Abstract
Objective: To evaluate the efficacy and tolerability of oral zolmitriptan as a short-term preventative therapy for menstrual migraine.
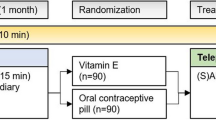
Methods: This was a randomized, double-blind, parallel group, placebo-controlled, multicentre, two-phase study. The results of the second phase are reported here (the first phase evaluated zolmitriptan in the acute treatment of menstrual migraine and is reported elsewhere). Women who successfully completed phase I (with either a positive or negative outcome, and who still fulfilled the inclusion criteria) were randomized to zolmitriptan 2.5 mg oral tablet three times daily, zolmitriptan 2.5 mg twice daily or placebo three times daily. Patients were treated for three consecutive menstrual cycles, starting 2 days prior to the expected onset of menses, for 7 days in total.
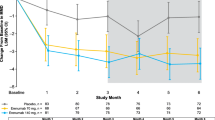
Results: Two hundred and fifty-three patients completed phase I and were eligible for phase II. The intention-to-treat population comprised 244 patients (zolmitriptan three times daily [n = 83]; zolmitriptan twice daily [n = 80]; placebo [n = 81]). Both zolmitriptan regimens demonstrated superior efficacy versus placebo, as measured by the proportion of patients with a ≥50% reduction in the frequency of menstrual migraine attacks (zolmitriptan three times daily [58.6%], p = 0.0007 vs placebo; zolmitriptan twice daily [54.7%], p = 0.002 vs placebo; placebo three times daily [37.8%]). The mean frequency of breakthrough migraine attacks per menstrual cycle was reduced accordingly. Fewer breakthrough attacks were treated with escape medication in the zolmitriptan three times daily (61.6% of attacks; p = 0.0004 vs placebo) and twice daily (60.7%; p = 0.0055 vs placebo) treatment groups than in the placebo group (74.4%). Short-term preventative therapy with zolmitriptan was well tolerated.
Conclusion: Zolmitriptan 2.5 mg oral tablet is effective and well tolerated as a short-term preventative therapy for menstrual migraine attacks.






Similar content being viewed by others
Notes
1 The use of trade names is for product identification purposes only and does not imply endorsement.
References
Lipton RB, Stewart WF, Diamond S, et al. Prevalence and burden of migraine in the United States: data from the American Migraine Study II. Headache 2001; 41: 646–57
Brandes JL. The influence of estrogen on migraine: a systematic review. JAMA 2006; 295: 1824–30
Somerville B. The role of estradiol withdrawal in the etiology of menstrual migraine. Neurology 1972; 22: 355–65
Kornstein SG, Parker AJ. Menstrual migraines: etiology, treatment, and relationship to premenstrual syndrome. Curr Opin Obstet Gynecol 1997; 9: 154–9
MacGregor EA, Chia H, Vohrah RC, et al. Migraine and menstruation: a pilot study. Cephalalgia 1990; 10: 305–10
MacGregor EA, Brandes J, Eikermann A, et al. Impact of migraine on patients and their families. The Migraine And Zolmitriptan Evaluation (MAZE) survey: phase III. Curr Med Res Opin 2004; 20: 1143–50
Stewart WF, Lipton RB, Chee E, et al. Menstrual cycle and headache in a population sample of migraineurs. Neurology 2000; 55: 1517–23
Massiou H. Is menstrually associated migraine difficult to treat? Cephalalgia 1999; 19Suppl. 24: 13–8
Couturier EGM, Bomhof MAM, Knuistingh Neven A, et al. Menstrual migraine in a representative Dutch population sample: prevalence, disability and treatment. Cephalalgia 2003; 23: 302–8
MacGregor EA, Hackshaw A. Prevalence of migraine on each day of the natural menstrual cycle. Neurology 2004; 63: 351–3
Granella F, Sances G, Allais G, et al. Characteristics of menstrual and nonmenstrual attacks in women with menstrually related migraine referred to headache centres. Cephalalgia 2004; 24: 707–16
Mannix LK, Files JA. The use of triptans in the management of menstrual migraine. CNS Drugs 2005; 19: 951–72
Newman LC, Lipton RB, Lay CL, et al. A pilot study of oral sumatriptan as intermittent prophylaxis of menstruation-related migraine. Neurology 1998; 51: 307–9
Newman L, Mannix LK, Landy S, et al. Naratriptan as short-term prophylaxis of menstrually associated migraine: a randomized, double-blind, placebo-controlled study. Headache 2001; 41: 248–56
Silberstein SD, Elkind AH, Schreiber C, et al. A randomized trial of frovatriptan for the intermittent prevention of menstrual migraine. Neurology 2004; 63: 261–9
