Abstract
Background
The involvement of lipids in carcinogenic and developmental processes has been reported in some malignancies, but their roles in gastric cancer remain to be analyzed. In this study, we compared the lipid content of gastric cancer tissue and adjacent nonneoplastic mucosa using imaging mass spectrometry.
Methods
Mass spectra were acquired from 12 sections of human gastric cancer tissue and adjacent nonneoplastic mucosa using a matrix-assisted laser desorption-ionization time-of-flight tandem mass spectrometry type mass spectrometer equipped with a 355 nm Nd:YAG laser. Protein expression of lysophosphatidylcholine acyltransferase 1 (LPCAT1), which converts lysophosphatidylcholine (LPC) to phosphatidylcholine (PC) in the presence of acyl-CoA in Lands’ cycle, was immunohistochemically analyzed in 182 gastric cancer specimens.
Results
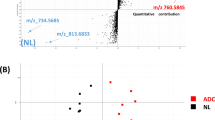
The averaged mass spectra from the cancer tissue and nonneoplastic mucosa were identical. Most of the signals that differed between cancer tissue and nonneoplastic mucosa corresponded to phospholipids, the majority of which were PC and LPC. Two signals, m/z 798.5 and 496.3, were higher and lower, respectively, in cancer tissues, predominantly in differentiated adenocarcinoma. A database search enabled identification of the ions at m/z 798.5 and m/z 496.3 as potassium-adducted PC (16:0/18:1) and proton-adducted LPC (16:0), respectively. Immunohistochemical analysis revealed that LPCAT1 was highly expressed in cancer lesions compared to nonneoplastic mucosa, predominantly in differentiated adenocarcinoma. LPCAT1 expression levels correlated positively with tumor differentiation and negatively with tumor depth, lymph node metastasis, and tumor stage.
Conclusions
Overexpressed LPCAT1 protein in gastric mucosa appears to play important roles in the tumorigenic process of gastric cancer by converting LPC to PC.





Similar content being viewed by others
References
Cancer statistics in Japan. http://www.fpcr.or.jp/publication/statistics.html (2010).
Yamaoka Y. Mechanisms of disease: Helicobacter pylori virulence factors. Nat Rev Gastroenterol Hepatol. 2010;7:629–641.
Santarelli RL, Pierre F, Corpet DE. Processed meat and colorectal cancer: a review of epidemiologic and experimental evidence. Nutr Cancer. 2008;60:131–144.
Ploss A, Dubuisson J. New advances in the molecular biology of hepatitis C virus infection: towards the identification of new treatment targets. Gut. 2012;61(Suppl 1):i25–i35.
Miki D, Ochi H, Hayes CN, Aikata H, Chayama K. Hepatocellular carcinoma: towards personalized medicine. Cancer Sci. 2012;103:846–850.
Mansilla F, da Costa KA, Wang S, et al. Lysophosphatidylcholine acyltransferase 1 (LPCAT1) overexpression in human colorectal cancer. J Mol Med. 2009;87:85–97.
Stoeckli M, Chaurand P, Hallahan DE, Caprioli RM. Imaging mass spectrometry: a new technology for the analysis of protein expression in mammalian tissues. Nat Med. 2001;7:493–496.
Shimma S, Sugiura Y, Hayasaka T, Zaima N, Matsumoto M, Setou M. Mass imaging and identification of biomolecules with MALDI-QIT-TOF-based system. Anal Chem. 2008;80:878–885.
Gessel MM, Norris JL, Caprioli RM. MALDI imaging mass spectrometry: spatial molecular analysis to enable a new age of discovery. J Proteomics. 2014;107:71–82.
Neubert P, Walch A. Current frontiers in clinical research application of MALDI imaging mass spectrometry. Expert Rev Proteomics. 2013;10:259–273.
Eriksson C, Masaki N, Yao I, Hayasaka T, Setou M. MALDI imaging mass spectrometry—a mini review of methods and recent developments. Mass Spectrom (Tokyo). 2013;2:S0022.
Morita Y, Ikegami K, Goto-Inoue N, et al. Imaging mass spectrometry of gastric carcinoma in formalin-fixed paraffin-embedded tissue microarray. Cancer Sci. 2010;101:267–273.
Tanaka H, Zaima N, Yamamoto N, et al. Imaging mass spectrometry reveals unique lipid distribution in primary varicose veins. Eur J Vasc Endovasc Surg. 2010;40:657–663.
Tanaka H, Zaima N, Yamamoto N, et al. Distribution of phospholipid molecular species in autogenous access grafts for hemodialysis analyzed using imaging mass spectrometry. Anal Bioanal Chem. 2011;400:1873–1880.
Tanaka H, Zaima N, Sasaki T, et al. Loss of lymphatic vessels and regional lipid accumulation is associated with great saphenous vein incompetence. J Vasc Surg. 2012;55:1440–1448.
Zhou X, Lawrence TJ, He Z, Pound CR, Mao J, Bigler SA. The expression level of lysophosphatidylcholine acyltransferase 1 (LPCAT1) correlates to the progression of prostate cancer. Exp Mol Pathol. 2012;92:105–110.
Setoguchi T, Kikuchi H, Yamamoto M, et al. Microarray analysis identifies versican and CD9 as potent prognostic markers in gastric gastrointestinal stromal tumors. Cancer Sci. 2011;102:883–889.
