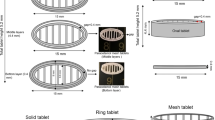
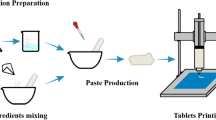
Abstract
Three-dimensional (3D)-printed tablets prepared using powder-based printing techniques like selective laser sintering (SLS) typically disintegrate/dissolve and release the drug within a few minutes because of their inherent porous nature and loose structure. The goal of this study was to demonstrate the suitability of SLS 3DP technology for fabricating sustained-release dosages utilizing Kollidon® SR (KSR), a matrix-forming excipient composed of polyvinyl acetate and polyvinylpyrrolidone (8:2). A physical mixture (PM), comprising 10:85:5 (% w/w) of acetaminophen (ACH), KSR, and Candurin®, was sintered using a benchtop SLS 3D printer equipped with a 2.3-W 455-nm blue visible laser. After optimization of the process parameters and formulation composition, robust 3D-printed tablets were obtained as per the computer-aided design (CAD) model. Advanced solid-state characterizations by powder X-ray diffraction (PXRD) and wide-angle X-ray scattering (WAXS) confirmed that ACH remained in its native crystalline state after sintering. In addition, X-ray micro-computed tomography (micro-CT) studies revealed that the tablets contain a total porosity of 57.7% with an average pore diameter of 24.8 μm. Moreover, SEM images exhibited a morphological representation of the ACH sintered tablets’ exterior surface. Furthermore, the KSR matrix 3D-printed tablets showed a sustained-release profile, releasing roughly 90% of the ACH over 12 h as opposed to a burst release from the free drug and PM. Overall, our work shows for the first time that KSR can be used as a suitable polymer matrix to create sustained-release dosage forms utilizing the digitally controllable SLS 3DP technology, showcasing an alternative technique and pharmaceutical excipient.
Graphical Abstract










Similar content being viewed by others
References
Jamróz W, Szafraniec J, Kurek M, Jachowicz R. 3D Printing in Pharmaceutical and Medical Applications – Recent Achievements and Challenges. Pharm Res. 2018;35(9):1–22. https://doi.org/10.1007/s11095-018-2454-x.
Basit AW, Gaisford S. 3D Printing of Pharmaceuticals [Internet]. Available from: http://www.springer.com/series/8825. Accessed 20 Sept 2022.
Foster KR. 3-Dimensional Printing in Medicine: Hype, Hope, and the Challenge of Personalized Medicine. Philos Eng Technol. 2017;26:211–28. https://doi.org/10.1007/978-3-319-45193-0_16.
Lim SH, Kathuria H, Tan JJY, Kang L. 3D printed drug delivery and testing systems — a passing fad or the future? Adv Drug Deliv Rev. 2018;132:139–68.
Trenfield SJ, Awad A, Madla CM, Hatton GB, Firth J, Goyanes A, et al. Shaping the future: recent advances of 3D printing in drug delivery and healthcare. Expert Opin Drug Deliv. 2019;16(10):1081–94. https://doi.org/10.1080/1742524720191660318.
Robles-Martinez P, Xu X, Trenfield SJ, Awad A, Goyanes A, Telford R, et al. 3D printing of a multi-layered polypill containing six drugs using a novel stereolithographic method. Pharmaceutics. 2019;11(6):274.
Fina F, Goyanes A, Rowland M, Gaisford S, Basit AW. 3D printing of tunable zero-order release Printlets. Polymers 2020;12(8):1769. Available from: https://www.mdpi.com/2073-4360/12/8/1769/htm. Accessed 20 Sept 2022.
Sun Y, Soh S. Printing Tablets with Fully Customizable Release Profiles for Personalized Medicine. Adv Mater. 2015;27(47):7847–53.
Giri BR, Song ES, Kwon J, Lee JH, Park JB, Kim DW. Fabrication of intragastric floating, controlled release 3D printed theophylline tablets using hot-melt extrusion and fused deposition modeling. Pharmaceutics. 2020;12(1):77.
Seoane-Viaño I, Trenfield SJ, Basit AW, Goyanes A. Translating 3D printed pharmaceuticals: From hype to real-world clinical applications. Adv Drug Deliv Rev. 2021;174:553–75.
Awad A, Trenfield SJ, Goyanes A, Gaisford S, Basit AW. Reshaping drug development using 3D printing. Drug Discov Today. 2018;23(8):1547–55.
Fina F, Goyanes A, Gaisford S, Basit AW. Selective laser sintering (SLS) 3D printing of medicines. Int J Pharm. 2017;529(1–2):285–93.
