Abstract
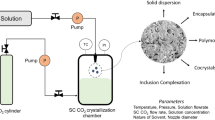
The purpose of this study was to design and build a supercritical CO2 anti-solvent (SAS) unit and use it to produce microparticles of the class II drug carbamazepine. The operation conditions of the constructed unit affected the carbamazepine yield. Optimal conditions were: organic solution flow rate of 0.15 mL/min, CO2 flow rate of 7.5 mL/min, pressure of 4,200 psi, over 3,000 s and at 33°C. The drug solid-state characteristics, morphology and size distribution were examined before and after processing using X-ray powder diffraction and differential scanning calorimetry, scanning electron microscopy and laser diffraction particle size analysis, respectively. The in vitro dissolution of the treated particles was investigated and compared to that of untreated particles. Results revealed a change in the crystalline structure of carbamazepine with different polymorphs co-existing under various operation conditions. Scanning electron micrographs showed a change in the crystalline habit from the prismatic into bundled whiskers, fibers and filaments. The volume weighted diameter was reduced from 209 to 29 μm. Furthermore, the SAS CO2 process yielded particles with significantly improved in vitro dissolution. Further research is needed to optimize the operation conditions of the self-built unit to maximize the production yield and produce a uniform polymorphic form of carbamazepine.







Similar content being viewed by others
References
A. J. Hickey, and D. Ganderton. Pharmaceutical process engineering, Marcel Dekker, New York, 2001.
M. E. Aulton. Pharmaceutics: the science of dosage form design, 2nd ed., Churchhill Livingstone, New York, 2002.
K. Okuyama, and I. W. Lenggoro. Preparation of nanoparticles via spray route. Chem. Eng. Sci. 58(3–6):537–547 (2003).
E. J. Beckman. Supercritical and near-critical CO2 in green chemical synthesis and processing. J. Supercrit. Fluids. 28:121–191 (2003).
E. L. V. Goetheer, M. A. G. Vorstman, and J. T. F. Keurentjes. Opportunities for process intensification using reverse micelles in liquid and supercritical carbon dioxide. Chem. Eng. Sci. 54(10):1589–1596 (1999).
B. W. Wenclawiak, A. Wolf, and S. Wilnewski. Extractability of As-chelates and solubility of different Rh, Pd-chelates in supercritical fluid CO2. In GHBrunner (ed.), Supercritical Fluids as Solvents and Reaction Media, Elsevier, Amsterdam, 2007, pp. 323–340.
Y. P. Sun, H. W. Rollins, B. Jayasundera, M. J. Meziani, and C. E. Bunker. In Y. P. Sun (ed.), Supercritical fluid technology in materials science and engineering: synthesis, properties, and applications, Marcel Dekker, New York, 2002, p. 491.
T. J. Young, S. Mawson, and K. P. Johnston. Rapid expansion from supercritical to aqueous solution to produce submicron suspensions of water-insoluble drugs. Biotechnol. Prog. 16:402–407 (2000).
N. Jovanovic, A. Bouchard, G. W. Hofland, G. J. Witkamp, D. J. A. Crommelin, and W. Jiskoot. Stabilization of proteins in dry powder formulations using supercritical fluid technology. Pharm. Res. 21(11):1955–1969 (2004).
S. Palakodaty, and P. York. Phase behavioral effects on particle formation processes using supercritical fluids. Pharm. Res. 16(7):976–985 (1999).
T. A. Ranjit, and R. B. Gupta. Formation of phenytoin nanoparticles using RESS-SC process. Int. J. Pharm. 308:190–199 (2006).
P. M. Gosselin, R. Thibert, M. Preda, and J. N. McMullen. Polymorphic properties of micronized carbamazepine produced by RESS. Int. J. Pharm. 252:225–233 (2003).
R. Bettini, L. Bonassi, V. Castoro, Rossi, L. Zema, A. Gazzaniga, and F. Giordano. Solubility and conversion of carbamazepine polymorphs in supercritical carbon dioxide. Eur. J. Pharm. Sci. 13(3):281–286 (2001).
M. Moneghini, I. Kikic, D. Voinovich, B. Perissutti, P. Alessi, A. Cortesi, F. Princivalle, and D. Solinas. Study of the solid state of carbamazepine after processing with gas anti-solvent technique. Eur. J. Pharm. Biopharm. 56(2):281–289 (2003).
J. O. Werling, and P. G. Debenedetti. Numerical modeling of mass transfer in the supercritical antisolvent process. J. Supercrit. Fluids. 16(2):167–181 (1999).
