Abstract
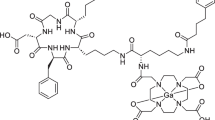
Gambogic acid (GA) has been proven to be a potent chemotherapeutic agent for the treatment of lung cancer in clinical trials. However, GA is limited in its therapeutic value by properties such as poor water solubility and low chemical stability. In clinical trials, cationic arginine (Arg) was added to solubilize GA, and this may also cause other side effects. Here, we have designed and developed a more efficient human serum albumin (HSA)-based delivery system for GA with low toxicity which helps improve its solubility, chemical stability and increases its antitumor efficacy. The GA-HSA nanoparticles (NPs) were prepared by albumin-bound (nabTM) technology, with a particle size of 135.2 ± 35.03 nm, a zeta potential of −21.81 ± 1.24 mV, and a high entrapment efficiency. Compared with GA-Arg solution, the physical and chemical stability of the NPs were improved when stored at pH 7.4 in PBS or freeze-dried. The in vitro drug release showed that GA-HSA NPs had a more sustained release than GA-Arg solution. Furthermore, HSA NPs improved the therapeutic efficacy of GA and were less toxic compared with GA-Arg solution in A549-bearing mice. Therefore, this delivery system is a promising polymeric carrier for GA when used for tumor therapy.









Similar content being viewed by others
References
Sesen MB, Kadir T, Alcantara RB, et al. Survival prediction and treatment recommendation with Bayesian techniques in lung cancer. AMIA Symposium. 2012;838–47.
Jemal A, Murray T, Ward E, et al. Cancer statistics, 2005. CA Cancer J Clin. 2005;55:10–30.
Zhu X, Zhang H, Lin Y, et al. Mechanisms of gambogic acid-induced apoptosis in non-small cell lung cancer cells in relation to transferring receptors. J Chemother. 2009;21(6):666–72.
Yang Y, Yang L, You QD, et al. Differential apoptotic induction of gambogic acid, a novel anticancer natural product, on hepatoma cells and normal hepatocytes. Cancer Lett. 2007;256(2):259–66.
Pandey MK, Sung B, Ahn KS, et al. Gambogic acid, a novel ligand for transferrin receptor, potentiates TNF-induced apoptosis through modulation of the nuclear factor-kappaB signaling pathway. Blood. 2007;110(10):3517–25.
Zhao L, Guo QL, You QD, et al. Gambogic acid induces apoptosis and regulates expressions of Bax and Bcl-2 protein in human gastric carcinoma MGC-803 cells. Biol Pharm Bull. 2004;27(7):998–1003.
Chen J, Gu HY, Lu N, et al. Microtubule depolymerization and phosphorylation of c-Jun N-terminal kinase-1 and p38 were involved in gambogic acid induced cell cycle arrest and apoptosis in human breast carcinoma MCF-7 cells. Life Sci. 2008;83(3–4):99–110.
Guo Q, Qi Q, You Q, et al. Toxicological studies of gambogic acid and its potential targets in experimental animals. Basic Clin Pharmacol Toxicol. 2006;99(2):178–84.
Qi Q, You Q, Gu H, et al. Studies on the toxicity of gambogic acid in rats. J Ethnopharmacol. 2008;117(3):433–8.
Hao K, Liu XQ, Wang GJ, et al. Pharmacokinetics, tissue distribution and excretion of gambogic acid in rats. Eur J Drug Metab Pharmacokinet. 2007;32(2):63–8.
Han QB, Cheung S, Tai J, et al. Stability and cytotoxicity of gambogic acid and its derivative, gambogoic acid. Biol Pharm Bull. 2005;28(12):2335–7.
Lundberg BB, Risovic V, Ramaswamy M, et al. A lipophilic paclitaxel derivative incorporated in a lipid emulsion for parenteral administration. J Control Release. 2003;86(1):93–100.
Peer D, Karp JM, Hong S, et al. Nanocarriers as an emerging platform for cancer therapy. Nat Nanotechnol. 2007;2(12):751–60.
Bae Y, Kataoka K. Intelligent polymeric micelles from functional poly(ethylene glycol)-poly(amino acid) block copolymers. Adv Drug Deliv Rev. 2009;61(10):768–84.
Egusquiaguirre SP, Igartua M, Hernández RM, et al. Nanoparticle delivery systems for cancer therapy: advances in clinical and preclinical research. Clin Transl Oncol. 2012;14(2):83–93.
Wiradharma N, Zhang Y, Venkataraman S, et al. Self-assembled polymer nanostructures for delivery of anticancer therapeutics. Nano Today. 2009;4(4):302–17.
Galisteo-González F, Molina-Bolívar JA. Systematic study on the preparation of BSA nanoparticles. Colloids Surf B: Biointerfaces. 2014;123(139):286–92.
Fu Q, Sun J, Zhang W, et al. Nanoparticle albumin-bound (NAB) technology is a promising method for anti-cancer drug delivery. Recent Pat Anticancer Drug Discov. 2009;4(3):262–72.
Gao S, Sun J, Fu D, et al. Preparation, characterization and pharmacokinetic studies of tacrolimus-dimethyl-β-cyclodextrin inclusion complex-loaded albumin nanoparticles. Int J Pharm. 2012;427(2):410–6.
Zhang ZH, Wang XP, Aym WY, et al. Studies on lactoferrin nanoparticles of gambogic acid for oral delivery. Drug Deliv. 2013;20(1):86–93.
Elzoghby AO, Samy WM, Elgindy NA. Albumin-based nanoparticles as potential controlled release drug delivery systems. J Control Release. 2012;157(2):168–82.
Zhao D, Zhao X, Zu Y, et al. Preparation, characterization, and in vitro targeted delivery of folate-decorated paclitaxel-loaded bovine serum albumin nanoparticles. Int J Nanomedicine. 2010;5(4):669–77.
Zu Y, Yu Z, Zhao X, et al. Optimization of the preparation process of vinblastine sulfate (VBLS)-loaded folate-conjugated bovine serum albumin (BSA) nanoparticles for tumor-targeted drug delivery using response surface methodology (RSM). Inter J Nanomedicine. 2008;4(1):321–33.
Rothweiler F, Michaelis M, et al. Preparation, characterization and maintenance of drug efficacy of doxorubicin-loaded human serum albumin (HSA) nanoparticles. Int J Pharm. 2007;341(1–2):207–14.
Wu L, Zhang J, Watanabe W. Physical and chemical stability of drug nanoparticles. Adv Drug Deliv Rev. 2011;63(6):456–69.
Ge F, Chen C, Liu D, et al. Study on the interaction between theasinesin and human serum albumin by fluorescence spectroscopy. J Lumin. 2010;130(1):168–73.
Li C, Zhang D, Guo H, et al. Preparation and characterization of galactosylated bovine serum albumin nanoparticles for liver-targeted delivery of oridonin. Int J Pharm. 2013;448(1):79–86.
Liu F, Li M, Liu C, et al. PH-sensitive self-assembled carboxymethyl chitosan-modified DNA/polyethylenimine complexes for efficient gene delivery. J Biomed Nanotechnol. 2014;10(11):3397–406.
Anhorn MG, Mahler HC, Langer K. Freeze drying of human serum albumin (HSA) nanoparticles with different excipients. Int J Pharm. 2008;363(1–2):162–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Y., Yang, Z., Tan, X. et al. Development of a More Efficient Albumin-Based Delivery System for Gambogic Acid with Low Toxicity for Lung Cancer Therapy. AAPS PharmSciTech 18, 1987–1997 (2017). https://doi.org/10.1208/s12249-016-0670-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-016-0670-4