Abstract—
One approach to the experimental evaluation of plant tolerance to abiotic stress-factors that can be performed under in vitro culture conditions is based on zygotic embryos used at a particular developmental stage embryo culture in vitro) as explants. The cultivation of immature embryos at-the critical stage of their relative autonomy is especially promising in this respect. Such embryos can themselves, independently of physiological factors of the maternal organism, give rise to full-fledged plants under adequate conditions in vitro and then ex vitro. This makes possible to bypass another stage of morphogenic callus formation and directly derive regenerants. This paper reviews the literature and the authors’ data dealing with the identification of the stage of relative autonomy of cereal embryogenesis in vivo. The use of the relatively autonomous embryos in the assessment of drought tolerance under selective conditions in vitro is also discussed. It is emphasized that the embryo culture in vitro, as a model system for evaluation of plant stress tolerance, is promising, because the embryo has all of the morphogenetic potentials of an adult organism. Furthermore, the morphogenetic reactions of plants in vivo and explants/regenerants in vitro are similar according to the principle of universality of plant morphogenesis in natural and experimental conditions.
Similar content being viewed by others
INTRODUCTION
Under extremely unstable environmental conditions (Global’nye izmemenia klimata…, 2009; Wang et al., 2019), the problem of stress tolerance of plants to harsh abiotic factors, especially drought, is obviously urgent. This is clearly stated in a vast amount of literature, including some recent reviews concerning cereal crops (Raveena et al., 2019; Sallam et al., 2019; Sattar et al., 2019; Pykalo et al., 2020).
Since the plants’ lifestyle is sessile, they have evolved various morphological, physiological, biochemical, and other means of strategic adaptations to biological stresses at different levels of structural organization (Wani et al., 2016; Yadav and Sharma, 2016; Sattar et al., 2019; Ghorbel et al., 2020; Plant Life…, 2020; Jogawat et al., 2021; Yadav et al., 2021). Nevertheless, methods promoting plant tolerance to abiotic stresses, along with the adaptive selection of cultivars of economically important crops, especially cereals, as a main food resource, are in intensive development.
The success in breeding for stress tolerance depends to a large extent on a correct estimation of this parameter in the newly created cultivars (Dragavtsev, 2019; Sallam et al., 2019). However, this estimation is sometimes problematic. Unlike plant tolerance to biotic stresses, which generally depends on monogenic traits, the control of complex responses of the plants to abiotic stresses is multigenic (Dubrovna, 2017; Falaknaz et al., 2019; Wang et al., 2019). The establishment of plant tolerance to abiotic stressors involves several transcription factors of the NAC, AP2/EREBP, MYB, WRKY, bZIP, ERF/DREB, and other families. Some of them participate in a general control of the development of the entire plant and its separate organs (Gupta et al., 2018; Baillo et al., 2019; Kimotho et al., 2019; Yang et al., 2021). Tolerance to abiotic stresses is also a complex trait, because the stressor affects various molecular, biochemical, and physiological events associated with the processes of plant growth and development. In general, it is difficult to monitor and regulate the nonspecific responses of plants to abiotic stresses (Inzhevatkin and Savchenko, 2016).
Various methods have been developed to assess the tolerance of genotypes of cereal crops to abiotic stresses for application under field conditions in vivo. The grain yield is a direct indicator in this regard (Grabovets and Fomenko, 2016; Demydov et al., 2021; etc.), although indirect morphological, phenotypic, and quantitative indices at different stages of plant development are also pertinent for the estimation of stress effects (Grabovets and Fomenko, 2016; Mehraban et al., 2018; Chaichi et al., 2019; Grzesiak et al., 2019; etc.). Genetic and molecular-genetic approaches (Gahlaut et al., 2016; Eid, 2018; Sonmezoglu and Terzi, 2018; Dehghani et al., 2019; Falaknaz et al., 2019; Leng and Zhao, 2019; Sakkar et al., 2019) involving mathematical apparatus (Abdolshahi et al., 2015; El-Mowafi et al., 2021) are also employed. Some physiological techniques are proposed to forecast the field tolerance of cereals, e.g., to drought. They are based on the content of the stress hormone ABA (Veselov et al., 2011), amino acids (Yadav et al., 2019), and water (Alabushev et al., 2019) in the leaves, together with the index of their stomatal conductivity (Kudoyarova et al., 2013).
All the same, there are constraints on the assessment of tolerance to abiotic stress-factors under field conditions for cereals, as well as for other plants. The final conclusion regarding the tolerance or sensitivity of a particular genotype can be drawn only after many years of laborious observations and experiments. In addition, the character and degree of the alteration caused by a tested stress-factor may vary from year to year; for example, primary and secondary types of drought may be formed (Pykalo et al., 2020).
Several laboratory methods of the physiological evaluation of the stress tolerance of cereals were proposed. Early diagnostics of the breeding material is traditionally carried out via the germination of caryopses and analysis of the seedling development in situ in solutions of osmotic agents simulating, for example, water deficiency (Parfenova et al., 2018; Seldimirova, 2019; etc.). Despite the relativity of such results, these methods enable the selection of prospective tolerant specimens. A complex assessment of the tolerance of the same cereal genotypes under both laboratory (in situ) and field conditions (in vivo) makes the results more reliable (Baimagambetova and Bulatova, 2013; Bychkova and Khkebova, 2015). More precise and sophisticated laboratory methods of the early diagnostics of the stress tolerance of cereal genotypes employ cytological and molecular markers that are detectable with light fluorescent and electron microscopy (Kononenko et al., 2020).
In recent years, approaches to laboratory diagnostics based on the biotechnological methods of culture in vitro of the cells, tissues, and organs have become more in demand for the evaluation of the stress tolerance of cereal crops (Dubrovnауа et al., 2017; Osnovy biotekhnologii …, 2017; Kruglova et al., 2018a; Pykalo et al., 2020; Perez-Clemente and Gomez-Cadenas, 2012; Maleki et al., 2019). Studies of the drought tolerance of this plant group in vitro are especially intensive (Rosseev, 2011; Tagimanova et al., 2013; Kruglova et al., 2019b; Farshadfar et al., 2012; Pykalo et al., 2018, 2019). A high effectiveness of culture in vitro methods was achieved with salinization (Khuder and Al-Taei, 2015; Alhasnawi et al., 2017; Kononenko et al., 2020), hypoxia and anoxia (Vartapetyan et al., 2014), high ionic concentrations of toxic metals (Baranova et al., 2015), and other stress factors affecting cereals.
Experimental evaluation of the stress tolerance of explants demonstrates several advantages of cultures in vitro. Large series of genotypes can be screened almost year-round under standard conditions in a relatively small working space. The reactions of a large number of explants (and later regenerants) can be monitored under a strict control with the necessary manipulations at all steps of plant development in vitro. The cultural conditions, which are adjusted correspondingly to the modeled extreme conditions in vivo, enable detailed analysis of the responses of explants and regenerants to a particular abiotiс stressor (or a complex of several stressors); this task is difficult under greenhouse or field conditions due to the high variability of these stresses. In addition, the stress agent, when added to the selective medium, makes direct contact with almost all of the cells of the explant and their genetic apparatus. It is also worth noting that in vitro selection can produce regenerants that are tolerant to several stress factors simultaneously (Zinchenko et al., 2013; Perez-Clemente and Gomez-Cadenas, 2012; Merks and Guravage, 2013; Abdelnour-Esquivel et al., 2020). In our opinion, the chief advantage of the discussed methods is based on the similarity of the morphogenetic reactions of plants in vivo and their explants/regenerants in vitro according to the principle of universality of plant morphogenesis under natural and experimental conditions (Batygina, 2014).