Kulinowski P, Malczewski P, Łaszcz M, Baran E, Milanowski B, Kuprianowicz M, et al. Development of Composite, Reinforced, Highly Drug-Loaded Pharmaceutical Printlets Manufactured by Selective Laser Sintering—In Search of Relevant Excipients for Pharmaceutical 3D Printing. Materials 2022;15(6):2142. Available from: https://www.mdpi.com/1996-1944/15/6/2142/htm
Yuan S, Shen F, Chua CK, Zhou K. Polymeric composites for powder-based additive manufacturing: Materials and applications. Prog Polym Sci. 2019;91:141–68.
Alhnan MA, Okwuosa TC, Sadia M, Wan KW, Ahmed W, Arafat B. Emergence of 3D Printed Dosage Forms: Opportunities and Challenges. Pharm Res. 2016;33(8):1817–32. https://doi.org/10.1007/s11095-016-1933-1.
Thakkar R, Davis DA, Williams RO, Maniruzzaman M. Selective Laser Sintering of a Photosensitive Drug: Impact of Processing and Formulation Parameters on Degradation, Solid State, and Quality of 3D-Printed Dosage Forms. Mol Pharm. 2021;18(10):3894–908. https://doi.org/10.1101/2021.04.09.439089v1.
Fina F, Madla CM, Goyanes A, Zhang J, Gaisford S, Basit AW. Fabricating 3D printed orally disintegrating printlets using selective laser sintering. Int J Pharm. 2018;541(1–2):101–7.
Jamróz W, Kurek M, Łyszczarz E, Szafraniec J, Knapik-Kowalczuk J, Syrek K, et al. 3D printed orodispersible films with Aripiprazole. Int J Pharm. 2017;533(2):413–20.
Awad A, Yao A, Trenfield SJ, Goyanes A, Gaisford S, Basit AW. 3D Printed Tablets (Printlets) with Braille and Moon Patterns for Visually Impaired Patients. Pharmaceutics 2020;12(2):172. Available from: https://www.mdpi.com/1999-4923/12/2/172/htm. Accessed 20 Sept 2022.
Titapiwatanakun V, Tankul J, Basit AW, Gaisford S. Laser irradiation to produce amorphous pharmaceuticals. Int J Pharm. 2016;514(1):282–9.
Titapiwatanakun V, Basit AW, Gaisford S. A New Method for Producing Pharmaceutical Co-crystals: Laser Irradiation of Powder Blends. Cryst Growth Des. 2016;16(6):3307–12. https://doi.org/10.1021/acs.cgd.6b00289.
Wang J, Goyanes A, Gaisford S, Basit AW. Stereolithographic (SLA) 3D printing of oral modified-release dosage forms. Int J Pharm. 2016;503(1–2):207–12.
Giri BR, Poudel S, Kim DW. Cellulose and its derivatives for application in 3D printing of pharmaceuticals. J Pharm Investig. 2021;51(1):1–22. https://doi.org/10.1007/s40005-020-00498-5.
Goodridge RD, Tuck CJ, Hague RJM. Laser sintering of polyamides and other polymers. Prog Mater Sci. 2012;57(2):229–67.
Evans R, Bourell D, Beaman JJ 2005 I, 2005 undefined. SLS materials development method for rapid manufacturing. repositories.lib.utexas.edu [Internet]. [cited 2022 Aug 29]; Available from: https://repositories.lib.utexas.edu/handle/2152/80056. Accessed 20 Sept 2022.
Gueche YA, Sanchez-Ballester NM, Cailleaux S, Bataille B, Soulairol I. Selective Laser Sintering (SLS), a New Chapter in the Production of Solid Oral Forms (SOFs) by 3D Printing. Pharmaceutics 2021;13(8):1212. Available from: https://www.mdpi.com/1999-4923/13/8/1212/htm. Accessed 20 Sept 2022.
Bühler V. Kollidon: Polyvinylpyrrolidone excipients for the pharmaceutical industry. BASF-The Chemical Company. 2008.
Davis DA, Thakkar R, Su Y, Williams RO, Maniruzzaman M. Selective Laser Sintering 3-Dimensional Printing as a Single Step Process to Prepare Amorphous Solid Dispersion Dosage Forms for Improved Solubility and Dissolution Rate. J Pharm Sci. 2021;110(4):1432–43.
Kulinowski P, Malczewski P, Pesta E, Łaszcz M, Mendyk A, Polak S, et al. Selective laser sintering (SLS) technique for pharmaceutical applications—Development of high dose controlled release printlets. Addit Manuf. 2021;38: 101761.