Moschiano F, Allais G, Grazzi L, et al. Naratriptan in the short-term prophylaxis of pure menstrual migraine. Neurol Sci 2005; 26Suppl. 2: S162–6
Campbell JC, Tobin J, Oleka N, et al. Frovatriptan for the short-term prevention of menstrual migraine: a retrospective subanalysis in triptan-experienced women. Headache 2006; 46: 843–4
MacGregor A, Pawsey S, Campbell J, et al. Use of frovatriptan for the short-term prevention of pure menstrual and menstrually-related migraine headaches: results of a 12-month, open-1abel, safety and tolerability trial. Headache 2006; 46: 844–5
Brandes JL, Smith T, Diamond M, et al. Open-1abel, long-term tolerability of naratriptan for short-term prevention of menstrually related migraine. Headache 2007; 47: 886–94
Mannix LK, Savani N, Landy S, et al. Efficacy and tolerability of naratriptan for short-term prevention of menstrually related migraine: data from two randomized, double-blind, placebo-controlled studies. Headache 2007; 47: 1037–49
Rapoport AM, Ramadan NM, Adelman JU, et al. Optimizing the dose of zolmitriptan (Zomig, *311C90) for the acute treatment of migraine. Neurology 1997; 49: 1210–8
Solomon GD, Cady RK, Klapper JA, et al. Clinical efficacy and tolerability of 2.5mg zolmitriptan for the acute treatment of migraine. The 042 Clinical Trial Study Group. Neurology 1997; 49: 1219–25
The long-term tolerability and efficacy of oral zolmitriptan (Zomig, 311C90) in the acute treatment of migraine: an international study. International 311C90 Long-term Study Group. Headache 1998; 38: 173–83
Tuchman M, Edvinsson L, Geraud G, et al. Zolmitriptan provides consistent migraine relief when used in the long-term. Curr Med Res Opin 1999; 15: 272–81
Loder E, Silberstein SD, Abu-Shakra S, et al. Efficacy and tolerability of oral zolmitriptan in menstrually associated migraine: a randomized, prospective, parallel-group, double-blind, placebo-controlled study. Headache 2004; 44: 120–30
Tuchman M, Hee A, Emeribe U, et al. Efficacy and tolerability of zolmitriptan oral tablet in the acute treatment of menstrual migraine. CNS Drugs 2006; 20: 1019–26
International Headache Society, Headache Classification Committee. Classification and diagnostic criteria for headache disorders, cranial neuralgias and facial pain. Cephalalgia 1988; 8Suppl. 7: 1–96
Dixon R, Warrander A. The clinical pharmacokinetics of zolmitriptan. Cephalalgia 1997; 17Suppl. 18: 15–20
International Headache Society, Headache Classification Committee. The international classification of headache disorders. 2nd ed. Cephalalgia 2004; 24Suppl. 1: 1–160
Acknowledgements
This study was supported by AstraZeneca. The authors would like to thank Steve Winter, Wolters Kluwer Health, who provided medical writing support funded by AstraZeneca. Dr Tuchman has received research funding from AstraZeneca, Allergan, GlaxoSmithKline, Merck, Sanofi, Ortho-McNeil, Pozen, Pfizer, Takeda, Eisai and Neurochem, and has received honoraria for speaking or consultations from AstraZeneca, GlaxoSmithKline, Merck, Pfizer and Eisai. Angela Hee was an employee of AstraZeneca at the time the study was completed. Ugochi Emeribe is an employee of AstraZeneca. Stephen Silberstein has received research funding from Abbott, Advanced Bionics, Advanced NeuroModulation Systems, AGA, Allergan, AstraZeneca, Eli Lilly, Endo Pharmaceuticals, GlaxoSmithKline, Medtronic, Merck, Ortho-McNeil, Pfizer, Pozen, ProEthic, Valeant Pharmaceuticals and Vernalis, and has received honoraria for acting as a speaker and/or advisory board member for Allergan, AstraZeneca, Endo Pharmaceuticals, GlaxoSmithKline, Medtronic, Merck, Ortho-McNeil, Pfizer, Pozen and Valeant Pharmaceuticals.
Author information
Authors and Affiliations
Corresponding author
Additional information
Although an employee of AstraZeneca at the time this study was completed, Angela Hee is currently employed by Johnson & Johnson Consumer & Personal Products Worldwide.
Rights and permissions
About this article
Cite this article
Tuchman, M.M., Hee, A., Emeribe, U. et al. Oral Zolmitriptan in the Short-Term Prevention of Menstrual Migraine. CNS Drugs 22, 877–886 (2008). https://doi.org/10.2165/00023210-200822100-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00023210-200822100-00007