Indeed, the use of explants/regenerants cultivated under selective conditions, like any other method, encounters specific restrictions. Thus, the-inoculation of explants into a culture is a stress impact itself, which implies cellular adaptation to the conditions in vitro. Moreover, prolonged cultivation may be accompanied by a decrease and even loss of the morphogenetic potential of the cells. Restrictions can also result from the variability of the genome and the heterogenicity of the cultured cells due to epigenetic peculiarities of the explant. The particular conditions of the selective systems in vitro can change the morphological, biochemical, and physiological parameters of the resultant regenerants and modify the changes in genetic parameters caused by somaclonal variability (Dubrovnауа, 2017; Kruglova et al., 2018a; Pykalo and Dubrovna, 2018; Ikeuchi et al., 2019). All of the this means that the obtained regenerants require scrutiny in field trials to verify their genetic stability.
In fact, cereal regenerants tolerant to various unfavorable abiotic factors, i.e., drought, have been derived under selective conditions of culture in vitro (Rosseev et al., 2011; Nikitina et al., 2014; Shupletsova and Shchennikova, 2016; Kruglova et al., 2019b; Farshadfar et al., 2012; Pykalo et al., 2018, 2019). An increased tolerance to drought remains in the progenies of the majority of the forms created via cellular selection in vitro; this indicates a mutational nature of the tolerance (Dubrovna, 2017).
A special method used to evaluate the plant stress tolerance in culture in vitro is based on the so-called embryo culture in vitro. The this approach is obviously useful, because the embryo possesses all of the morphogenetic potencies of the adult organism (Terekhin, 1996), including the capacity to cope with stresses, although this trait is not necessarily implemented.
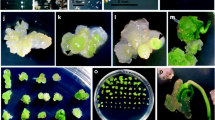
Intense studies have found two means of regenerant formation in-embryo culture in vitro—direct and indirect. In the second, regenerants emerge via the formation of morphogenic calli. The authors previously reviewed studies on the tolerance of cereals to abiotic stressors in which morphogenetic embryonic calli in vitro and the regenerants derived from them were analyzed (Kruglova et al., 2018a, 2021a; Zinatullina, 2020). The present review surveys the literature data and the authors’ data concerning the evaluation of the drought tolerance of cereals with the use of regenerants directly derived from embryo culture, without the stage of morphogenic calli.
CULTURE OF AUTONOMOUS EMBRYOS IN VITRO IN THE EVALUATION OF THE DROUGHT TOLERANCE OF CEREALS
Embryo culture in Vitro as a Biotechnological Approach
The embryo culture method is based on the cultivation of zygotic embryos of different ages in vitro (Plant Embryo Culture …, 2011). Hannig (1904, cited by Raghavan, 2003) was the first to isolate aseptically mature zygotic embryos from the plants of Raphanus and Cochlearia genera and to maintain them on sucrose-amended mineral medium until the formation of seedlings. This method was later actively improved by many workers with the use of both angiospermous and gymnospermous plants from different families. The evolution of the method of embryo culture in vitro is detailed by Raghavan (2003) and Haslam and Yeung (2011).
At present, the cultivation of embryos of different ages in vitro is used in several tasks: the determination of the physiological factors controlling embryonic morphogenesis and differentiation, developmental studies of the embryos that arose after extracorporal pollination of the ovules, analysis of premature seed germination, and the search for possible means of overcoming seed dormancy (Batygina, 2014; Raghavan, 2003; Haslam and Yeung, 2011; Hussain et al., 2012). This method is indispensable for embryo rescue of the haploid embryos of barley. These embryos are prepared with the so-called bulbosum method, which involves an interspecific crossing between the cultural forms of Hordeum vulgare L. barley and a haploproducer—the perennial, self-sterile, bulbous form of Hordeum bulbosum 2x barley (Kumari et al., 2018). The method is distinguished as a means of the production and conservation of the amphidiploid and interploid interspecific hybrids in biotechnological studies of cereals (D’yachuk et al., 2009) and as an effective procedure for regenerant preparation via the formation of morphogenic calli from immature embryos (see details in Kruglova et al., 2021b).
Meanwhile, the embryo culture in vitro techniques is one of few methods, or maybe the only one, that enables the direct derivation of cereal regenerated plants, bypassing the stage of morphogenic-callus formation. Both mature (Rostami et al., 2013; Delporte et al., 2014; etc.) and immature (Kruglova and Seldimirova, 2011; Ignatova, 2011; Goleva et al., 2014; Nikitina and Khlebova, 2014; Bychkova, 2016; Bychkova et al., 2016; Osnovy biotekhnologii…, 2017; Kruglova et al., 2019b; Zuraida et al., 2011; Zhang et al., 2015; Noga et al., 2016) cereal embryos are used for that purpose. A culture of immature embryos was found to be the most efficient.
It should be noted that biotechnological publications describing the immature embryos cultured in vitro—with the rare exception of Zuraida et al. (2011) and Kruglova et al. (2019b)—do not usually indicate the developmental stage passed by the inoculated embryos. As a rule, the time elapsed from the pollination of the ovules to the inoculation of the incubation medium with the embryos is specified. More rarely, the length of the inoculated embryos is given. Some papers mention that an “immature embryo” (without further details) was used as an explant. This situation seems to be a consequence of the absence of a conventional periodization of cereal embryogenesis. This developmental scheme should be based on a detailed histology indicating clear morphological and temporal boundaries of the embryonic stages and should be convenient for biotechnological practice, especially for mass seasonal work. Actually, we have elaborated such a periodization for spring soft wheat (Kruglova, 2012b, 2013b), which is not widely recognized as yet.
Meanwhile, the data on a particular stage of embryogenesis are essential upon the use of embryo culture in vitro if it is necessary to inoculate an immature embryo, which can directly originate the seedling and the subsequent regenerated plant. Embryos of the same genotype but different ages manifest different responses to the same cultural conditions in vitro. This was demonstrated, for example, in a detailed study of the vast collection of cultivars and hybrid lines of spring soft wheat (Kruglova, 2012a, 2012b, 2014; Kruglova et al., 2018b, 2018c). In terms of the regarded problem, it is important to characterize the exact developmental stage from which the embryo can give rise to a full-fledged plant. This significant item will be considered below on the example of cereal family.
Recognition of the Critical Stage of Relative Autonomy as a Methodical Approach to the Direct Derivation of Regenerants in the Embryo Culture In Vitro
The plant embryonic development is an integral process in which the mature embryo forms from the initial totipotent cell—a zygote—according to a certain pattern of cell divisions (Embriologia tsvetkovykh…, 2000; Batygina, 2014; Methods in…, 2008; De Vries and Weijers, 2017). Embryogenetic laws (the law of origin, the law of numbers, the law of disposition, and the law of parsimony) (Shamrov, 1997) characterize the complexity of this process.
The embryogenesis of cereals is especially complex. The dorsoventral structure of their embryos is established from the first steps of their development (from the zygote). The specificity of their organogenesis brings about a unique structure comprising such highly-differentiated organs as the epiblast, scutellum, and ligula. These peculiarities allow the definition of a special type of cereal embryogenesis termed Graminad (Batygina, 2014, and earlier). The validity of this assignment was supported by studies of different cereal species (Kruglova et al., 2019a).
