Abstract
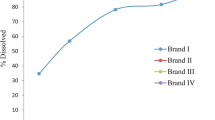
Controlled-release (CR) tablet formulation of olanzapine was developed using a binary mixture of Methocel® K100 LV-CR and Ethocel® standard 7FP premium by the dry granulation slugging method. Drug release kinetics of CR tablet formulations F1, F2, and F3, each one suitably compressed for 9-, 12-, and 15-kg hardness, were determined in a dissolution media of 0.1 N HCl (pH 1.5) and phosphate buffer (pH 6.8) using type II dissolution apparatus with paddles run at 50 rpm. Ethocel® was found to be distinctly controlling drug release, whereas the hardness of tablets and pH of the dissolution media did not significantly affect release kinetics. The CR test tablets containing 30% Methocel® and 60% Ethocel® (F3) with 12-kg hardness exhibited pH-independent zero-order release kinetics for 24 h. In vivo performance of the CR test tablet and conventional reference tablet were determined in rabbit serum using high-performance liquid chromatography coupled with electrochemical detector. Bioavailability parameters including C max, T max, and AUC 0–48 h of both tablets were compared. The CR test tablets produced optimized C max and extended T max (P < 0.05). A good correlation of drug absorption in vivo and drug release in vitro (R 2 = 0.9082) was observed. Relative bioavailability of the test tablet was calculated as 94%. The manufacturing process employed was reproducible and the CR test tablets were stable for 6 months at 40 ± 2°C/75 ± 5% relative humidity. It was concluded that the CR test tablet formulation successfully developed may improve tolerability and patient adherence by reducing adverse effects.





Similar content being viewed by others
References
Ciudad A, Gutiérrez M, Cañas F, Gibert J, Gascón J, Carrasco J-L, et al. Safety and effectiveness of olanzapine in monotherapy: a multivariate analysis of a naturalistic study. Prog Neuro-Psychopharmacol Biol Psychiatry. 2005;29(6):944–51. doi:10.1016/j.pnpbp.2005.04.037.
Lieberman JA. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia: efficacy, safety and cost outcomes of CATIE and other trials. J Clin Psychiatry. 2007;68(2):e04.
Aravagiri M, Ames D, Wirshing W, Marder S. Plasma level monitoring of olanzapine in patients with schizophrenia: determination by high-performance liquid chromatography with electrochemical detection. Ther Drug Monit. 1997;19(3):307.
Callaghan J, Bergstrom R, Ptak L, Beasley C. Olanzapine: pharmacokinetic and pharmacodynamic profile. Clin Pharmacokinet. 1999;37(3):177–93.
Catlow J, Barton R, Clements M, Gillespie T, Goodwin M, Swanson S. Analysis of olanzapine in human plasma utilizing reversed-phase high-performance liquid chromatography with electrochemical detection. J Chromatogr B Biomed Sci Appl. 1995;668(1):85–90.
Mauri M, Volonteri L, Colasanti A, Fiorentini A, De Gaspari I, Bareggi S. Clinical pharmacokinetics of atypical antipsychotics: a critical review of the relationship between plasma concentrations and clinical response. Clin Pharmacokinet. 2007;46(5):359–88.
Hiemke C, Dragicevic A, Gründer G, Hätter S, Sachse J, Vernaleken I, et al. Therapeutic monitoring of new antipsychotic drugs. Ther Drug Monit. 2004;26(2):156.
Leon J, Susce MT, Pan RM, Wedlund PJ, Orrego ML, Diaz FJ. A study of genetic (CYP2D6 and ABCB1) and environmental (drug inhibitors and inducers) variables that may influence plasma risperidone levels. Pharmacopsychiatry. 2007;40(3):93–102.
Lee PE, Sykora K, Gill SS, Mamdani M, Marras C, Anderson G, et al. Antipsychotic medications and drug-induced movement disorders other than parkinsonism: a population-based cohort study in older adults. J Am Geriatr Soc. 2005;53(8):1374–9.
Ray WA, Chung CP, Murray KT, Hall K, Stein CM. Atypical antipsychotic drugs and the risk of sudden cardiac death. N Engl J Med. 2009;360(3):225–35.
