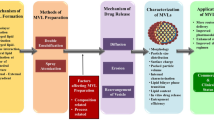
Abstract
Lamellar liposome technology has been used for several decades to produce sustained-release drug formulations for parenteral administration. Multivesicular liposomes are structurally distinct from lamellar liposomes and consist of an aggregation of hundreds of water-filled polyhedral compartments separated by bi-layered lipid septa. The unique architecture of multivesicular liposomes allows encapsulating drug with greater efficiency, provides robust structural stability and ensures reliable, steady and prolonged drug release.
The favourable characteristics of multivesicular liposomes have resulted in many drug formulations exploiting this technology, which is proprietary and referred to as DepoFoam™. Currently, two formulations using multivesicular liposome technology are approved by the US FDA for clinical use, and many more formulations are at an experimental developmental stage. The first clinically available formulation contains the antineoplastic agent cytarabine (DepoCyt™) for its intrathecal injection in the treatment of malignant lymphomatous meningitis. Intrathecal injection of DepoCyt™ reliably results in the sustained release of cytarabine and produces cytotoxic concentrations in cerebrospinal fluid (CSF) that are maintained for at least 2 weeks. Early efficacy data suggest that DepoCyt™ is fairly well tolerated, and its use allows reduced dosing frequency from twice a week to once every other week and may improve the outcome compared with frequent intrathecal injections of unencapsulated cytarabine. The second available formulation contains morphine (DepoDur™) for its single epidural injection in the treatment of postoperative pain. While animal studies confirm that epidural injection of DepoDur™ results in the sustained release of morphine into CSF, the CSF pharmacokinetics have not been determined in humans. Clinical studies suggest that the use of DepoDur™ decreases the amount of systemically administered analgesics needed for adequate postoperative pain control. It may also provide superior pain control during the first 1–2 postoperative days compared with epidural administration of unencapsulated morphine or intravenous administration of an opioid. However, at this timepoint the overall clinical utility of DepoDur™ has yet to be defined and some safety concerns remain because of the unknown CSF pharmacokinetics of DepoDur™ in humans.
The versatility of multivesicular liposome technology is reflected by the many agents including small inorganic and organic molecules and macromolecules including proteins that have successfully been encapsulated. Data concerning many experimental formulations containing antineoplastic, antibacterial and antiviral agents underscore the sustained, steady and reliable release of these compounds from multivesicular liposomes after injection by the intrathecal, subcutaneous, intramuscular, intraperitoneal and intraocular routes. Contingent on the specific formulation and manufacturing process, agents were released over a period of hours to weeks as reflected by a 2- to 400-fold increase in elimination half life. Published data further suggest that the encapsulation process preserves bioactivity of agents as delicate as proteins and supports the view that examined multivesicular liposomes were non-toxic at studied doses. The task ahead will be to examine whether the beneficial structural and pharmacokinetic properties of multivesicular liposome formulations will translate into improved clinical outcomes, either because of decreased drug toxicity or increased drug efficacy.




Similar content being viewed by others
Notes
The use of trade names is for product identification purposes only and dose not imply endorsement.