The zygotic embryo is known to pass through several discrete, interrelated stages in its morphogenesis. The stages differ from each other in their morphological processes, functional load, duration, and significance for subsequent development. Despite the diversity of the concurring processes, the goal of each developmental stage is to implement the morphogenetic potential of the embryo and to execute of the ontogenetic developmental program of the whole individual. The embryo demonstrates the properties of a dynamic system with a pulse mode of functioning of its elements (Batygina, 2014).
Certain stages of embryogenesis are considered to be critical.
The literature offers different, definitive criteria for the critical stages of plant embryogenesis. T.B. Batygina and V.E. Vasilyeva (1983) proposed a systemic approach to define these stages. It is based on embryonic differentiation with allowance for morphogenetic and morphophysiological correlations in the development of embryonic structures. Here, the critical stages are put forward as the time intervals for changes in the structural–functional characteristics of embryonic development and the adjacent tissues of a fruit and seed. I.I. Shamrov et al. (Shamrov, 2008; Shamrov and Anisimova, 2003; еtc.) dealt with the periodization of the development of the ovules and seeds of flowering plants and considered the morphogenetic and morphophysiological correlations. The authors gave the term critical stages to the rather short time periods related to the structural–functional rearrangements in the ovule and seed. The particular stages are named after the main embryonic structures that arose in this period. In the opinion of Wareing and Phillips (1981), at the critical stages of plant embryogenesis, the embryonic development is reprogrammed to alternative pathways, and certain parts of the embryo are determined for their forthcoming differentiation. This viewpoint conforms in many respects to the concept of an embryonic induction in animals: interaction of the embryonic layers has a form-building effect via the target tissue, which becomes determined for a particular developmental type. Afterwards, the determined state of the tissue is implemented during the subsequent differentiation (Gilbert, 2018).
Batygina and Vasilyeva (Batygina, 1987, 2014; Batygina and Vasilyeva, 1987a, 1987b, 1988; Vasilyeva and Batygina, 1997, 2006) carried out a series of studies analyzing the critical stage of the relative autonomy of the embryos of flowering plants as a manifestation of the autonomization of individual ontogenesis. In complete autonomy, all structural and functional links of the embryo with the maternal organism (fruit or seed) are ceased; the embryo, independent of the surrounding tissues, is capable of self-regulation and seedling formation. In relative autonomy, the embryo, unlike a completely autonomous one, becomes independent of the physiological and biochemical maternal factors (primarily hormones). In other words, a strict developmental determination of the embryo, as a novel organism, is fixed at the stage of relative autonomy. Since the establishment of autonomy is a long and complex process, the concept of the “degree of autonomy” is proposed as a quantitative and temporal measure of the embryonic dependence on the maternal organism.
Issues of the relative autonomy of cereal embryos were considered in a the study (Kruglova et al., 2020) in comparison with different families of flowering plants. The authors speculate that, since this critical stage of embryogenesis is determined to a large extent by structural traits of the cereal embryos, its identification can favor the solution of some basic problems of theoretical biology and the evolution of higher plants. Moreover, data on the relative autonomy of the embryo may be of practical importance for the identification and assessment of the immature status of the cereal embryos. This use of material would be optimal in embryo culture in vitro in order to direct the derivation of the regenerated plants under conditions in vitro and, thereafter, ex vitro.
Two criteria are proposed for the identification of the stage of relative autonomy of the plant embryos. The first is the ability of the embryos to accomplish embryogenesis and to form normal seedlings in vitro on hormone-free medium (Batygina and Vasilyeva, 1987a). The second, more rigorous one is the ability of the embryos not only to complete embryogenesis and to produce seedlings on a hormone-free medium but also to originate full-grown, fertile regenerants ex vitro with seeds that can be assessed for their germinability under laboratory conditions (Kruglova, 2013a).
The stage of relative autonomy of developing cereal embryos was thoroughly determined for barley and wheat. According to the first criterion, immature embryos of the cultural forms of barley (Hordeum vulgare L.) become relatively autonomic on days 10–14, depending on the genotype (Ignatova, 2011). By the second criterion, the relative autonomy of the embryos was found to occur 12–15 days after pollination, depending on the genotype; this analysis was carried out on the vast collection of cultivars and hybridous combinations of spring soft wheat Triticum aestivum L. (Kruglova, 2014; Kruglova et al., 2018b, 2018c). The inconsistence between barley and wheat with respect to the times of the establishment of relative autonomy may be attributed to the difference between climatic conditions in their cultivation zones and weather conditions during vegetation. In addition, the taxon-dependent specificity of this critical stage may play some role, because particular species differ in their morphogenetic, physiological, and biochemical embryonic traits at this stage. The examined zygotic/somatic embryos of dicotyledonous plants also demonstrate a similar taxon-specific dispersion in the appearance of the stage of relative autonomy (Vasilyeva and Batygina, 1997).
There are only a few published studies specially focused on the recognition of the histological status of the cereal embryos at the critical stage of relative autonomy. In this regard, Vasilyeva and Batygina (1997) point to the stage of differentiation of the scutellum and the root apex. In the opinion of Kruglova et al. (2020), the critical stage of relative autonomy of the cereal embryos is the stage of the differentiation of all organs pertinent to the embryo: the scutellum (cotyledon), the ligula, the plumule (which consists of the shoot apex and the first leaf), the coleoptile, the epiblast, the coleorhiza, the mesocotyle, and the embryonic root. This histological status was found in wheat embryos on days 12–15 postpollination (Kruglova et al., 2018b, 2018c). This again confirms the reason for the use of the second, more rigorous criterion of the embryonic relative autonomy, at least in the tested wheat genotypes.
Seldimirova et al. (2017b) reported the results of ultrastructural observations of cells of wheat immature embryos on days 15–17 postpollination. This age partially corresponds to the stage of relative autonomy. The scutellum cells of the embryo contain lipid droplets and large amyloplasts that accumulate starch. There are well-developed mitochondria and plastids as single chloroplasts in the cells of the shoot apex. This is experimental evidence of the synthesis of constitutive compounds in these cells and their high metabolic activity.
The readiness of embryos to develop independently from the stage of relative autonomy is also characterized by physiological signs, mainly the availability of their endogenous hormones. The independence of the relatively autonomic embryos of exogenous hormones is confirmed by their successful cultivation on a hormone-free medium in vitro.
It is known that the autologous hormonal system of cereals, like that of other plants, is progressively formed in the course of embryogenesis and actively participates in the regulation of the embryonic growth and development (Kruglova et al., 2019a). It is feasible that the key hormones of morphogenesis—auxins, which govern cellular growth, and cytokinins, which govern cell divisions (Medvedev and Sharova, 2011)—are present in the cereal embryos at the stage of relative autonomy (12–15 days postpollination), although these facts have not been specially explored. All the same, this suggestion agrees with the dynamics of hormones as related to the dry mass accumulation in the developing wheat caryopses. Actually, on day 12 after pollination, the contents of IAA and cytokinins show a sharply increase that corresponds to the rapid enlargement of the caryopsis (Hess et al., 2002). In addition, the cytokinin level is maximum in the barley caryopsis after 14 days postpollination (Seldimirova et al., 2018). In addition, the deep staining for IAA occurs in the cells of the apical part of the embryo and the cells of the developing organs of the wheat embryo at the phase of organogenesis, including the stage of differentiation of the shoot apex and organs (Seldimirova et al., 2017a).
In general, the cereal embryo, after reaching the critical stage of relative autonomy, possesses morphological, histological, and physiological (hormonal) statuses that allowing its autonomic development independent of maternal organism. In practical biotechnological studies of the embryo culture in vitro, it is the relative autonomy as a developmental stage of inoculating immature embryos that gives rise to normal seedlings and later plants under both in vitro and ex vitro conditions.