Fernandez HH, Trieschmann ME, Friedman JH. Treatment of psychosis in Parkinson’s disease: safety considerations. Drug Saf. 2003;26(9):643–59.
Katz I. Optimizing atypical antipsychotic treatment strategies in the elderly. J Am Geriatr Soc. 2004;52(12 Suppl):S272.
Martindale SS. The complete drug reference. 35th ed. London: The Pharmaceutical Press; 2008.
Svarstad B, Shireman T, Sweeney J. Using drug claims data to assess the relationship of medication adherence with hospitalization and costs. Psychiatr Serv. 2001;52(6):805.
Gerlach J. Improving outcome in schizophrenia: the potential importance of EPS and neuroleptic dysphoria. Ann Clin Psychiatry. 2002;14(1):47–57. doi:10.1023/A:1015276028425.
Weiden P, Mackell J, McDonnell D. Obesity as a risk factor for antipsychotic noncompliance. Schizophr Res. 2004;66(1):51–7.
Keith S. Advances in psychotropic formulations. Prog Neuro-Psychopharmacol Biol Psychiatry. 2006;30(6):996–1008.
Ganesan S, Agambaram V, Randeree F, Eggens I, Huizar K, Meulien D. Switching from other antipsychotics to once-daily extended release quetiapine fumarate in patients with schizophrenia. Curr Med Res Opin. 2008;24(1):21–32.
Cotter L, Eadie M, Hooper W, Lander C, Smith G, Tyrer J. The pharmacokinetics of carbamazepine. Eur J Clin Pharmacol. 1977;12(6):451–6.
Barakat N, Elbagory I, Almurshedi A. Formulation, release characteristics and bioavailability study of oral monolithic matrix tablets containing carbamazepine. AAPS PharmSciTech. 2008;9(3):931–8.
Vivek K, Reddy H, Murthy R. Investigations of the effect of the lipid matrix on drug entrapment, in vitro release, and physical stability of olanzapine-loaded solid lipid nanoparticles. AAPS PharmSciTech. 2007;8(4):16–24.
Merchant H, Shoaib H, Tazeen J, Yousuf R. Once-daily tablet formulation and in vitro release evaluation of cefpodoxime using hydroxypropyl methylcellulose: a technical note. AAPS PharmSciTech. 2006;7(3):178–83.
Agrawal A, Neau S, Bonate P. Wet granulation fine particle ethylcellulose tablets: effect of production variables and mathematical modeling of drug release. AAPS J. 2003;5(2):48–60.
Ravi PR, Kotreka UK, Saha RN. Controlled release matrix tablets of zidovudine: effect of formulation variables on the in vitro drug release kinetics. AAPS PharmSciTech. 2008;9(1):302–13.
Jamzad S, Fassihi R. Development of a controlled release low dose class II drug—Glipizide. Int J Pharm. 2006;312(1–2):24–32.
Higuchi T. Mechanism of sustained-action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. J Pharm Sci. 1963;52(12):1145–9.
Hixson A, Crowell J. Dependence of reaction velocity upon surface and agitation. Ind Eng Chem. 1931;23(10):1160–8.
Korsmeyer R, Gurny R, Doelker E, Buri P, Peppas N. Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm. 1983;15(1):25–35.
Shah VP, Tsong Y, Sathe P, Liu JP. In vitro dissolution profile comparison—statistics and analysis of the similarity factor, f2. Pharm Res. 1998;15(6):889–96.
FDA. Guidance for industry. Dissolution testing of immediate release solid oral dosage forms. Rockville; 1997.
Nagarwal R, Ridhurkar D, Pandit J. In vitro release kinetics and bioavailability of gastroretentive cinnarizine hydrochloride tablet. AAPS PharmSciTech. 2010;11:294–303.
Malkawi A, Al-Ghananeem A, Crooks P. Development of a GC-MS assay for the determination of fentanyl pharmacokinetics in rabbit plasma after sublingual spray delivery. AAPS J. 2008;10(2):261–7.
Charde S, Mudgal M, Kumar L, Saha R. Development and evaluation of buccoadhesive controlled release tablets of lercanidipine. AAPS PharmSciTech. 2008;9(1):182–90.