References
Kim S, Turker MS, Chi EY, et al. Preparation of multivesicular liposomes. Biochem Biophys Acta 1983; 728: 339–48
Mantripragada S. DepoFoam technology for sustained release injectable drug delivery. Drug Delivery Systems and Sciences 2001; 1: 13–6
Mantripragada S. A lipid based depot (DepoFoam technology) for sustained release drug delivery. Prog Lipid Res 2002; 41(5): 392–406
Xiao C, Qi X, Maitani Y, et al. Sustained release of cisplatin from multivesicular liposomes: potentiation of antitumor efficacy against S180 murine carcinoma. J Pharm Sci 2004; 93(7): 1718–24
Murry DJ, Blaney SM. Clinical pharmacology of encapsulated sustained-release cytarabine. Ann Pharmacother 2000; 34(10): 1173–8
Chowdhary S, Chamberlain M. Leptomeningeal metastases: current concepts and management guidelines. J Natl Compr Canc Netw 2005; 3(5): 693–703
Fleischhack G, Jaehde U, Bode U. Pharmacokinetics following intraventricular administration of chemotherapy in patients with neoplastic meningitis. Clin Pharmacokinet 2005; 44(1): 1–31
Howell SB. Clinical applications of a novel sustained-release injectable drug delivery system: DepoFoam technology. Cancer J 2001; 7(3): 219–27
Spector MS, Zasadzinski JA, Sankaram MB. Topology of multivesicular liposomes, a model biliquid foam. Langmuir 1996; 12(20): 4704–8
Ellena JF, Le M, Cafisco DS, et al. Distribution of phopsholipids and triglycerides in multivesicular lipid particles. Drug Deliv 1999; 6(2): 97–106
Kohn FR, Malkmus SA, Brownson EA, et al. Fate of the predominant phospholipid component of DepoFoam drug delivery matrix after intrathecal administration of sustainedrelease encapsulated cytarabine in rats. Drug Delivery Systems and Sciences 1998; 5: 143–51
Yanez AM, Wallace M, Ho R, et al. Touch-evoked agitation produced by spinally administered phospholipid emulsion and liposomes in rats: structure-activity relation. Anesthesiology 1995; 82(5): 1189–98
Wallace MS, Yanez AM, Ho RJ, et al. Antinociception and side effects of liposome-encapsulated alfentanil after spinal delivery in rats. Anesth Analg 1994; 79(4): 778–86
Isackson J, Wallace MS, Ho RJ, et al. Antinociception and side effects of L- and D-dipalmitoylphosphatidyl choline liposomeencapsulated alfentanil after spinal delivery in rats. Pharmacol Toxicol 1995; 77(5): 333–40
Ramprasad MP, Amini A, Kararli T, et al. The sustained granulopoietic effect of progenipoietin encapsulated in multivesicular liposomes. Int J Pharm 2003; 261(1–2): 93–103
Langsten MV, Ramprasad MP, Kararli TT, et al. Modulation of the sustained delivery of myelopoietin (Leridistim) encapsulated in multivesicular liposomes (DepoFoam). J Control Release 2003; 89(1): 87–99
Zhong H, Deng Y, Wang X, et al. Multivesicular liposome formulation for the sustained delivery of breviscapine. Int J Pharm 2005; 301(1–2): 15–24
Kim S, Howell SB. Multivesicular liposomes containing cytarabine entrapped in the presence of hydrochloric acid for intracavitary chemotherapy. Cancer Treat Rep 1987; 71(7–8): 705–11
Viscusi ER. DepoDur: a new drug formulation with unique safety considerations [newsletter]. Indianapolis (IN): Anesthesia Patient Safety Foundation, 2005: 50–1
Chatelut E, Suh P, Kim S. Sustained-release methotrexate for intracavitary chemotherapy. J Pharm Sci 1994; 83(3): 429–32
Katre NV, Asherman J, Schaefer H, et al. Multivesicular liposome (DepoFoam) technology for the sustained delivery of insulin-like growth factor-I (IGF-I). J Pharm Sci 1998; 87(11): 1341–6
Gambling D, Hughes T, Martin G, et al. A comparison of Depodur, a novel, single-dose extended-release epidural morphine, with standard epidural morphine for pain relief after lower abdominal surgery. Anesth Analg 2005; 100(4): 1065–74
Roy R, Kim S. Multivesicular liposomes containing bleomycin for subcutaneous administration. Cancer Chemother Pharmacol 1991; 28(2): 105–8
Chatelut E, Kim T, Kim S. A slow-release methotrexate formulation for intrathecal chemotherapy. Cancer Chemother Pharmacol 1993; 32(3): 179–82
