Summary
Over the last decade, chloroquine-resistant falciparum malaria has spread to other areas from its original foci in Southeast Asia and South America. Additionally, new knowledge about the life-cycle of the malaria parasite, and about the pharmacokinetic properties of antimalarial drugs, has emerged. It is appropriate to reassess our approach to prevention and management of malaria with these factors in mind.
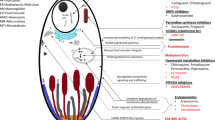
Antimalarial drugs can be classified in two ways: (a) biologically as tissue schizontocides, hypnozoitocides, blood schizontocides, gametocytocides or sporontocides; or (b) by a mixed chemical/biological classification as 8-aminoquinolines, antimetabolites and (again) blood schizontocides.
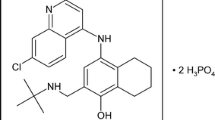
Chloroquine resistance in P. falciparum can now be found in most areas where malaria occurs. Malarial strains moderately resistant to the chloroquine group of drugs (chloroquine and mepacrine) are generally susceptible to the aryl amino alcohols such as quinine. Indeed, quinine is the most widely used drug for treating malaria due to chloroquine-resistant strains, followed by a 7-day course of tetracycline where some resistance to quinine is also found. Alternatively, the course of quinine may be followed by sulfadoxine/ pyrimethamine or the newer quinoline derivative, mefloquine. Quinidine has also shown activity against quinine-resistant strains. Prophylaxis of chloroquine-resistant strains is best undertaken with daily proguanil (chloroguanide), and weekly chloroquine.
In severe malaria, including cerebral malaria, an intravenous loading dose of quinine should be considered, and plasma concentration monitoring may be advisable to assist with dosage adjustment. In patients with severe renal insufficiency, there is evidence that the elimination of chloroquine is prolonged, and dosage adjustments may be nécessary.
Other recent findings on the pharmacodynamic properties, mechanisms of action and toxicity of antimalarial drugs are also discussed.
Similar content being viewed by others
References
Bergqvist Y, Domeij-Nyberg B. Distribution of chloroquine and its metabolite desethylchloroquine in human blood cells and its implications for the quantitative determination of these compounds in plasma. Journal of Chromatography 272: 137–148, 1983
Bradley DJ, Hall AP, Peters W, Warhurst DC. Fansidar in malaria prophylaxis. British Medical Journal 291: 136, 1985
Brooks MH, Mallon JP, Bartelloni PJ. Quinine, pyrimethamine and sulphordimethoxine: clinical response, plasma levels and urinary excretion during initial attack of naturally acquired falciparum malaria. Clinical and Pharmacological Therapeutics 10: 85–91, 1969
Bruce-Chwatt LJ. Essential Malariology, 2nd ed., Heinemann, London, 1985
Bruce-Chwatt LJ, Hutchinson DBA. Maloprim and agranulocytosis. Lancet 2: 1487–1488, 1983
Bruce-Chwatt LJ, Black, RH, Canfield CJ, Clyde DF, Peters W, Wernsdorfer WH (Eds) Chemotherapy of malaria, 2nd ed., World Health Organization, Geneva, 1981
Chawira AN, Warhurst DC, Robinson BL, Peters W. Development of resistance to ginghaosu (artemisinin) in rodent malarias. Transactions of the Royal Society of Tropical Medicine and Hygiene 79: 275, 1985
Chevli R, Fitch CD. The antimalarial drug mefloquine binds to membrane phospholipids. Antimicrobial Agents and Chemotherapy 21: 581–586, 1982
Chiodini PL, Somerville M, Salam I, Tubbs HR, Wood MJ, et al. Exchange transfusion in severe falciparum malaria. Transactions of the royal Society for Tropical Medicine and Hygiene 79: 865–866, 1985
Chongsuphajaisiddhi T, Sabcharoen A, Attanath P. In vitro and in vivo sensitivity of falciparum malaria to quinine in Thai children. Annals of Tropical Pediatrics 1: 21–26, 1981