Relatively Autonomic Cereal Embryos in Evaluation of Drought Tolerance under Selective Conditions In Vitro
Physiological drought is an adverse combination of meteorological conditions under which plants undergo long-term water deficiency in the air and soil (Kuznetsov and Dmitrieva, 2011). This is one of the most common abiotic stress-factors that occur in both arid and semiarid areas. This causes devastating losses to agricultural production and endangers food security (Plant Life …, 2020). It has been suggested that a shortage of soil water is much more injurious to plant production than the sum of all other stress factors (Dubrovnауа, 2017; Pykalo et al., 2020). In general, it is impossible to overestimate the significance of research on the various consequences of drought and the formation of adaptive tolerance of agricultural crops, including cereals, to this stress factor.
The cultivation of relatively autonomic embryos on selective nutrient medium simulating water deficiency is a promising biotechnological approach that enable the recognition of drought-tolerant plants among those already available, as well as newly created cultivars and hybrid combinations. In fact, the degree of structural and functional differentiation of this embryo is determined not only by its intrinsic genotype (which determines the type of embryogenesis and the specificity of development) but also the genotype of the entire maternal organism (this determines the conditions inside the developing seed, which are indirectly related with the whole maternal organism) and the capacity of the individual to withstand drought (Vasilyeva and Batygina, 1997).
To artificially simulate drought, osmotic agents are added to the cultivation medium for cereals and other plant families in vitro. For this purpose, polyethylene glycol with a molecular mass of 6000 (PEG 6000) or 10 000 (PEG 10 000), mannitol, sorbitol, sucrose, or sodium chloride are used (Nikitina et al., 2013, 2014; Bychkova and Khlebova, 2015; Seldimirova, 2019; Pykalo et al., 2018, 2019; etc.). PEG 6000 is especially popular. Because of its high molecular mass, it does not permeate into the cells but collapses the cell walls and shrinks the protoplast, which well mimics the water balance of osmotically-stressed cells (Freitas et al., 2020). Meanwhile, the mannitol-containing, selective, in vitro system is more efficient, since it completely eliminates sensitive cells and favors better viability of the surviving plant regenerants (Dubrovna, 2017; Pykalo et al., 2020). In general, the choice of particular drought-simulating agents is determined by the purpose and conditions of the given experiment.
Therefore, the examination of the immature, relatively autonomic cereal embryos in cultures in vitro on selective media containing drought simulators appears to be promising. Nevertheless, these studies are rather poorly represented in the literature. This might be a consequence of the inadequate development of this biotechnology (Dubrovnауа et al., 2014, cited by Pykalo et al., 2020), and this opinion is difficult to doubt.
Nikitina et al. (2014) compared cultivars of spring soft and hard wheat that differ in their drought tolerance under field conditions. In vitro cultured immature embryos were tested, and the effectiveness of different techniques of cell selection (rigorous, soft, and mixed) for tolerance to NaCl-induced osmotic stress was assessed. The authors did not use the term “embryo culture in vitro.” Comparison of the level of the field tolerance of a cultivar and the reaction to the sampling technique showed that success in regeneration upon a soft selection is determined to a greater extent by the regeneration potential of the genotype in vitro than its drought tolerance. Rigorous selection with a sublethal dose of sodium chloride, as a selective agent to pick out the stress-tolerant regenerants of soft wheat, is possible only for genotypes with high regeneration potential. The results of the study agree with the recognized opinion that regeneration in the culture in vitro is primarily determined by the traits of the genotype of the donor plant (Ikeuchi et al., 2019; Sugimoto et al., 2019). The work (Nikitina et al., 2014) is also valuable in that it compared the stress tolerance of several cultivars of hard wheat, which is rarely studied in vitro in this respect.
N.N. Kruglova (Kruglova, 2012c, 2013a, 2014; Kruglova et al., 2018b, 2018c, 2019b) reported thorough studies of relatively autonomic immature embryos. The material was taken from a vast collection of parental cultivars and hybrid combinations from direct and reciprocal crosses of spring soft wheat under selective conditions of cultures in vitro. A rigorous criterion of drought tolerance of the immature embryos was used. It was based on their ability to give rise to seedlings that develop to the phenological stage of tillering. The development occurs under in vitro conditions simulating water deficiency via the addition of PEG 6000 at sublethal concentration 12–14%. In addition, the seedling development until the phenological stage of full grain ripeness in a soil culture ex vitro, as well as the laboratory germinability of the caryopses in situ, were estimated. The experiments identified wheat genotypes displaying both the capacity of immature embryos to form seedlings under drought-mimicking conditions and a high laboratory germinability of the caryopses. It is interesting that there was no correlation between the parental cultivars and their hybrids in the drought tolerance/sensitivity of the immature embryos. In some cases, crossings of drought-sensitive cultivars yielded hybrids with immature embryos that exhibited sufficiently high tolerance under selective conditions in vitro. Presumably, this responsivity of the hybrid embryos is explained by the complex interaction of the parental genotypes; this trait may be controlled by multiple genes, which is typical of quantitative traits (Dragavtsev, 2019). The drought-tolerant genotypes were identified after many years of exploration and were recommended as initial forms for breeding programs to create zoned cultivars that are promising towards drought tolerance. The authors emphasize the potential value of the identification and selection in vitro of drought-tolerant immature embryos at the stage of relative autonomy. This approach enables express diagnostics of drought tolerance of each newly bred hybrid combination and subsequent wheat cultivar. In this case, the screening goes faster, because the hybrid combination is inspected for drought tolerance at the earliest ontogenic stage (the embryo) rather than routine laboratory analysis of the mature grain or field trials. This conclusion is pertinent to selection studies on any cereal.
CONCLUSIONS
The stress tolerance of plants is a complex process that is determined by quantitative traits of multigenic inheritance. For this reason, the development of different model systems to study this biological phenomenon is actually promising. These systems include in vitro cultures of the cells, tissues, and organs. This approach makes it possible to gain insight into the general relationships and characteristics of plant responses to abiotic stressors and can point to means to control plant adaptations to stress conditions.
The division of embryo culture in vitro as the cultivation of immature zygotic embryos at the stage of relative autonomy can be an experimental approach to the study of the cellular and tissue mechanisms of plant responses to various stress factors. An argument in favor of the use of this technique is the fact that the embryo possesses all of the morphogenetic potencies of the adult organism and that the responses of plants in vivo and explants in vitro (here, autonomic immature embryos) are similar because of the universality of the morphogenetic pathways.
Although embryo cultures in vitro have been studied for more than 110 years, there is a rather limited number of works using this method for the stress-assessment of plants, in contrast to the use of embryonic callus cultures in vitro for similar purposes. However, the biotechnological use of immature embryos at the stage of relative autonomy can favor the straightforward derivation of stress-tolerant regenerants. The advantage of this approach consists of the inoculation of immature embryos that are already independent of physiological factors of the maternal organism. The structural and functional readiness of relatively autonomic embryos upon further normal development and the formation of full-fledged regenerants will save working time. In the case of wheat, the time economy is at least 15 days in comparison with the technique of mature embryos. Furthermore, the-culture in vitro of immature autonomic embryos makes it possible to avoid the costly and laborious steps of the formation of morphogenic calli and the subsequent initiation of plant regeneration via the alternation of nutrient media. This is especially significant if it is necessary to minimize the somaclonal variability, which is usually associated with callus cultures in vitro.