Bonetti A, Chatelut E, Kim S. An extended-release formulation of methotrexate for subcutaneous administration. Cancer Chemother Pharmacol 1994; 33(4): 303–6
Assil KK, Lane J, Weinreb RN. Sustained release of the antimetabolite 5-fluorouridine-5′-monophosphate by multivesicular liposomes. Ophthalmic Surg 1988; 19(6): 408–13
Assil KK, Hartzer M, Weinreb RN, et al. Liposome suppression of proliferative vitreoretinopathy: rabbit model using antimetabolite encapsulated liposomes. Invest Ophthalmol Vis Sci 1991; 32(11): 2891–7
Gariano RF, Assil KK, Wiley CA, et al. Retinal toxicity of the antimetabolite 5-fluorouridine 5′-monophosphate administered intravitreally using multivesicular liposomes. Retina 1994; 14(1): 75–80
Grayson LS, Hansbrough JF, Zapata-Sirvent RL, et al. Pharmacokinetics of DepoFoam gentamicin delivery system and effect on soft tissue infection. J Surg Res 1993; 55(5): 559–64
Grayson LS, Hansbrough JF, Zapata-Sirvent R, et al. Soft tissue infection prophylaxis with gentamicin encapsulated in multivesicular liposomes: results from a prospective, randomized trial. Crit Care Med 1995; 23(1): 84–91
Roehrborn AA, Hansbrough JF, Gualdoni B, et al. Lipid-based slow-release formulation of amikacin sulfate reduces foreign body-associated infections in mice. Antimicrob Agents Chemother 1995; 39(8): 1752–5
Huh J, Chen JC, Furman GM, et al. Local treatment of prosthetic vascular graft infection with multivesicular liposome-encapsulated amikacin. J Surg Res 1998; 74(1): 54–8
Assil KK, Frucht-Perry J, Ziegler E, et al. Tobramycin liposomes: single subconjunctival therapy of pseudomonal keratitis. Invest Ophthalmol Vis Sci 1991; 32(13): 3216–20
Frucht-Perry J, Assil KK, Ziegler E, et al. Fibrin-enmeshed tobramycin liposomes: single application topical therapy of Pseudomonas keratitis. Cornea 1992; 11(5): 393–7
Jain SK, Jain RK, Chourasia MK, et al. Design and development of multivesicular liposomal depot delivery system for controlled systemic delivery of acyclovir sodium. AAPS Pharm-SciTech 2005; 6(1): E35–41
Kim S, Scheerer S, Geyer MA, et al. Direct cerebrospinal fluid delivery of an antiretroviral agent using multivesicular liposomes. J Infect Dis 1990; 162(3): 750–2
Bonetti A, Kim S. Pharmacokinetics of an extended-release human interferon alpha-2b formulation. Cancer Chemother Pharmacol 1993; 33(3): 258–61
Qiu J, Wei XH, Geng F, et al. Multivesicular liposome formulations for the sustained delivery of interferon alpha-2b. Acta Pharmacol Sin 2005; 26(11): 1395–401
Li H, An JH, Park JS, et al. Multivesicular liposomes for oral delivery of recombinant human epidermal growth factor. Arch Pharm Res 2005; 28(8): 988–94
Ramprasad MP, Anantharamaiah GM, Garber DW, et al. Sustained-delivery of an apolipoprotein E-peptidomimetic using multivesicular liposomes lowers serum cholesterol levels. J Control Release 2002; 79(1–3): 207–18
Kaplan JG, DeSouza TG, Farkash A, et al. Leptomeningeal metastases: comparison of clinical features and laboratory data of solid tumors, lymphomas and leukemias. J Neurooncol 1990; 9(3): 225–9
Zimm S, Collins JM, Miser J, et al. Cytosine arabinoside cerebrospinal fluid kinetics. Clin Pharmacol Ther 1984; 35(6): 826–30
Bleyer WA, Dedrick RL. Clinical pharmacology of intrathecal methotrexate: I. Pharmacokinetics in nontoxic patients after lumbar injection. Cancer Treat Rep 1977; 61(4): 703–8
Graham FL, Whitmore GF. The effect of 1-beta-D-arabinofuranosylcytosine on growth, viability, and DNA synthesis of mouse L-cells. Cancer Res 1970; 30(11): 2627–35
Kim S, Kim DJ, Howell SB. Modulation of the peritoneal clearance of liposomal cytosine arabinoside by blank liposomes. Cancer Chemother Pharmacol 1987; 19(4): 307–10
Kim S, Howell SB. Multivesicular liposomes containing cytarabine for slow-release Sc administration. Cancer Treat Rep 1987; 71(5): 447–50
Kim S, Kim DJ, Geyer MA, et al. Multivesicular liposomes containing 1-beta-D-arabinofuranosylcytosine for slow-release intrathecal therapy. Cancer Res 1987; 47(15): 3935–7
