Abstract
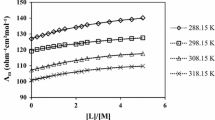
Conductometric titrations were performed in pure and binary solvent solutions of ethyl acetate (EtOAc), methyl acetate (MeOAc) and methanol (MeOH) with acetonitrile (AN) at 288, 298, 308, and 318 K to determine the stoichiometry, the complex stability constants and the standard thermodynamic parameters for the complexation of nickel(II) and cobalt(II) cations with 1,13-bis(8-quinolyl)-1,4,7,10,13-pentaoxatridecane (kryptofix 5). The stability constants of the resulting 1:1 complexes formed between the metal cations and the ligand in different solvent mixtures were determined by computer fitting of the conductance-mole ratio data. The results revealed that the stability order of (kryptofix 5)-Ni2+ and (kryptofix 5)-Co2+ complexes changes with nature and composition of the solvent system. There is a non-linear relationship between the logK f values of complexes and the mole fraction of acetonitrile in the mixed solvent system. In addition, the conductometric data show that the stoichiometry of the complexes formed between the nickel(II) and cobalt(II) cations with the acyclic ligand changes with the nature of the solvent. The enthalpy and entropy values for the 1: 1 [ML] complexation reactions were evaluated from the temperature dependence of the formation constants. Thermodynamically, in most of systems, the complexation processes of nickel(II) and cobalt(II) cations with kryptofix 5, are both enthalpy and entropy stabilized and the values of these parameters are influenced by the nature and composition of the binary mixed solvent solutions.
Similar content being viewed by others
References
M. Lamsa, K. Raitamaa, and J. Pursiainen, J. Phys. Org. Chem. 12, 557 (1999).
K.-S. Jeong, Y. L. Cho, and S. Y. Pyun, Tetrahedron Lett. 36, 2827 (1995).
L. E. Echegoyen, H. K. Yoo, V. J. Gatto, G. W. Gokel, and L. Echegoyen, J. Am. Chem. Soc. 111, 2440 (1989).
E. Weber and F. Vogtle, Tetrahedron Lett. 29, 2415 (1975).
F. Faridbod, M. R. Ganjali, R. Dinarvand, P. Norouzi, and S. Riahi, Sensors 8, 1645 (2008).
H. Tsukube, J. I. Uenishi, N. Kojima, and O. Yonemitsu, J. Tetrahedron Lett. 36, 2257 (1995).
K. Chayama and E. Sekido, J. Anal. Sci. 6, 883 (1990).
H. Tsukube, Tetrahedron Lett. 23, 2109 (1982).
C. Sirlin, M. Burgard, and M. J. F. Leroy, J. Membr. Sci. 54, 299 (1990).
F. Christy and P. S. Shrivastav, Crit. Rev. Anal. Chem. 41, 236 (2011).
P. D. Bernardo, A. Melchior, M. Tolazzi, and P. L. Zanonato, Coord. Chem. Rev. 256, 328 (2012).
N. S. Hush and J. R. Reimers, Chem. Rev. 100, 775 (2000).
M. R. Gholami and A. H. Yangieh, Int. J. Chem. Kinet. 33, 118 (2001).
C. Reichardt, Solvents and Solvent Effects in Organic Chemistry (VCH, New York, 1999).
Y. Marcus, J. Chem. Soc. Perkin. Trans. 2 1994, 1015 (1994).
V. S. Ijeri and A. K. Srivastava, Polyhedron. 22, 569 (2003).
G. H. Rounaghi, E. Razavipanah, and F. Kaveh, J. Incl. Phenom. Macrocycl. Chem. 68, 245 (2010).
G. H. Rounaghi, M. Mohajeri, M. Doaei, and A. Ghaemi, J. Incl. Phenom. Macrocycl. Chem. 67, 443 (2010).
G. H. Rounaghi, M. H. Soorgi, and M. S. Kazemi, Russ. J. Inorg. Chem. 55, 355 (2010).
G. H. Rounaghi, E. Nazari, A. Ghaemi, and M. Mohajeri, J. Coord. Chem. 63, 2349 (2010).
Y. Takeda and T. Kimura, J. Incl. Phenom. Mol. Recognit. Chem. 11, 159 (1991).
Genplot, A Data Analysis and Graphical Plotting Program for Scientists and Engineers (Computer Graphic Service, Ltd, Ithaca, New York, USA, 1989).
T. Madrakian and M. Shamsipur, Polish J. Chem. 73, 1405 (1999).
M. Payehghadr, A. Zamani, A. R. S. Sadaghiani, and M. Taghdiri, J. Incl. Phenom. Macrocycl. Chem. 62, 255 (2008).
M. Shamsipur, N. S. Gilani, M. K. Rofouei, and T. Madrakian, J. Mol. Liquids 160, 154 (2011).
P. A. Mosier-Boss and A. I. Popov, J. Am. Chem. Soc. 107, 6168 (1985).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Zarifpour, M., Rounaghi, G.H. & Tarahomi, S. Thermodynamic study of complex formation between Ni2+ and Co2+ cations and 1,13-bis(8-quinolyl)-1,4,7,10,13-pentaoxatridecane (kryptofix 5) in binary mixed nonaqueous solutions. Russ. J. Phys. Chem. 88, 1996–2003 (2014). https://doi.org/10.1134/S0036024414110211
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024414110211