Abstract
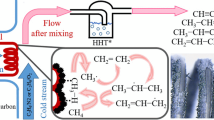
Oxidative transformations of methane on a catalyst (0.9 wt % of La2O3 + 0.1 wt % of CeO2)/MgO located inside the pores of a ceramic membrane occur at temperatures as low as 550°C with a high selectivity that was not previously observed, and terminate mainly with the formation of synthesis gas (carbon monoxide and hydrogen). The observed result is composed of the thermolysis reaction of methane yielding hydrogen and carbon, and comprises the subsequent reverse Buduar reaction. The reforming of carbon dioxide runs intensively when a methane-carbon dioxide mixture is fed into a membrane reactor at 650°C.
Similar content being viewed by others
References
Dedov, A.G., Loktev, A.S., Men’shikov, V.A., Moiseev, I.I., et al., Khim. Tekhnol., 2006, no. 4, pp. 5–11.
Makhlin, V.A., Dedov, A.G., Loktev, A.S., Moiseev, I.I., et al., Khim. Tekhnol., 2006, no. 7, pp. 29–34.
Arutyunov, V.S. and Krylov, O.V., Okislitel’nye prevrashcheniya metana (Oxidative Transformations of Methane), Moscow: Nauka, 1998.
Dedov, A.G., Loktev, A.S., Men’shikov, V.A., Moiseev, I.I., et al., Dokl. Akad. Nauk, 2001, vol. 380, no. 6, pp. 791–794 [Dokl. Chem. (Engl. Transl.), vol. 380, no. 6, pp. 301–304].
Borovinskaya, I.P., Merzhanov, A.G., and Uvarov, V.I., RF Patent no. 2175904, Byull. Izobret., 2002, no. 32.
Tsodikov, M.V., Teplyakov, V.V., Magsumov, M.I., et al., Kinet. Katal., 2006, vol. 47, no. 1, pp. 29–39 [Kinet. Catal. (Engl. Transl.), vol. 47, no. 1, pp. 132–135].
Teplyakov, V.V., Pisarev, G.I., Magsumov, M.I., et al, Catal. Today, 2006, vol. 118, pp. 7–11.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.G. Dedov, A.S. Loktev, K.V. Parkhomenko, M.V. Tsodikov, V.V. Teplyakov, V.I. Uvarov, A.S. Fedotov, I.I. Moiseev, 2008, published in Khimicheskaya Tekhnologiya, 2008, Vol. 9, No. 5, pp. 208–212.
Rights and permissions
About this article
Cite this article
Dedov, A.G., Loktev, A.S., Parkhomenko, K.V. et al. Oxidative transformations of methane: From the fixed bed to nanoreactors. Theor Found Chem Eng 43, 494–498 (2009). https://doi.org/10.1134/S0040579509040241
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0040579509040241