1983) Transposable elements in yeast. In: Shapiro JA (ed) Mobile Genetic Elements, pp 300–328. New York: Academic Press/Harcourt Brace Jovanovich
Rubin GM (1983) Dispersed repetitive DNAs in Drosophila. In: Shapiro JA (ed) Mobile Genetic Elements, pp 329–362. New York: Academic Press/Harcourt Brace Jovanovich
Sachs MM, Peacock WJ, Dennis ES and Gerlack WL (1983) Maize Ac/Ds controlling elements. A molecular viewpoint. Maydica 28: 289–303
Schwartz D and Dennis E (1986) Transposase activity of the Ac controlling element in maize is regulated by its degree of methylation. Mol Gen Genet 206: 476–482
Schwarz-Sommer Zs, Gierl A, Klosgen RB, Wienand U, Peterson PA and Saedler H (1984) The Spm (En) transposable element controls the excision of a 2-kb DNA insert at the wxm−8 allele of Zea mays. EMBO J 3: 1021–1028
Shapiro JA (1983) Genetic reorganization in cell lineages. In: Shapiro JA, (ed) Mobile Genetic Elements, pp xi-xvi. New York: Academic Press/Harcourt Brace Jovanovich
Shure M, Wessler S and Federoff N (1983) Molecular identification and isolation of the Waxy locus in maize. Cell 35: 225–233
Somerville CR (1986) Analysis of photosynthesis with mutants of higher plants and algae. Ann Rev Plant Physiol 37: 467–507
Southern EM (1975) Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol 98: 503–517
Taylor LP, Chandler VL and Walbot V (1986) Insertion of 1.4 kb and 1.7 kb Mu elements into the Bronze-1 gene of Zea mays L. Maydica 31: 31–45
Taylor WC, Barkan A and Martienssen RA (1988) The use of nuclear mutants in the analysis of chloroplast development. Developmental Genet 8: 305–320
Thomas PE, Rayn D and Levin W (1976) An improved staining procedure for the detection of the peroxidase activity of cytochrome P-450 on sodium dodecyl sulfate polyacrylamide gels. Anal Biochem 75: 168–176
Tittgen J, Hermans J, Steppuhn J, Jansen T, Jannson C, Anderson B, Nechushtai R, Nelson N and Herrmann RG (1986) Isolation of cDNA clones for fourteen nuclear-encoded thylakoid membrane proteins. Mol Gen Genet 204: 258–265
Walbot V, Chandler VL, Taylor LP and McLaughlin P (1987) Regulation of transposable element activities during the development and evolution of Zea mays L. In: Development as an Evolutionary Process, pp 265–284. New York: Alan R Liss
Webber DF (1986) The production and utilisation of monosomic Zea mays in cytogenetic studies. In: Reddy GM and Co EH (eds) Gene Structure and Function in Higher Plants, pp 191–204. Dehli: Oxford and IBH Publishing
Young RA and Davis RW (1983) Efficient isolation of genes by using antibody probes. Proc Natl Acad Sci USA 80: 1194–1198
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cook, W.B., Miles, D. Transposon mutagenesis of nuclear photosynthetic genes in Zea mays . Photosynth Res 18, 33–59 (1988). https://doi.org/10.1007/BF00042979
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00042979