Abstract
Background
The duodenum is a rare site of primary gastrointestinal stromal tumor (GIST). Overall (OS) and disease-free survival (DFS) after limited resection (LR) versus pancreaticoduodenectomy (PD) were studied.
Methods
All patients who underwent surgery for primary, localized duodenal GIST between 2000 and 2011 were identified from four prospective institutional databases. OS and DFS were calculated by Kaplan–Meier method. Univariate analysis was performed.
Results
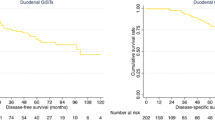
Eighty-four patients (median follow-up 42 months) underwent LR (n = 56, 67 %) or PD (n = 28, 33 %). Patients in the PD group had a larger median tumor size (7 cm vs. 5 cm, p = 0.024) and higher mitotic count (39 % vs. 19 % >5/50 high-power fields, p = 0.05). Complications were observed in five patients (9 %) in the LR group and ten patients (36 %) in the PD group. OS and DFS for the entire cohort were 89 % and 64 % at 5 years, respectively. No difference in outcome between LR and PD were observed. Eleven patients were treated with preoperative IM. A major RECIST response was obtained in nine (80 %), whereas two had stable disease. Twenty-three patients received postoperative Imatinib (IM). A trend toward a better OS in IM-treated patients could be detected only in the high-risk group.
Conclusions
Type of duodenal resection does not impact outcome. The choice should be determined by duodenal site of origin and tumor size. IM may be considered in cases at high risk of recurrence; in neoadjuvant setting, IM might facilitate resection and possibly increase the chance of preserving normal biliary and pancreatic anatomy.



Similar content being viewed by others
References
Joensuu H, Fletcher C, Dimitrijevic S, Silberman S, Roberts P, Demetri G. Management of malignant gastrointestinal stromal tumours. Lancet Oncol. 2002;3:655–64.
Miettinen M, Lasota J. Gastrointestinal stromal tumors—definition, clinical, histological, immunohistochemical, and molecular genetic features and differential diagnosis. Virchows Arch. 2001;438:1–12.
Joensuu H. Gastrointestinal stromal tumor. Ann Oncol. 2006;17:280–286.
Buchs NC, Bucher P, Gervaz P, Ostermann S, Pugin F, Morel P. Segmental duodenectomy for gastrointestinal stromal tumor of the duodenum. World J Gastroenterol. 2010;16:2788–92.
Casali PG, Lost L, Reichardt, Schlemmer M, Blay JY. Gastrointestinal stromal tumours: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol. 2009;20:64–7.
Fiore M, Palassini E, Fumagalli E, et al. Preoperative imatinib mesylate for unresectable or locally advanced primary gastrointestinal stromal tumors (GIST). EJSO. 2009;35:739–45.
Dematteo RP, Ballman KV, Antonescu CR, et al. Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: a randomised, double-blind, placebo-controlled trial. Lancet. 2009;373:1097–104.
Joensuu H, Eriksson M, Sundby Hall K, et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA. 2012;307(12):1265–72.
Joensuu H. Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Hum Pathol. 2008;39(10):1411–9.
Firth D. Bias reduction of maximum likelihood estimates. Biometrika. 1993;80:27–38.
Heinze G, Schemper M. A solution to the problem of monotone likelihood in Cox regression. Biometrics. 2001;7:114–9.
Heinze G, Ploner M. SAS and SPLUS programs to perform Cox regression without convergence problems. Comput Methods Programs Biomed. 2002;67:217–23.
Beham A, Schaefer IM, Cameron S, von Hammerstein K, Füzesi L, Ramadori G, Ghadimi MB. Duodenal GIST: a single-center experience. Int J Colorectal Dis. 2012. doi:10.1007/s00384-012-1432-8.
Chung JC, Chu CW, Cho GS, Shin EJ, Lim CW, Kim HC, Song PO. Management and outcome of gastrointestinal stromal tumors of the duodenum. J Gastrointest Surg. 2010;14:880–3.
Tien YW, Lee CY, Huang CC, Hu RH, Lee PH. Surgery for gastrointestinal stromal tumors of the duodenum. Ann Surg Oncol. 2010;17:109–14.
Gronchi A, Raut CP. The combination of Surgery and Imatinib in GIST: a reality for localized tumors at high risk, an open issue for metastatic ones. Ann Surg Oncol. 2012;19:1051–5.
Verweij J, Casali PG, Zalcberg J, et al. Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: randomised trial. Lancet. 2004;364:1127–34.
Blanke C, Rankin C, Demetri GD, et al. Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the KIT receptor tyrosine kinase: S0033. J Clin Oncol. 2008;26:626–32.
Heinrich MC, Corless CL, Blanke CD, et al. Molecular correlates of imatinib resistance in gastrointestinal stromal tumors. J Clin Oncol. 2006;24(29):4764–74.
Heinrich MC, Owzar K, Corless CL, et al. Correlation of kinase genotype and clinical outcome in the North American Intergroup Phase III Trial of imatinib mesylate for treatment of advanced gastrointestinal stromal tumor: CALGB 150105 Study by Cancer and Leukemia Group B and Southwest Oncology Group. J Clin Oncol. 2008;26(33):5360–7.
Corless CL, Ballman BV, Antonescu C, et al. Relation of tumor pathologic and molecular features to outcome after surgical resection of localized primary gastrointestinal stromal tumor (GIST): results of the intergroup phase III trial ACOSOG Z9001. J Clin Oncol. 2010;28(Suppl):10006.
Gastrointestinal Stromal Tumor Meta-Analysis Group (MetaGIST). Comparison of two doses of imatinib for the treatment of unresectable or metastatic gastrointestinal stromal tumors: a meta-analysis of 1,640 patients. J Clin Oncol. 2010;28(7):1247–53.
Debiec-Rychter M, Sciot R, Le Cesne A, et al. KIT mutations and dose selection for imatinib in patients with advanced gastrointestinal stromal tumours. Eur J Cancer. 2006;42(8):1093–103.
Emile JF, Brahimi S, Coindre JM, et al. Frequencies of KIT and PDGFRA mutations in the MolecGIST prospective population-based study differ from those of advanced GISTs. Med Oncol. 2011. doi:10.1007/s12032-011-0074-y.
Disclosures
C.P. Raut received honoraria from Novartis Pharma. U. Roenellenfitsch received travel supports from Novartis Pharma. P. Rukowski received honoraria and travel supports from Novartis Pharma and Pfizer; compensations for advisory boards from Novartis Pharma. P. Hohenberger received honoraria and compensations for advisory boards from Novartis Pharma. A. Gronchi received honoraria and compensations for advisory boards from Novartis Pharma.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Colombo, C., Ronellenfitsch, U., Yuxin, Z. et al. Clinical, Pathological and Surgical Characteristics of Duodenal Gastrointestinal Stromal Tumor and Their Influence on Survival: A Multi-Center Study. Ann Surg Oncol 19, 3361–3367 (2012). https://doi.org/10.1245/s10434-012-2559-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2559-0