In general, cultures in vitro of immature embryos at the stage of relative autonomy can be regarded as a convenient and reliable biotechnological tool for the express-testing of genotypes of economically valuable plants against various stressors and as a measure to create the stress-tolerant zoned cultivars as a final purpose of breeding.
Further development of biotechnologies involving the in vitro cultivation of relatively autonomic embryos can be highly successful with the use of molecular-genetic approaches and data on functional genomics and transgenesis. This will widen the possibilities of classical breeding, which increases the stress tolerance of cereals and other economically valuable plants.
REFERENCES
Abdelnour-Esquivel, A., Perez, J., Rojas, M., et al., Use of gamma radiation to induce mutations in rice (Oryza sativa L.) and the selection of lines with tolerance to salinity and drought, In Vitro Cell. Dev. Biol.: Plant, 2020, vol. 56, pp. 88–97. https://doi.org/10.1007/s11627-019-10015-5
Abdolshahi, R., Nazari, M., Safarian, A., et al., Integrated selection criteria for drought tolerance in wheat (Triticum aestivum L.) breeding programs using discriminant analysis, Field Crops Res., 2015, vol. 174, pp. 20–29.
Alabushev, A.V., Ionova, E.V., Likhovidova, V.A., et al., Estimation of drought tolerance of winter soft wheat in conditions of modeled drought, Zemledelie, 2019, no. 7, pp. 35–37.
Alhasnawi, A.N., Zain, Ch.R., Kadhimi, A.A., et al., Accumulation of antioxidants in rice callus (Oryza sativa L.) induced by β-glucan and salt stress, Austral. J. Crop Sci., 2017, vol. 11, pp. 118–125.
Baillo, E.H., Kimotho, R.N., Zhang, Z., and Xu, P., Transcription factors associated with abiotic and biotic stress tolerance and their potential for crops improvement, Genes, 2019, vol. 10, no. 10, р. 771. https://doi.org/10.3390/genes10100771
Baimagambetova, K. and Bulatova, K., Gradual assessment of varieties and lines of spring wheat for drought tolerance, Sel. Semenarstvo, 2013, vol. 19, no. 2, pp. 27–34.
Baranova, E.N., Chaban, I.A., Kononenko, N.V., et al., Morphofunctional characteristics of barley calli tolerant to the toxic effect of aluminum, Biol. Membr., 2015, vol. 32, no. 3, pp. 1–13.
Batygina, T.B., Khlebnoe zerno (Bread Grain), Leningrad: Nauka, 1987.
Batygina, T.B., Biologiya razvitiya rastenii (Plant Developmental Biology), St. Petersburg: DEAN, 2014.
Batygina, T.B. and Vasil’eva, V.E., System approach to differentiation of a embryo of angiosperms, Ontogenez, 1983, vol. 14, no. 3, pp. 304–311.
Batygina, T.B. and Vasil’eva, V.E., Applied aspects of embryology. Embryo autonomy and embryo culture of flowering plants, Bot. Zh., 1987a, vol. 72, no. 2, pp. 155–161.
Batygina, T.B. and Vasilyeva, V.E., Some aspects of embryo-culture (autonomy of embryo) of flowering plants, Phytomorphology, 1987b, vol. 37, pp. 283–288.
Batygina, T.B. and Vasilyeva, V.E., Some aspects of autonomy of embryo in flowering plants, Phytomorphology, 1988, vol. 38, pp. 293–297.
Bychkova, O.V., Evaluation of the effectiveness of morphogenesis and regeneration of spring durum wheat in vitro, Acta Biol. Sib., 2016, no. 2 (1), pp. 139–149.
Bychkova, O.V. and Khlebova, L.P., Physiological assessment of drought resistance of spring durum wheat, Acta Biol. Sib., 2015, vol. 1, nos. 1–2, pp. 107–116.
Bychkova, O.V., Ereshchenko, D.V., and Rozova, M.A., Comparative assessment of the use of mature and immature embryos of spring durum wheat in culture, Acta Biol. Sib., 2016, vol. 2, no. 2, pp. 76–80.
Chaichi, M., Sanjarian, F., Razavi, K., and Gonzalez-Hernandez, J.L., Phenotypic diversity among Iranian bread wheat landraces, as a screening tool for drought tolerance, Acta Physiol. Plant., 2019, vol. 41, p. 90.
De Vries, S.C. and Weijers, D., Plant embryogenesis, Curr. Biol., 2017, vol. 27, pp. 870–873.
Dehghani, I., Mostajeran, A., Esmaeili, A., and Ghannadian, M., The role of DREB2 gene in drought tolerance of common wheat (Triticum aestivum L.) associated with Azospirillum brasilense, Appl. Ecol. Environ. Res., 2019, vol. 17, pp. 4883–4902.
Delporte, F., Pretova, A., Du Jardin, P., et al., Morpho-histology and genotype dependence of in vitro morphogenesis in mature embryo cultures of wheat, Protoplasma, 2014, vol. 251, pp. 1455–1470.
Demydov, O., Khomenko, S., Fedorenko, M., et al., Stability and plasticity of collection samples of durum spring wheat in the forest-steppe conditions of Ukraine, Am. J. Agric. For., 2021, vol. 9, pp. 83–88.
Dragavtsev, V.A., Increase of crop yields based on the theory of ecological and genetic organization of quantitative traits, Byull. Gl. Nauchn. Bot. Sada, 2019, no. 132, pp. 17–28.
Dubrovnaya, O.V., Wheat breeding in vitro for resistance to abiotic stress factors, Fiziol. Rast. Genet., 2017, vol. 49, no. 4, pp. 279–292.
D’yachuk, T.I., Khomyakova, O.V., Stolyarova, S.V., et al., Cellular biotechnologies in creation of initial material for triticale breeding, Agrar. Vestn. Yugo-Vost., 2009, no. 2, pp. 9–10.
Eid, M., Validation of SSR molecular markers linked to drought tolerant in some wheat cultivars, J. Plant Breed. Genet., 2018, vol. 6, pp. 95–109.
El-Mowafi, H.F., Al Kahtani, M.D.F., Abdallah, R.M., et al., Combining ability and gene action for yield characteristics in novel aromatic cytoplasmic male sterile hybrid rice under water-stress conditions, Agriculture, 2021, vol. 11, no. 3, р. 226. https://doi.org/10.3390/agriculture11030226
Embryology of Flowering Plants: Terminology and Concepts, Vol. 3: Reproductive Systems, Batygina, T.B., Ed., Boca Raton, Fl: CRC Press, 2009.
Falaknaz, M., Aalami, A., Mehrabi, A.A., et al., Assessing Aegilops tauschii genotypes to drought stress using tolerance indices, Cereal Res., 2019, vol. 8, pp. 483–494.
Farshadfar, E., Jamshidi, B., Cheghamirza, K., et al., Evaluation of drought tolerance in bread wheat (Triticum aestivum L.) using in vivo and in vitro techniques, Ann. Biol. Res., 2012, vol. 3, pp. 465–476.
Freitas, W.C., Medina, P.F., Giomoto, G.S., and Almeida, J.A.S., PEG 6000 and sucrose in the control of the direct somatic embryogenesis capacity in Coffea arabica L., J. Global Biosci., 2020, vol. 9, pp. 7364–7376.
Gahlaut, V., Jaiswal, V., Kumar, A., et al., Transcription factors involved in drought tolerance and their possible role in developing drought tolerant cultivars with emphasis on wheat (Triticum aestivum L.), Theor. Appl. Genet., 2016, vol. 129, pp. 2019–2042.