Chou AC, Chevli R, Fitch CD. Ferriprotoporphyrin IX fulfils the criteria for identification as the chloroquine receptor of malaria parasites. Biochemistry 19: 1543–1549, 1980
Cohen SF, Phifer KD, Yielding KL. Complex formation between chloroquine and ferrihaemic acid in vitro and its effect on the antimalarial action of chloroquine. Nature 202: 805–806, 1964
Davies EE, Warhurst DC, Peters W. The chemotherapy of rodent malaria XXI. Action of quinine and WR 122,455, (a 9-phen-anthrene methanol) on the fine structure of Plasmodium berghei in mouse blood. Annals of Tropical Medicine and Parasitology 69: 147–153, 1975
Diribe CO, Warhurst DC. Inhibitors of chloroquine uptake. Transactions of the Royal Society of Tropical Medicine and Hygiene 74: 675–676, 1980
Diribe CO, Warhurst DC. A study of the uptake of chloroquine in malaria infected erythrocytes. Biochemical Pharmacology 34: 3019–3027, 1985
Diribe CO, Warhurst DC. The effect of ionophores and ouabain on uptake of chloroquine by malaria parasites. Submitted for publication, 1986
Eckman JR, Modler S, Eaton JW, Berger E, Engel RR. Host heme catabolism in drug sensitive and drug resistant malaria. Journal of Laboratory and Clinical Medicine 90: 767–770, 1977
Ellis DS, Li ZL, Gu HM, Peters W, Robinson BL et al. The chemotherapy of rodent malaria XXXIX: Ultrastructural changes following treatment with artemisinine of Plasmodium berghei infection in mice, with observations of the localisation of [3H] dihydroartemisinine by erythrocytes infected with Plasmodium falciparum. Annals of Tropical Medicine and Parasitology 79: 367–374, 1985
Fitch CD. Mode of action of antimalarial drugs. In Evered and Whelan. Malaria and the red cell, CIBA Foundation Symposium 94, pp. 222–232, and Pitman, London, 1983
Friedman MJ, Roth EF, Nagel RC, Trager W. Plasmodium falciparum: physiological interactions with the human sickle cell. Experimental Parasitology 47: 73–80, 1979
Garnham PCC. Malaria parasites and other haemosporidia, Blackwell, Oxford, 1966
Garnham PCC. The myth of quartan malaria (Haemamoeba laverania var quartana Labbe, 1894). Transactions of the Royal Society of Tropical Medicine and Hygiene 75: 616–617, 1981
Garnham PCC. Life cycles. In Peters and Richards (Eds) Antimalarial drugs I, pp. 3–30. Springer Verlag, Berlin, 1984
Gu HM, Lu BF, Qu ZX. Activities of 25 derivatives of artemisinine against chloroquine-resistant Plasmodium berghei. Acta Pharmacologica Sinica 1: 48–50, 1980
Gu HM, Warhurst DC, Peters W. Rapid action of ginghaosu and related drugs on incorporation of [3H] isoleucine by Plasmodium falciparum in vitro. Biochemical Pharmacology 32: 2463–2466, 1983
Gu HM, Warhurst DC, Peters W. Uptake of [3H] dihydroartemisinine by erythrocytes infected with Plasmodium falciparum in vitro. Transactions of the Royal Society of Tropical Medicine and Hygiene 78: 265–270, 1984
Gustafsson LL, Walker O, Alvan G, Beerman B, Estevez F, et al. Disposition of chloroquine in man after single intravenous and oral doses. British Journal of Clinical Pharmacology 15: 471–479, 1983
Gutteridge WE, Dave D, Richards WHG. Conversion of dihy-droorotate to orotate in parasitic protozoa. Biochimica Biophysica Acta 581: 390–401, 1979
Hall AP. The treatment of severe falciparum malaria. Transactions of the Royal Society of Tropical Medicine and Hygiene 71: 367–368, 1977
Hammond DJ, Burchell JR, Pudney M. Inhibition of pyrimidine synthesis de novo in Plasmodium falciparum by 2-(4-t-butyl-cyclohexyl)-3-hydroxy-1, 4-naphthoquinone in vitro. Molecular and Biochemical Parasitology 14: 97–109, 1985
Hatton CSR, Peto TEA, Bunch C, Pasvol G, et al. Frequency of severe neutropenia associated with amodiaquine prophylaxis against malaria. Lancet 1: 411–414, 1986
Hernborg A. Stevens-Johnson syndrome after mass prophylaxis with sulfadoxine for cholera in Mozambique. Lancet 2: 1072–1073, 1985