Kim S, Khatibi S, Howell SB, et al. Prolongation of drug exposure in cerebrospinal fluid by encapsulation into DepoFoam. Cancer Res 1993; 53(7): 1596–8
Kim S, Chatelut E, Kim JC, et al. Extended CSF cytarabine exposure following intrathecal administration of DTC 101. J Clin Oncol 1993; 11(11): 2186–93
Chamberlain MC, Khatibi S, Kim JC, et al. Treatment of leptomeningeal metastasis with intraventricular administration of depot cytarabine (DTC 101): a phase I study. Arch Neurol 1993; 50(3): 261–4
Chamberlain MC, Kormanik P, Howell SB, et al. Pharmacokinetics of intralumbar DTC-101 for the treatment of leptomeningeal metastases. Arch Neurol 1995; 52(9): 912–7
Bomgaars L, Geyer JR, Franklin J, et al. Phase I trial of intrathecal liposomal cytarabine in children with neoplastic meningitis. J Clin Oncol 2004; 22(19): 3916–21
Glantz MJ, LaFollette S, Jaeckle KA, et al. Randomized trial of a slow-release versus a standard formulation of cytarabine for the intrathecal treatment of lymphomatous meningitis. J Clin Oncol 1999; 17(10): 3110–6
Glantz MJ, Jaeckle KA, Chamberlain MC, et al. A randomized controlled trial comparing intrathecal sustained-release cytarabine (DepoCyt) to intrathecal methotrexate in patients with neoplastic meningitis from solid tumors. Clin Cancer Res 1999; 5(11): 3394–402
Jaeckle KA, Phuphanich S, Bent MJ, et al. Intrathecal treatment of neoplastic meningitis due to breast cancer with a slowrelease formulation of cytarabine. Br J Cancer 2001; 84(2): 157–63
Cole BF, Glantz MJ, Jaeckle KA, et al. Quality-of-life-adjusted survival comparison of sustained-release cytosine arabinoside versus intrathecal methotrexate for treatment of solid tumor neoplastic meningitis. Cancer 2003; 97(12): 3053–60
Jaeckle KA, Batchelor T, O’Day SJ, et al. An open label trial of sustained-release cytarabine (DepoCyt) for the intrathecal treatment of solid tumor neoplastic meningitis. J Neurooncol 2002; 57(3): 231–9
Apfelbaum JL, Chen C, Mehta SS, et al. Postoperative pain experience: results from a national survey suggest postoperative pain continues to be undermanaged. Anesth Analg 2003; 97(2): 534–40
Svensson I, Sjostrom B, Haljamae H. Assessment of pain experiences after elective surgery. J Pain Symptom Manage 2000; 20(3): 193–201
Shea RA, Brooks JA, Dayhoff NE, et al. Pain intensity and postoperative pulmonary complications among the elderly after abdominal surgery. Heart Lung 2002; 31(6): 440–9
Tsui SL, Law S, Fok M, et al. Postoperative analgesia reduces mortality and morbidity after esophagectomy. Am J Surg 1997; 173(6): 472–8
Pavlin DJ, Chen C, Penaloza DA, et al. Pain as a factor complicating recovery and discharge after ambulatory surgery. Anesth Analg 2002; 95(3): 627–34
Perkins FM, Kehlet H. Chronic pain as an outcome of surgery: a review of predictive factors. Anesthesiology 2000; 93(4): 1123–33
Quality improvement guidelines for the treatment of acute pain and cancer pain. American Pain Society Quality of Care Committee. JAMA 1995; 274(23): 1874–80
Agency for Health Care Policy and Research. Acute pain management: operative or medical procedures and trauma. Clinical Practice Guideline. Rockville (MD): Agency for Health Care Policy and Research, 1992
Richman JM, Wu CL. Epidural analgesia for postoperative pain. Anesthesiol Clin North America 2005; 23(1): 125–40
Jain S, Datta S. Postoperative pain management. Chest Surg Clin N Am 1997; 7(4): 773–99
Sinatra RS, Torres J, Bustos AM. Pain management after major orthopaedic surgery: current strategies and new concepts. J Am Acad Orthop Surg 2002; 10(2): 117–29
Block BM, Liu SS, Rowlingson AJ, et al. Efficacy of postoperative epidural analgesia: a meta-analysis. JAMA 2003; 290(18): 2455–63
Rawal N, Sjostrand U, Dahlstrom B. Postoperative pain relief by epidural morphine. Anesth Analg 1981; 60(10): 726–31
Fuller JG, McMorland GH, Douglas MJ, et al. Epidural morphine for analgesia after caesarean section: a report of 4880 patients. Can J Anaesth 1990; 37(6): 636–40