Ghorbel, M., Saibi, W., and Brini, F., Abiotic stress signaling in Brassicaceae plants, J. Soil Plant Biol., 2020, vol. 1, pp. 138–150.
Gilbert, S.F., Developmental Biology, Mayers, R., Ed., 2018, 11th ed. https://www.pdfdrive.com/developmental-biology-e188565455.html.
Global’nye izmeneniya klimata i prognoz riskov v sel’skom khozyaistve Rossii (Global Climate Changes and Forecast of Risks for Russian Agriculture), Ivanov, A.L. and Kiryushin, V.I., Eds., Moscow: Rossel’khozakademiya, 2009.
Goleva, G.G., Batluk, Yu.A., Vashchenko, T.G., et al., Obtaining plants-regenerants of winter soft wheat (Triticum aestivum L.) in vitro, Vestn. Voronezh. Gos. Agrar. Univ., S-kh. Nauki, 2014, no. 3 (42), pp. 17–22.
Grabovets, A.I. and Fomenko, M.A., Improvement of wheat breeding procedure in conditions of insufficient moisture, Zernovye Krupyanye Kul’t., 2016, no. 2 (18), pp. 48–53.
Grzesiak, M.T., Hordynska, N., Maksymowicz, A., et al., Variation among spring wheat (Triticum aestivum L.) genotypes in response to the drought stress. II—Root system structure, Plants, 2019, vol. 8, no. 12, р. 584. https://doi.org/10.3390/plants8120584
Gupta, S., Mishra, V.K., Kumari, S., et al., Deciphering genome-wide WRKY gene family of Triticum aestivum L. and their functional role in response to abiotic stress, Genes Genomics, 2019, vol. 41, pp. 79–94. https://doi.org/10.1007/s13258-018-0742-9
Ignatova, S.A., Kletochnye tekhnologii v rastenievodstve, genetike i selektsii vozdelyvaemykh rastenii: zadachi, vozmozhnosti, razrabotki sistem in vitro (Cell Technologies in Plant Cultivation, Genetics, and Selection of Crop Plants: Objectives, Opportunities, and Cultivation in Vitro), Odessa: Astroprint, 2011.
Ikeuchi, M., Favero, D.S., Sakamoto, Y., et al., Molecular mechanisms of plant regeneration, Ann. Rev. Plant B-iol., 2019, vol. 70, pp. 377–406.
Inzhevatkin, E.V. and Savchenko, A.A., The nonspecific metabolic reaction of cells to extreme exposures, Biol. Bull. (Moscow), 2016, vol. 43, no. 1, pp. 2–11.
Jogawat, A., Yadav, B., Lakra, N., et al., Crosstalk between phytohormones and secondary metabolites in the drought stress tolerance of crop plants: a review, Phy-siol. Plant., 2021, vol. 172, no. 2, pp. 1106–1132. https://doi.org/10.1111/ppl.13328
Haslam, T.M. and Yeung, E.C., Zygotic embryo culture: an overview, in Plant Embryo Culture: Methods and Protocols, Methods Mol. Biol., vol. 710, Thorpe, T.A. and Yeung, E.C., Eds., Dordrecht: Springer-Verlag, 2011, ch. 1, pp. 3–16.
Hess, J.R., Carman, J.G., and Banowetz, G.M., Hormones in wheat kernels during embryony, Plant Physio-l., 2002, vol. 159, pp. 379–386.
Hussain, A., Qarshi, I.A., Nazir, H., et al., Plant tissue culture: current status and opportunities, in Recent Advances in Plant in Vitro Culture, London: InTechOpen, 2012, pp. 1–21. https://doi.org/10.5772/50568
Khuder, H.H. and AL-Taei, Yu.I.H., Effect of salt stress on some growth indicators and cellular components of wheat (Triticum aestivum L.) callus, Int. J. Appl. Agric. Sci., 2015, vol. 1, pp. 91–94.
Kimotho, R.N., Baillo, E.H., and Zhang, Z., Transcription factors involved in abiotic stress responses in maize (Zea mays L.) and their roles in enhanced productivity in the post genomics era, Peer J., 2019, vol. 7, p. e7211. https://doi.org/10.7717/peerj.7211
Kononenko, N., Baranova, E., Dilovarova, T., et al., Oxidative damage to various root and shoot tissues of durum and soft wheat seedlings during salinity, Agriculture, 2020, vol. 10, no. 3, p. 55. https://doi.org/10.3390/agriculture10030055
Kruglova, N.N., Optimization of biotechnological production of wheat plants in vitro, Izv. Ufimsk. Nauchn. Tsentra, Ross. Akad. Nauk, 2012a, no. 3, pp. 57–61.
Kruglova, N.N., Periodization of wheat germ development as a methodological aspect for biotechnological developments, Izv. Ufimsk. Nauchn. Tsentra, Ross. Akad. Nauk, 2012b, no. 2, pp. 21–24.
Kruglova, N.N., Assessment of the collection of spring soft wheat genotypes by the resistance of autonomous embryos on selective media simulating a drought in vitro, Izv. Samar. Nauchn. Tsentra, Ross. Akad. Nauk, 2012c, vol. 14, no. 1 (9), pp. 2243–2245.
Kruglova, N.N., Identification of the critical stage of wheat germ autonomy in vitro, Izv. Ufimsk. Nauchn. Tsentra, Ross. Akad. Nauk, 2013a, no. 1, pp. 42–45.
Kruglova, N.N., Periodization of wheat embryo structure on the base of anatomy and morphology criteria, Mod. Phytomorphol., 2013b, vol. 4, pp. 181–183.
Kruglova, N.N., Identification of autonomy of a wheat germ as a stage in the development of express diagnostic of biotechnological obtaining of drought-resistant plants, Perm. Agrar. Vestn., 2014, no. 1 (5), pp. 38–43.
Kruglova, N.N. and Sel’dimirova, O.A., Regeneratsiya pshenitsy in vitro i ex vitro: tsitologicheskie aspekty (Regeneration of Wheat Plants in Vitro and ex Vitro: Cytogenetic Aspects), Ufa: Gilem, 2011.
Kruglova, N.N., Seldimirova, O.A., and Zinatulina, A.E., In vitro callus as a model system for the study of plant stress-resistance to abiotic factors (on the example of cereals), Biol. Bull. Rev., 2018a, vol. 8, pp. 518–526.
Kruglova, N.N., Sel’dimirova, O.A., Zinatullina, A.E., and Veselov, D.C., The critical stage of wheat germ autonomy in planta, Biomika, 2018b, vol. 10, no. 1, pp. 1–6.
Kruglova, N.N., Sel’dimirova, O.A., Zinatullina, A.E., and Nikonov, V.I., Identification of the relative autonomy in planta of zygotic embryos of spring soft wheat for optimization of biotechnological studies, Izv. Ufimsk. Nauchn. Tsentra Ross. Akad. Nauk, 2018c, no. 3, pp. 28–33.
Kruglova, N.N., Sel’dimirova, O.A., Zinatullina, A.E., and Nikonov, V.I., Identification of drought-resistant wheat genotypes in vitro culture of immature embryos, Vestn. Bashkir. Gos. Agrar. Univ., 2019, vol. 52, no. 4, pp. 37–41.
Kruglova, N.N., Seldimirova, O.A., and Zinatulina, A.E., Structural features and hormonal regulation of the zygotic embryogenesis in cereals, Biol. Bull. Rev., 2020a, vol. 10, pp. 115–126.
Kruglova, N.N., Titova, G.E., Seldimirova, O.A., et al., Embryo of flowering plants at the critical stage of embryogenesis relative autonomy (by example of cereals), Russ. J. Dev. Biol., 2020b, vol. 51, no. 1, pp. 1–15.