Homewood CA, Warhurst DC, Peters W, Baggaley VC. Lyso-somes, pH, and the antimalarial action of chloroquine. Nature 235: 50–52, 1972
Homewood CA, Moore GA, Warhurst DC, Atkinson EM. Purification and some properties of malaria pigment. Annals of Tropical Medicine and Parasitology 69: 283–287, 1975
Jaffe, JJ. Dihydrofolate reductase in parasitic protozoa and helminths. In Van Den Bossche (Ed.) Comparative Biochemistry of Parasites, pp. 219–233, Academic Press, London, 1972
Krotoski WA. Discovery of the hypnozoite and a new theory of malaria relapse. Transactions of the Royal Society of Tropical Medicine and Hygiene 79: 1–11, 1985
Li G, Arnold K, Guo X, Jian H, Fu L. Randomised study of mefloquine, ginghaosu, and pyrimethamine-sulfadoxine in patients with falciparum malaria. Lancet 2: 1360–1361, 1984
Lullmann H, Plosch H, Ziegler CA. Ca replacement by cationic amphiphilic drugs from lipid monolayers. Biochemical Pharmacology 29: 2969–2974, 1980
McLarty DG, Jaatinen M, Aubert B, Webber RH, Kihamia CH, et al. Prophylaxis of malaria in non-immune residents in Dar Es Salaam, Tanzania. Lancet 2: 656–658, 1984
McChesney JD, Baker JK, Clark AM, Hufford CD. Metabolism of antimalarial drug candidates. In Wernsdorfer & Trigg (Eds) Modern design of antimalarial drugs, pp. 57–82, UNDP/World Bank/WHO, Geneva, 1983
Mikkelsen RB, Tanabe K, Wallach DFH. Membrane potential of Plasmodium-infected erythrocytes. Journal of Cell Biology 93: 685–689, 1982
MMWR. Revised recommendations for preventing malaria in travelers to areas with chloroquine-resistant Plasmodium falciparum. Morbidity and Mortality Weekly Reports 34: 185–190, 1985
Moreau S, Perty B, Biguet J. Interaction de la chloroquine avec la ferriprotoporphyrin IX: etude par resonance magnetique nucleaire. Biochimie 64: 1015–1025, 1982
Pasvol G, Wilson RJM. The interaction of malaria parasites with red blood cells. British Medical Bulletin 38: 187–192, 1982
Peters W. Drugs that affect hypnozoites of Plasmodium. Transactions of the Royal Society of Tropical Medicine and Hygiene 77: 742, 1983
Peters W. The problem of drug resistance in malaria. Parasitology 90: 705–715, 1985
Peters W, Hall AP. The treatment of severe falciparum malaria. British Medical Journal 291: 1146–1147, 1985
PHLS. Prevention of malaria in pregnancy and early childhood. British Medical Journal 289: 1296–1297, 1984
Playfair JHL. Immunity to malaria. British Medical Bulletin 38: 153–159, 1982
Qinghaosu Antimalaria Coordinating Research Group. Antimalaria studies on qinghaosu. Chinese Medical Journal 92: 811–816, 1979
Rombo L, Stenbeck J, Lobel HO, Campbell CC, Papioanou M, et al. Does chloroquine contribute to the risk of serious adverse reactions to Fansidar? Lancet 2: 1298–1299, 1985
Salako L, Walker O, Iyun AO. Pharmacokinetics of chloroquine in renal insufficiency. African Journal of Medicine and Medical Sciences 13: 177–182, 1984
Sansonetti PJ, Lebras C, Verdier F, Charmot G, Dupont B, et al. Chloroquine-resistant Plasmodium falciparum in Cameroon. Lancet 1: 1154–1155, 1985
Shann F, Stace J, Edstein M. Pharmacokinetics of quinine in children. Journal of Pediatrics 106: 506–510, 1985
Sherman IW. Biochemistry of plasmodium (malaria parasites). Microbiological Reviews 43: 453–495, 1979
Strother A, Allahyari R, Buchhoĺz J, Fraser IM, Tilton BE. In vitro metabolism of the antimalarial agent primaquine by mouse liver enzymes and identification of a methemoglobin-forming metabolite. Drug Metabolism and Diposition 12: 35–44, 1984
Targett GAT. Interactions between chemotherapy and immunity. In Peters & Richards (Eds) Antimalarial drugs I, pp. 331–348, Springer Verlag, Berlin, 1984
Trenholme GH, Williams RL, Rieckmann KH, Frischer H, Carson PE. Quinine disposition during malaria and induced fever. Clinical Pharmacology and Therapeutics 19: 459–467, 1976