Awad A, Fina F, Goyanes A, Gaisford S, Basit AW. 3D printing: Principles and pharmaceutical applications of selective laser sintering. Int J Pharm. 2020;586: 119594.
Brika SE, Letenneur M, Dion CA, Brailovski V. Influence of particle morphology and size distribution on the powder flowability and laser powder bed fusion manufacturability of Ti-6Al-4V alloy. Addit Manuf. 2020;31: 100929.
Dong Z, Liu Y, Wen W, Ge J, Liang J. Effect of hatch spacing on Melt pool and as-built quality during selective laser melting of stainless steel: Modeling and experimental approaches. Materials 2018;12(1):50. Available from: https://www.mdpi.com/1996-1944/12/1/50/htm. Accessed 20 Sept 2022.
Senthilkumaran K, Pandey PM, Rao PVM. Influence of building strategies on the accuracy of parts in selective laser sintering. Mater Des. 2009;30(8):2946–54.
Lekurwale S, Karanwad T, Banerjee S. Selective laser sintering (SLS) of 3D printlets using a 3D printer comprised of IR/red-diode laser. Ann 3D Print Med. 2022;6:100054.
Sinico M, Jadhav SD, Witvrouw A, Vanmeensel K, Dewulf W. A micro-computed tomography comparison of the porosity in additively fabricated CuCr1 alloy parts using virgin and surface-modified powders. Materials 2021;14(8):1995. Available from: https://www.mdpi.com/1996-1944/14/8/1995/htm. Accessed 20 Sept 2022.
du Plessis A, Yadroitsev I, Yadroitsava I, le Roux SG. X-Ray Microcomputed Tomography in Additive Manufacturing: A Review of the Current Technology and Applications. 3D Print Addit Manuf. 2018;5(3):227–47. https://doi.org/10.1089/3dp.2018.0060.
Allahham N, Fina F, Marcuta C, Kraschew L, Mohr W, Gaisford S, et al. Selective Laser Sintering 3D Printing of Orally Disintegrating Printlets Containing Ondansetron. Pharmaceutics 2020;12(2):110. Available from: https://www.mdpi.com/1999-4923/12/2/110/htm. Accessed 20 Sept 2022.
Zapata F, López-Fernández A, Ortega-Ojeda F, Quintanilla G, García-Ruiz C, Montalvo G. Introducing ATR-FTIR Spectroscopy through Analysis of Acetaminophen Drugs: Practical Lessons for Interdisciplinary and Progressive Learning for Undergraduate Students. J Chem Educ. 2021;98(8):2675–86. https://doi.org/10.1021/acs.jchemed.0c01231.
Arias JL, Gómez-Gallo A, Delgado ÁV, Gallardo V. Study of the stability of Kollidon® SR suspensions for pharmaceutical applications. Colloids Surf A Physicochem Eng Asp. 2009;338(1–3):107–13.
Ahmed J, Giri BR, Thomas L, Al-Attar H, Maniruzzaman M. Continuous manufacturing of vitamin D3 and iron enriched granules by means of a novel twin-screw dry granulation process. Powder Technol. 2022;412: 117975.
The VBP excipients for, 2008 undefined. Kollidon. pharmacompass.com [Internet]. [cited 2022 Sep 6]; Available from: https://www.pharmacompass.com/pAssets/pdf/edqm/application/gmp-kollidon.pdf. Accessed 20 Sept 2022.
Acknowledgements
The authors would also like to thank Dr. Jessica A. Maisano and Mark Costello for their assistance with X-ray Micro-CT analysis, as well as the University of Texas High-Resolution X-ray CT Facility, which is supported by the NSF EAR-2223808.
Author information
Authors and Affiliations
Contributions
Bhupendra Raj Giri: conceptualization, methodology, formal analysis, investigation, data curation, writing–original draft, writing (review and editing); Mohammed Maniruzzaman: conceptualization, resources, supervision, project administration, funding acquisition.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare the following conflict of interest. All authors are co-inventors of related intellectual property (IP). M.M., an author of this manuscript, holds stock in, serves on a scientific advisory board for, or is a consultant for CoM3D Ltd., (Surrey, UK) and Septum Solutions LLC. (Texas, USA). The terms of this arrangement have been reviewed and approved by the University of Texas at Austin in accordance with its policy on objectivity in research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Giri, B.R., Maniruzzaman, M. Fabrication of Sustained-Release Dosages Using Powder-Based Three-Dimensional (3D) Printing Technology. AAPS PharmSciTech 24, 4 (2023). https://doi.org/10.1208/s12249-022-02461-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-022-02461-z