Kruglova, N.N., Sel’dimirova, O.A., and Zinatulina, A.E., Callus cultures in vitro in the experimental evaluation of drought resistance of cereals, Tavricheskii Vestn. Agrar. Nauki, 2021a, no. 1 (25), pp. 124–139.
Kruglova, N.N., Titova, G.E., Seldimirova, O.A., and Zinatulina, A.E., Cytophysiological features of the cereal-based experimental system “embryo in vivo–callus in vitro,” Russ. J. Dev. Biol., 2021, vol. 52, no. 4, pp. 199–214.
Kudoyarova, G.R., Kholodova, V.P., and Veselov, D.S., Current state of the problem of water relations in plants under water deficit, Russ. J. Plant Physiol., 2013, vol. 60, no. 2, pp. 165–175.
Kumari, P., Thaneshwari, and Rahul, Embryo rescue in horticultural crops, Int. J. Curr. Microbiol. Appl. Sci., 2018, vol. 7, pp. 3350–3358.
Kuznetsov, V.V. and Dmitrieva, G.A., Fiziologiya rastenii (Plant Physiology), Moscow: Abris, 2011.
Leng, P. and Zhao, J., Transcription factors as molecular switches to regulate drought adaptation in maize, Theor. Appl. Genet., 2020, vol. 133, no. 5, pp. 1455–1465. https://doi.org/10.1007/s00122-019-03494-y
Maleki, M., Ghorbanpour, M., Nikabadi, S., et al., In vitro screening of crop plants for abiotic stress tolerance, in Recent Approaches in Omics for Plant Resilience to Climate Change, Wani, S., Ed., Cham: Springer-Verlag, 2019, pp. 75–91.
Medvedev, S.S. and Sharova, E.I., Biologiya razvitiya rastenii, Tom 1. Nachala biologii razvitiya rastenii. Fitogormony (Developmental Biology of the Plants, Vol. 1: Fundamentals of Developmental Biology of the Plants. Phytohormones), St. Petersburg: S.-Peterb. Gos. Univ., 2011.
Mehraban, A., Tobe, A., Gholipouri, A., et al., Evaluation of drought tolerance indices and yield stability of wheat cultivars to drought stress in different growth stage, World J. Environ. Biosci., 2018, vol. 7, pp. 8–14.
Merks, R.M.H. and Guravage, M.A., Building simulation models of developing plant organs, in Plant Organogenesis: Methods and Protocols, Methods Mol. Biol., vol. 959, De Smet, I., Ed., New York: Springer-Verlag, 2013, pp. 333–352.
Nikitina, E.D. and Khlebova, L.P., Influence of temperature and light on direct germination of immature embryos of Triticum aestivum L. in culture in vitro, Izv. Altai. Gos. Univ., Biol. Nauki, 2014, vol. 2, no. 3, pp. 46–50.
Nikitina, E.D., Khlebova, L.P., Sokolova, G.G., and Ereshchenko, O.V., Creation of stress-resistant spring soft wheat using cell selection in vitro, Izv. Altai. Gos. Univ., Biol. Nauki, 2013, no. 3, pp. 95–98.
Nikitina, E.D., Khlebova, L.P., and Ereshchenko, O.V., Cell breeding of spring wheat for resistance to abiotic stresses, Izv. Altai. Gos. Univ., Biol. Nauki, 2014, vol. 2, no. 3, pp. 50–54.
Noga, A., Skrzypek, E., Warchoł, M., et al., Conversion of oat (Avena sativa L.) haploid embryos into plants in relation to embryo developmental stage and regeneration media, In Vitro Cell. Dev. Biol.: Plant, 2016, vol. 52, pp. 590–597.
Osnovy biotekhnologii rastenii (Fundamentals of Plant Biotechnology), Kuluev, B.R., Kruglova, N.N., Zaripova, A.A., and Farkhutdinov, R.G., Eds., Ufa: Bashkir. Gos. Univ., 2017.
Parfenova, E.S., Shamova, M.G., Nabatova, N.A., et al., Assessment of the relative drought resistance of winter rye varieties by the germination on a sucrose solution, Mezhdunar. Zh. Prikl. Fundam. Issled., 2018, no. 11-2, pp. 347–351.
Pérez-Clemente, R.M. and Gómez-Cadenas, A., In vitro tissue culture, a tool for the study and breeding of plants subjected to abiotic stress conditions, in Recent Advances in Plant In Vitro Culture, Leva, A. and Rinaldi, L., Eds., London: InTechOpen, 2012. https://doi.org/10.5772/50671
Pikalo, S., Demidov, O., Yurchenko, T.I., et al., Assessment methods of drought resistance of selected wheat materials, Visn. L’viv. Univ., Ser. Biol., 2020, no. 82, pp. 63–79.
Plant Embryo Culture: Methods and Protocols, Thorpe, T.A. and Yeung, E.C., Eds., Dordrecht: Springer-Verlag, 2011.
Plant Embryogenesis, Methods Mol. Biol., vol. 427, Suarez, M.E. and Bozhkov, P.V., Eds., Totowa, N.J.: Humana, 2008.
Plant Life under Changing Environment: Responses and Management, Tripathi, D.K. et al., Eds., Amsterdam: Elsevier, 2020.
Pykalo, S.V. and Dubrovna, O.V., Variability of the triticale genome in vitro, Cytol. Genet., 2018, vol. 52, pp. 385–393.
Pykalo, S.V., Demydov, O.A., Prokopik, N.I., et al., In vitro screening of the spring wheat F2 hybrids for water deficit resistance, ScienceRise: Biol. Sci., 2018, vol. 3, no. 12, pp. 12–18.
Pykalo, S., Demydov, O., Yurchenko, T., et al., Comparative assessment of methods for evaluation of drought tolerance in winter bread wheat varieties, ScienceRise: Biol. Sci., 2019, vol. 4, no. 19, рр. 17–21. https://doi.org/10.15587/2519-8025.2019.186813
Raghavan, V., One hundred years of zygotic embryo culture investigations, In Vitro Cell. Dev. Biol.: Plant, 2003, vol. 39, pp. 437–442.
Raveena, Bharti, R., and Chaudhary, N., Drought resistance in wheat (Triticum aestivum L.): a review, Int. J. Curr. Microbiol. Appl. Sci., 2019, vol. 8, pp. 1780–1792.
Rosseev, V.M., Belan, I.A., and Rosseeva, L.P., Testing of spring soft wheat for drought reistance in vitro, Vestn. Altai. Gos. Agrar. Univ., 2011, vol. 76, no. 2, pp. 32–34.
Rostami, H., Giri, A., Nejad, A.S.M., et al., Optimization of multiple shoot induction and plant regeneration in Indian barley (Hordeum vulgare) cultivars using mature embryos, Saudi J. Biol. Sci., 2013, vol. 20, pp. 251–255.
Sakkar, T., Thankappan, R., Mishra, G.P., and Nawa-de, B.D., Advances in the development and use of DREB for improved abiotic stress tolerance in transgenic crop plants, Physiol. Mol. Biol. Plants, 2019, vol. 25, pp. 1323–1334.
Sallam, A., Alqudah, A.M., Dawood, M. F., et al., Drought stress tolerance in wheat and barley: advances in physiology, breeding and genetics research, Int. J. Mol. Sci., 2019, vol. 20. https://doi.org/10.3390/ijms20133137
Sattar, S., Afzal, R., Bashir, I., et al., Biochemical, molecular and morpho-physiological attributes of wheat to upgrade grain production and compete with water stress, Int. J. Inn. Appl. Agric. Res., 2019, vol. 3, pp. 510–528.
