Abstract
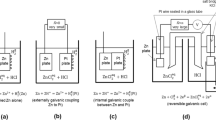
This article discusses changes in time of certain electrochemical properties (corrosion potential, Ψ′-potential, and corrosion inhibition coefficient) under residual protective action within corrosion of Zn and Fe. These properties reflect stage-by-stage desorption of preliminary adsorbed inhibitor. The pattern of their changes in time is explained in terms of changes in adsorption interaction metal-inhibitor taking into account the essence of metal and inhibitor. Rather high coefficients of acid-corrosion inhibition of Fe and Zn are attributed to the simultaneous action of some specific factors, which increase adsorption of inhibitor on metals.
Similar content being viewed by others
References
Grigor’ev, V.P., Shpan’ko, S.P., Boginskaya, V.V., and Plekhanova, E.V., Fizikokhim. Poverkhn. Zashch. Mater., 2010, no. 1, pp. 88–92.
Grigor’ev, V.P., Shpan’ko, S.P., Plekhanova, E.V., et al., Korroziya: Materialy, Zashchita, 2011, no. 4, pp. 39–43.
Khimiya i periodicheskaya sistema (Chemistry and Periodic System) Saito, K, Eds., Moscow: Mir, 1982.
Antropov, L.I., Zashch. Met., 1977, vol. 13, no. 4, pp. 387–399.
Antropov, L.I., Makushin, E.M., and Panasenko, V.F., Ingibitory korrozii metallov (Metal Corrosion Inhibitors), Kiev: Tekhnika, 1981.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.P. Grigor’ev, E.V. Plekhanova, A.S. Burlov, I.S. Vasil’chenko, T.A. Kuz’menko, 2013, published in Korroziya: Materialy, Zashchita, 2013, No. 5, pp. 22–27.
Rights and permissions
About this article
Cite this article
Grigor’ev, V.P., Plekhanova, E.V., Burlov, A.S. et al. Changes in certain electrochemical properties of Fe and Zn electrodes within residual protective action of an organic inhibitor. Prot Met Phys Chem Surf 50, 866–869 (2014). https://doi.org/10.1134/S2070205114070077
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205114070077