S. Budavari, M. J. O’Neil, A. Smith, P. E. Heckelman, and J. F. Kinneary. The Merck index: an encyclopedia of chemicals, drugs and biologicals, 12th ed., Chapman & Hall, London, 1996.
K. Parfitt. Martindale: the complete drug reference, 32rd ed., Pharmaceutical, London, 1999.
FDA. Waiver of in vivo bioavailability and bioequivalence studies for immediate-release solid oral dosage forms based on a biopharmaceutics classification system, 2007. http://www.fda.gov/cder/guidance/3618fnl.pdf (accessed 18/11/07).
M. Moneghini, I. Kikic, D. Voinovich, B. Perissutti, and J. Filipovic-Grcic. Processing of carbamazepine-PEG 4000 solid dispersions with supercritical carbon dioxide: preparation, characterisation, and in vitro dissolution. Int. J. Pharm. 222(1):129–138 (2001).
A. D. Edwards, B. Y. Shekunov, A. Kordikowski, R. T. Forbes, and P. York. Crystallization of pure anhydrous polymorphs of carbamazepine by solution enhanced dispersion with supercritical fluids (SEDS™). J. Pharm. Sci. 90(8):1115–1124 (2001).
P. A. Kikic, A. Cortesi, F. Eva, A. Fogar, M. moneghini, B. Perissutti, and D. Voinovich. Supercritical anti-solvent precipitation processes: different ways for improving the performance of drugs. In A. Bertucco (ed.), Chemical Engineering Transactions, Vol. 2, AIDIC, Milano, 2002, pp. 821–826.
J. C. De La Fuente, C. J. Peters, and A. J. De Swaan. Volume expansion in relation to the gas-antisolvent process. J. Supercrit. Fluids. 17(1):13–23 (2000).
J. C. De La Fuente, A. Shariati, and C. J. Peters. On the selection of optimum thermodynamic conditions for the GAS process. J. Supercrit. Fluids. 32(1)–3:55–61 (2004).
M. Perrut, J. Jung, and F. Leboeuf. Enhancement of dissolution rate of poorly-soluble active ingredients by supercritical fluid processes Part I: Micronization of neat particles. Int. J. Pharm. 288(1):3–10 (2005).
A. Nokhodchi, N. Bolourtchian, and R. Dinarvand. Dissolution and mechanical behaviors of recrystallized carbamazepine from alcohol solution in the presence of additives. J. Cryst. Growth. 274(3–4):573–584 (2005).
L. E. O’Brien, P. Timmins, A. C. Williams, and P. York. Use of in situ FT-Raman spectroscopy to study the kinetics of the transformation of carbamazepine polymorphs. J. Pharm. Biomed. Anal. 36(2):335–340 (2004).
C. McGregor, M. H. Saunders, G. Buckton, and R. D. Saklatvala. The use of high-speed differential scanning calorimetry (Hyper-DSCä) to study the thermal properties of carbamazepine polymorphs. Thermochim. Acta. 417(2):231–237 (2004).
A. L. Grzesiak, M. Lang, K. Kim, and A. J. Matzger. Comparison of the four anhydrous polymorphs of carbamazepine and the crystal structure of form I. J. Pharm. Sci. 92(11):2260–2271 (2003).
B. Subramaniam, R. A. Rajewski, and K. Snavely. Pharmaceutical processing with supercritical carbon dioxide. J. Pharm. Sci. 86(8):885–890 (1997).
R. B. Gupta. Supercritical fluid technology for particle engineering. In U. B. Kompella, and R. B. Gupta (eds.), Nanoparticle technology for drug delivery, Vol. 159, Taylor and Francis, New York, 2006, pp. 53–84.
H. Ohde, J. M. Rodruguez, X. R. Ye, and C. M. Wai. Synthesizing silver halide nanoparticles in supercritical carbon dioxide utilizing a water-in-CO2 microemulsion. Chem. Comm. 18:2353–2354 (2000).
S. Sethia, and E. Squillante. Solid dispersion of carbamazepine in PVP K30 by conventional solvent evaporation and supercritical methods. Int. J. Pharm. 272:1–10 (2004).
B. C. Hancock, and M. Parksi. What is the true solubility advantage for amorphous pharmaceuticals? Pharm. Res. 17(4):397–404 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meng, D., Falconer, J., Krauel-Goellner, K. et al. Self-built Supercritical CO2 Anti-solvent Unit Design, Construction and Operation using Carbamazepine. AAPS PharmSciTech 9, 944–952 (2008). https://doi.org/10.1208/s12249-008-9130-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-008-9130-0