Sel’dimirova, O.A., Testing of selective agents to evaluate the drought tolerance of spring soft wheat, Ekobiotekh, 2019, vol. 2, no. 1, pp. 51–62.
Sel’dimirova, O.A., Galin, I.R., Kruglova, N.N., and Veselov, D.S., Distribution of IAA and ABA in developing wheat germs in vivo, Izv. Ufimsk. Nauchn. Tsentra, Ross. Akad. Nauk, 2017a, no. 3 (1), pp. 114–118.
Seldimirova, O.A., Kruglova, N.N., Titova, G.E., and Batygina, T.B., Comparative ultrastructural analysis of the in vitro microspore embryoids and in vivo zygotic embryos of wheat as a basis for understanding of cytophysiological aspects of their development, Russ. J. Dev. Biol., 2017b, vol. 48, no. 3, pp. 185–197.
Sel’dimirova, O.A., Kruglova, N.N., Galin, I.R., and Veselov, D.S., Comparative evaluation of IAA, ABA, and cytokinin levels during the embryogenesis in vivo of barley sv. Steptoe and its ABA-deficient mutant AZ34, Ekobiotekh, 2018, vol. 1, no. 3, pp. 134–142.
Shamrov, I.I., Semyazachatok tsvetkovykh rastenii: stroenie, funktsii, proiskhozhdenie (The Ovule of Flowering Plants: Structure, Functions, and Origin), Moscow: KMK, 2008.
Shamrov, I.I., Embryogeny, in Embryology of Flowering Plants: Terminology and Concepts, Vol. 2: The Seed, Batygina, T.B., Ed., Boca Raton, FL: CRC Press, 2009, part 3, pp. 175–186.
Shamrov, I.I. and Anisimova, G.M., Critical stages of ovule and seed development, Acta Biol. Cracov, Ser. Bot., 2003, vol. 45, pp. 167–172.
Shupletsova, O.N. and Shchennikova, I.N., The use of cell technologies in creation of new barley varieties resistant to aluminum toxicity and drought, Vavilovskii Zh. Gene-t. Sel., 2016, vol. 20, no. 5, pp. 623–628.
Sonmezoglu, O.A. and Terzi, B., Characterization of some bread wheat genotypes using molecular markers for drought tolerance, Physiol. Mol. Biol. Plants, 2018, vol. 24, pp. 159–166.
Sugimoto K., Temman, H., Kadokura, S., and Matsuna-ga, S., To regenerate or not to regenerate: factors that drive plant regeneration, Curr. Opin. Plant Biol., 2019, vol. 47, pp. 138–150.
Tagimanova, D.S., Ergalieva, A.Zh., Raiser, O.B., and Khapilina, O.N., In vitro evaluation of spring soft wheat genotypes for drought resistance, Biotekhnol.: Teor. Prakt., 2013, no. 2, pp. 42–46.
Terekhin, E.S., Semya i semennoe razmnozhenie (A Seed and Seed Reproduction), St. Petersburg: Mir i Sem’ya-95, 1996.
Vartapetyan, B.B., Dolgikh, Yu.I., Polyakova, L.I., et al., Biotechnological approaches to creation of plants tolerant to hypoxia and anoxia, Acta Nat., 2014, vol. 6, no. 2 (21), pp. 21–33.
Vasilyeva, V.E. and Batygina, T.B., Autonomy of the embryo, in Embryology of Flowering Plants: Terminology and Concepts, Vol. 2: The Seed, Batygina, T.B., Ed., Boca Raton, FL: CRC Press, 2006, pp. 375–382.
Veselov, S.Yu., Sharipova, G.V., Timergalin, M.D., et al., Forecast of drought resistance by the content of abscisic acid and analysis of possible simplifying of the procedure for its quantitative assessment in wheat plants, Izv. Samar. Nauchn. Tsentra, Ross. Akad. Nauk, 2011, vol. 13, no. 5 (3), pp. 17–20.
Wang, X., Zenda, T., Liu, S., et al., Comparative proteomics and physiological analyses reveal important maize filling-kernel drought-responsive genes and metabolic pathways, Int. J. Mol. Sci., 2019, vol. 20, no. 15, p. 3743. https://doi.org/10.3390/ijms20153743
Wani, S., Kumar, V., Shriram, V., and Sah, S.K., Phytohormones and their metabolic engineering for abiotic stress tolerance in crop plants, Crop J., 2016, vol. 4, pp. 164–176.
Wareing, F.P. and Phillips, I.D.J., Growth and Differentiation in Plants, Oxford: Pergamon, 1981.
Yadav, S. and Sharma, K.D., Molecular and morphophysiological analysis of drought stress in plants, in Plant Growth, Rigobelo, E.C., Ed., London: InTechOpen, 2016, ch. 10. https://doi.org/10.5772/65246
Yadav, A.K., Carroll, A.J., Estavillo, G.M., et al., Wheat drought tolerance in the field is predicted by amino acid responses to glasshouse-imposed drought, J. Exp. Bot., 2019, vol. 70, pp. 4931–4948.
Yadav, B., Jogawat, A., Rahman, M.S., et al., Secondary metabolites in the drought stress tolerance of crop plants: a review, Gene Rep., 2021, vol. 23. https://doi.org/10.1016/j.genrep.2021.101040
Yang, C., Wang, D., Zhang, C., et al., Comprehensive analysis and expression profiling of PIN, AUX/LAX, and ABCB auxin transporter gene families in Solanum tuberosum under phytohormone stimuli and abiotic stresses, Biology, 2021, vol. 10, no. 2, p. 127. https://doi.org/10.3390/biology10020127
Zhang, W., Wang, X., Fan, R., et al., Effects of inter-culture, arabinogalactan proteins, and hydrogen peroxide on the plant regeneration of wheat immature embryos, J. Integr. Agric., 2015, vol. 14, pp. 11–19.
Zinatullina, A.E., Model system “embryo-embryonal callus” in express assessment of stress and anti-stress effects (by the example of cereals), Ekobiotekh, 2020, vol. 3, no. 1, pp. 38–50.
Zinchenko, M.A., Dubrovnaya, O.V., and Bavol, A.V., Cellular selection of soft wheat for resistance to a complex of stress factors and analysis of obtained forms, Izv. Samar. Nauchn. Tsentra, Ross. Akad. Nauk, 2013, vol. 15, no. 3 (5), pp. 1610–1614.
Zuraida, A.R., Naziah, B., Zamri, Z, and Sreeramanan, S., Efficient plant regeneration of Malaysian indica rice MR 219 and 232 via somatic embryogenesis system, Acta Physiol. Plant., 2011, vol. 33, pp. 1913–1921.
Funding
This study was carried out on project no. AAAA-A18-118022190099-6 within the framework of the State Assignment of Ministry of Science and Higher Education of the Russian Federation, no. 075-00326-19-00.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflicts of interest.
Statement on the welfare of humans or animals. This article does not contain any studies involving humans or animals performed by any of the authors.
Additional information
Translated by A. Aver’yanov
Rights and permissions
About this article
Cite this article
Kruglova, N.N., Zinatullina, A.E. In Vitro Culture of Autonomous Embryos as a Model System for the Study of Plant Stress Tolerance to Abiotic Factors (on the Example of Cereals). Biol Bull Rev 12, 201–211 (2022). https://doi.org/10.1134/S2079086422020050
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2079086422020050