Kasper SC, Mattiuz EL, Swanson SP, Chiu JA, Johnson JT, Garner CO. Determination of olanzapine in human breast milk by high-performance liquid chromatography with electrochemical detection. J Chromatogr B Biomed Sci Appl. 1999;726(1–2):203–9. doi:10.1016/S0378-4347(99)00017-1.
Shargel L, Yu A. Bioavailability and bioequivalence. In: Shargel L, Yu AB (eds) Applied biopharmaceutics and pharmacokinetics, 4th ed. New York: McGraw-Hill Medical Publishing Company; 1999.
Wagner JG, Nelson E. Kinetic analysis of blood levels and urinary excretion in the absorptive phase after single doses of drug. J Pharm Sci. 1964;53:1392–403.
Robinson DG, Woerner MG, Alvir JMJ, Bilder RM, Hinrichsen GA, Lieberman JA. Predictors of medication discontinuation by patients with first-episode schizophrenia and schizoaffective disorder. Schizophr Res. 2002;57(2–3):209–19.
Rowe RC, Sheskey PJ, Owen SC. Handbook of pharmaceutical excipient. London: Pharmaceutical Press; 2006.
Chowhan Z. Role of binders in moisture-induced hardness increase in compressed tablets and its effect on in vitro disintegration and dissolution. J Pharm Sci. 1980;69(1):1–3.
Shah S, Asghar S, Choudhry M, Akash M, Rehman N, Baksh S. Formulation and evaluation of natural gum-based sustained release matrix tablets of flurbiprofen using response surface methodology. Drug Dev Ind Pharm. 2009;35:1470–8.
Bettini R, Catellani P, Santi P, Massimo G, Peppas N, Colombo P. Translocation of drug particles in HPMC matrix gel layer: effect of drug solubility and influence on release rate. J Control Release. 2001;70(3):383–91.
Peppas NA. Analysis of Fickian and non-Fickian drug release from polymers. Pharm Acta Helv. 1985;60(4):110–1.
Ritger P, Peppas N. A simple equation for description of solute release. II: Fickian and anomalous release from swellable devices. J Control Release. 1987;5(1):37–42.
Ritger P, Peppas N. A simple equation for description of solute release. I: Fickian and non-Fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs. J Control Release. 1987;5(1):23–36.
Rekhi GS, Nellore RV, Hussain AS, Tillman LG, Malinowski HJ, Augsburger LL. Identification of critical formulation and processing variables for metoprolol tartrate extended-release (ER) matrix tablets. J Control Release. 1999;59(3):327–42.
Perez-Marcos B, Ford JL, Armstrong DJ, Elliott PN, Rostron C, Hogan JE. Influence of pH on the release of propranolol hydrochloride from matrices containing hydroxypropylmethylcellulose K4M and carbopol 974. J Pharm Sci. 1996;85(3):330–4.
Fukui A, Fujii R, Yonezawa Y, Sunada H. Analysis of the release process of phenylpropanolamine hydrochloride from ethylcellulose matrix granules III. Effects of the dissolution condition on the release process. Chem Pharm Bull. 2006;54(8):1091–6.
Kasim N, Whitehouse M, Ramachandran C, Bermejo M, Lennernas H, Hussain A, et al. Molecular properties of WHO essential drugs and provisional biopharmaceutical classification. Mol Pharm. 2004;1(1):85–96.
Acknowledgment
We acknowledge the help of Dr. Nadeem Irfan Bukhari, Assistant Professor of Pharmaceutics for analyzing the pharmacokinetic data; the University of Peshawar for financial support, Colorcon Asia Pacific, India for generously gifting Ethocel standard 7FP premium and Methocel K100 LV-CR; Bryon Pharma, and Pakistan for the provision of some chemicals and manufacturing facilities. We are obliged to Danis Pharma, Islamabad, for gifting olanzapine powder.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Badshah, A., Subhan, F. & Rauf, K. Controlled Release Matrix Tablets of Olanzapine: Influence of Polymers on the In Vitro Release and Bioavailability. AAPS PharmSciTech 11, 1397–1404 (2010). https://doi.org/10.1208/s12249-010-9510-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-010-9510-0