Domingues P, Domingues MR, Amado FM, Ferrer-Correia AJ. Characterization of sodiated glycerol phosphatidylcholine phospholipids by mass spectrometry. Rapid Commun Mass Spectrom. 2001;15:799–804.
Hsu FF, Turk J, Thukkani AK, Messner MC, Wildsmith KR, Ford DA. Characterization of alkylacyl, alk-1-enylacyl and lyso subclasses of glycerophosphocholine by tandem quadrupole mass spectrometry with electrospray ionization. J Mass Spectrom. 2003;38:752–763.
Pulfer M, Murphy RC. Electrospray mass spectrometry of phospholipids. Mass Spectrom Rev. 2003;22:332–364.
Weintraub ST, Pinckard RN, Hail M. Electrospray ionization for analysis of platelet-activating factor. Rapid Commun Mass Spectrom. 1991;5:309–311.
Reis A, Domingues P, Ferrer-Correia AJ, Domingues MR. Tandem mass spectrometry of intact oxidation products of diacylphosphatidylcholines: evidence for the occurrence of the oxidation of the phosphocholine head and differentiation of isomers. J Mass Spectrom. 2004;39:1513–1522.
Satoh T, Kubo A, Shimma S, Toyoda M. Mass spectrometry imaging and structural analysis of lipids directly on tissue specimens by using a spiral orbit type tandem time-of-flight mass spectrometer, spiralTOF-TOF. Mass Spectrom (Tokyo). 2012;1:A0013.
Chen X, Hyatt BA, Mucenski ML, Mason RJ, Shannon JM. Identification and characterization of a lysophosphatidylcholine acyltransferase in alveolar type II cells. Proc Natl Acad Sci USA. 2006;103:11724–11729.
Lands WE. Metabolism of glycerolipids. 2. The enzymatic acylation of lysolecithin. J Biol Chem. 1960;235:2233–2237.
Soupene E, Fyrst H, Kuypers FA. Mammalian acyl-CoA: lysophosphatidylcholine acyltransferase enzymes. Proc Natl Acad Sci USA. 2008;105:88–93.
Nakanishi H, Shindou H, Hishikawa D, et al. Cloning and characterization of mouse lung-type acyl-CoA: lysophosphatidylcholine acyltransferase 1 (LPCAT1). Expression in alveolar type II cells and possible involvement in surfactant production. J Biol Chem. 2006;281:20140–20147.
Schone C, Hofler H, Walch A. MALDI imaging mass spectrometry in cancer research: combining proteomic profiling and histological evaluation. Clin Biochem. 2013;46:539–545.
van Meer G. Cellular lipidomics. EMBO J. 2005;24:3159–3165.
Stubbs CD, Smith AD. The modification of mammalian membrane polyunsaturated fatty acid composition in relation to membrane fluidity and function. Biochim Biophys Acta. 1984;779:89–137.
Exton JH. Phosphatidylcholine breakdown and signal transduction. Biochim Biophys Acta. 1994;1212:26–42.
Aboagye EO, Bhujwalla ZM. Malignant transformation alters membrane choline phospholipid metabolism of human mammary epithelial cells. Cancer Res. 1999;59:80–84.
Morita Y, Sakaguchi T, Ikegami K, et al. Lysophosphatidylcholine acyltransferase 1 altered phospholipid composition and regulated hepatoma progression. J Hepatol. 2013;59:292–299.
Wu M, Tu T, Huang Y, Cao Y. Suppression subtractive hybridization identified differentially expressed genes in lung adenocarcinoma: ERGIC3 as a novel lung cancer–related gene. BMC Cancer. 2013;13:44.
Grupp K, Sanader S, Sirma H, et al. High lysophosphatidylcholine acyltransferase 1 expression independently predicts high risk for biochemical recurrence in prostate cancers. Mol Oncol. 2013;7:1001–1011.
Endoh Y, Tamura G, Watanabe H, Ajioka Y, Motoyama T. The common 18–base pair deletion at codons 418–423 of the E-cadherin gene in differentiated-type adenocarcinomas and intramucosal precancerous lesions of the stomach with the features of gastric foveolar epithelium. J Pathol. 1999;189:201–206.
Saito A, Shimoda T, Nakanishi Y, Ochiai A, Toda G. Histologic heterogeneity and mucin phenotypic expression in early gastric cancer. Pathol Int. 2001;51:165–171.
Smith MG, Hold GL, Tahara E, El-Omar EM. Cellular and molecular aspects of gastric cancer. World J Gastroenterol. 2006;12:2979–2990.
Gomceli I, Demiriz B, Tez M. Gastric carcinogenesis. World J Gastroenterol. 2012;18:5164–5170.
Acknowledgment
This work was supported in part by the Ministry of Education, Culture, Sports, Science, and Technology of Japan Grants-in-Aid 22791270 (H. Kikuchi), 20670004 (M. Setou), and 21390376 (H. Konno), and the Development of System and Technology for Advanced Measurement and Analysis (M. Setou).
Disclosure
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Takashi Uehara and Hirotoshi Kikuchi have contributed equally to this article, and both should be considered first author.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Uehara, T., Kikuchi, H., Miyazaki, S. et al. Overexpression of Lysophosphatidylcholine Acyltransferase 1 and Concomitant Lipid Alterations in Gastric Cancer. Ann Surg Oncol 23 (Suppl 2), 206–213 (2016). https://doi.org/10.1245/s10434-015-4459-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4459-6