Rauck RL, Raj PP, Knarr DC, et al. Comparison of the efficacy of epidural morphine given by intermittent injection or continuous infusion for the management of postoperative pain. Reg Anesth 1994; 19(5): 316–24
Pastor MC, Sanchez MJ, Casas MA, et al. Thoracic epidural analgesia in coronary artery bypass graft surgery: seven years’ experience. J Cardiothorac Vasc Anesth 2003; 17(2): 154–9
Flisberg P, Rudin A, Linner R, et al. Pain relief and safety after major surgery: a prospective study of epidural and intravenous analgesia in 2696 patients. Acta Anaesthesiol Scand 2003; 47(4): 457–65
Pan PH, Bogard TD, Owen MD. Incidence and characteristics of failures in obstetric neuraxial analgesia and anesthesia: a retrospective analysis of 19,259 deliveries. Int J Obstet Anesth 2004; 13(4): 227–33
Horlocker TT, Wedel DJ, Benzon H, et al. Regional anesthesia in the anticoagulated patient: defining the risks (the second ASRA Consensus Conference on Neuraxial Anesthesia and Anticoagulation). Reg Anesth Pain Med 2003; 28(3): 172–97
Brooks K, Pasero C, Hubbard L, et al. The risk of infection associated with epidural analgesia. Infect Control Hosp Epidemiol 1995; 16(12): 725–8
Kim T, Kim J, Kim S. Extended-release formulation of morphine for subcutaneous administration. Cancer Chemother Pharmacol 1993; 33(3): 187–90
Kim T, Murdande S, Gruber A, et al. Sustained-release morphine for epidural analgesia in rats. Anesthesiology 1996; 85(2): 331–8
Yaksh TL, Provencher JC, Rathbun ML, et al. Pharmacokinetics and efficacy of epidurally delivered sustained-release encapsulated morphine in dogs. Anesthesiology 1999; 90(5): 1402–12
Yaksh TL, Provencher JC, Rathbun ML, et al. Safety assessment of encapsulated morphine delivered epidurally in a sustained-release multivesicular liposome preparation in dogs. Drug Deliv 2000; 7(1): 27–36
Viscusi ER, Martin G, Hartrick CT, et al. Forty-eight hours of postoperative pain relief after total hip arthroplasty with a novel, extended-release epidural morphine formulation. Anesthesiology 2005; 102(5): 1014–22
Carvalho B, Riley E, Cohen SE, et al. Single-dose, sustainedrelease epidural morphine in the management of postoperative pain after elective cesarean delivery: results of a multicenter randomized controlled study. Anesth Analg 2005; 100(4): 1150–8
Hartrick CT, Martin G, Kantor G, et al. Evaluation of a singledose, extended-release epidural morphine formulation for pain after knee arthroplasty. J Bone Joint Surg Am 2006; 88(2): 273–81
Viscusi ER, Kopacz D, Hartrick C, et al. Single-dose extendedrelease epidural morphine pharmacokinetics. Am J Ther 2006; 13(5): 423–31
Gould EM, Manvelian G. A pooled analysis of extended-release epidural morhphine pharmacokinetics [abstract no. A770]. Anesthesiology 2005; 103: 28
American Society of Anesthesiologists Task Force on Acute Pain Management. Practice guidelines for acute pain management in the perioperative setting: an updated report by the American Society of Anesthesiologists Task Force on Acute Pain Management. Anesthesiology 2004; 100(6): 1573–81
Contopoulos-Ioannidis DG, Giotis ND, Baliatsa DV, et al. Extended-interval aminoglycoside administration for children: a meta-analysis. Pediatrics 2004; 114(1): e111–8
Barza M, Ioannidis JP, Cappelleri JC, et al. Single or multiple daily doses of aminoglycosides: a meta-analysis. BMJ 1996; 312(7027): 338–45
Ali MZ, Goetz MB. A meta-analysis of the relative efficacy and toxicity of single daily dosing versus multiple daily dosing of aminoglycosides. Clin Infect Dis 1997; 24(5): 796–809
Acknowledgements
No sources of funding were used to assist in the preparation of this review. The authors have no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Angst, M.S., Drover, D.R. Pharmacology of Drugs Formulated with DepoFoam™. Clin Pharmacokinet 45, 1153–1176 (2006). https://doi.org/10.2165/00003088-200645120-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200645120-00002