Warhurst DC. Chemotherapeutic agents and malaria research. In Taylor & Muller (Eds) Chemotherapeutic Agents in the study of parasites, pp. 1–28, Blackwell, Oxford, 1973
Warhurst, DC. The quinine-haemin interaction and its relationship to antimalarial activity. Biochemical Pharmacology 30: 3323–3327, 1981a
Warhurst DC. Cinchona alkaloids and malaria. Lancet 2: 1346, 1981b
Warhurst DC. Why are primaquine and other 8-aminoquinolines particularly effective against the mature gametocytes and the hypnozoites of malaria? Annals of Tropical Medicine and Parasitology 78: 165, 1984a
Warhurst DC. Drug-induced clumping test. In Peters & Richards (Eds) Antimalarial drugs I, pp. 303–329, Springer Verlag, Berlin, 1984b
Warhurst DC. Antimalarial blood schizontocides: why a permease is necessary. Parasitology Today, in press, 1986
Warhurst DC, Thomas SC. Pharmacology of the malaria parasite: a study of of dose-response relationships in chloroquine-induced autophagic vacuole formation in Plasmodium berghei. Biochemical Pharmacology 24: 2047–56, 1975a
Warhurst DC, Thomas SC. Localisation of mepacrine in Plasmodium berghei and Plasmodium falciparum by fluorescence microscopy. Annals of Tropical Medicine and Parasitology 69: 417–420, 1975b
Warhurst DC, Thomas SC. Effect of some metabolic inhibitors upon chloroquine-induced pigment clumping (CIPC) in Plasmodium berghei. Annals of Tropical Medicine and Parasitology 72: 203–211, 1978
Watkins WM, Sixsmith DG, Chulay JD, Spencer HC. Antagonism of sulfadoxine and pyrimethamine antimalarial activity in vitro by p-amino benzoic acid, p-aminobenzoylglutamic acid, and folic acid. Molecular and Biochemical Parasitology 14: 55–61, 1985
White NJ. Clinical pharmacokinetics of antimalarial drugs. Clinical Pharmacokinetics 10: 187–215, 1985
White NJ, Warrell DA. Clinical management of chloroquine-resistant falciparum malaria in Southeast Asia. Tropical Doctor 13: 153–158, 1983
White NJ, Looaresuwan S, Warrell DA, Chongsuphajaisiddhi T, Bunnag D, et al. Quinidine in falciparum malaria. Lancet 2: 1069–1071, 1981
White, NJ, Looaresuwan S, Warrell DA, Warrell MJ, Bunnag D, et al. Quinine pharmacokinetics and toxicity in cerebral and uncomplicated falciparum malaria. American Journal of Medicine 73: 564–572, 1982
White NJ, Looaresuwan S, Warrell DA. Quinine and quinidine: a comparison of EKG effects during the treatment of malaria. Journal of Cardiovascular Pharmacology 5: 173–175, 1983
White NJ, Looaresuwan S, Warrell DA, Warrell MJ. Chantharanich P, et al. Quinine loading dose in cerebral malaria. American Journal of Tropical Medicine and Hygiene 32: 1–5, 1983a
White NJ, Warrell DJ, Chantavanich P, Looaresuwan S, Warrell MJ, et al. Severe hypoglycemia and hyperinsulinemia in falciparum malaria. New England Journal of Medicine 309: 61–66, 1983b
WHO. Malaria Chemoprophylaxis. WHO Weekly Epidemiological Record 60: 181–188, 1985
WHO. Severe and complicated malaria (World Health Organization Malaria Action Programme). Transactions of the Royal Society of Tropical Medicine and Hygiene 80 (Suppl.): 1–50, 1986
Yamada KA, Sherman IW. Plasmodium lophurae: composition and properties of hemozoin, the malarial pigment. Experimental Parasitology 48: 61–74, 1979
Yayon A, Cabantchick ZI, Ginsberg H. Identification of the acidic compartment of Plasmodium falciparum-infected erythrocytes as the target of the antimalarial drug chloroquine. EMBO Journal 3: 2695–2700, 1984
Author information
Authors and Affiliations
Additional information
This document represents the personal views of the author, and should not be regarded as an expression of opinion of the PHLS Malaria Reference Laboratory.
Rights and permissions
About this article
Cite this article
Warhurst, D.C. Antimalarial Drugs. Drugs 33, 50–65 (1987). https://doi.org/10.2165/00003495-198733010-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-198733010-00003