Abstract
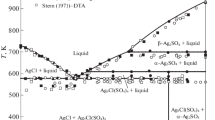
Phase equilibria in the systems zinc chloride - silver chloride and zinc bromide - silver bromide were investigated by differential scanning calorimetry, electric conductivity measurements and X-ray diffraction methods. No intermediate compounds were found. Solubility of components in the solid-state is negligible. Both systems are of the eutectic type. The eutectic temperatures in the ZnCl2-AgCl system and in the ZnBr2-AgBr system are 504 K and 551 K, respectively, and the compositions of the eutectic point are 53.5 mol% AgCl and 59.4 mol% AgBr, respectively.
Similar content being viewed by others
References
B. J. Aylett, The Chemistry of Zinc, Cadmium and Mercury, Pergamon Press 1973, pp. 209–214.
F. I. Galeener, J. C. Mikkelsen, Jr., A. C. Wright, R. N. Sinclair, J. A. E. Desa and J. Wong, J. Non-Cryst. Solids, 42 (1980) 23.
C. A. Angell and J. Wong, J. Chem. Phys., 53 (1979) 2053.
H. Hu, F. Ma and J. D. Mackenzie, J. Non-Cryst. Solids, 55 (1983) 169.
E. Kartini, M. F. Collins, F. Mezei and E. C. Svensson, Physica B, 241–243 (1998) 909.
‘Kristallstrukturdaten anorganischer Verbindungen’ in: Landolt-Börnstein, Neue Serie III/7a, Springer Verlag, Berlin 1973, p. 362–363.
J. Brynestad and H. L. Yakel, Inorg. Chem., 17 (1978) 1376.
H. L. Yakel and J. Brynestad, Inorg. Chem., 17 (1978) 3294.
F. A. Cotton, G. Wilkinson, C. A. Murillo and M. Bochmann, Advanced Inorganic Chemistry, 6th ed., John Wiley & Sons, Inc., New York 1999, p. 605.
D. Cubiciotti and H. Eding, J. Chem. Phys., 40 (1964) 978.
O. Kubaschewski and C. B. Alcock, Metallurgical Thermochemistry, 5th ed., Pergamon Press, Oxford 1979, p. 334.
N. I. Glistenko and G. I. Artemova, Trudy Khim. Fak. Voronezh. Gos. Univ., 32 (1953) 87.
Spravochnik po plavkostei sistem iż bezvodnykh neorganicheskikh solei, Vol. I, H. K. Voskresenskaya, Ed., Izd. Akad. Nauk SSSR, Moscow 1961, p. 34.
Ternary Alloys, A Comprehensive Compendium of Evaluated Constitutional Data and Phase Diagrams, Vol. 1, G. Petzow, G. Effenberg, Eds, VCH Verlagsgesellschaft mbH., Weinheim 1988, p. 522.
A. P. Palkin and N. A. Shchirova, Zh. Neorg. Khim., 1 (1956) 2155.
A. Wojakowska and E. Kundys, J. Mater. Sci., 25 (1990) 3780.
G. J. Janz, R. P. T. Tomkins, C. B. Allen, J. R. Downey, Jr. and S. K. Singer, J. Phys. Chem. Ref. Data, 6 (1977) 409.
E. Kordes, G. Ziegler and H. J. Proeger, Z. Elektrochem., 58 (1954) 168.
J. E. Ricci, ‘Phase Diagrams of Fused Salts’ in Molten Salt Chemistry, M. Blander, Ed., Interscience Publishers (John Wiley), New York 1964, 239–365.
G. Tamman, Z. Anorg. Chem., 47 (1905) 24.
L. Rycerz, E. Ingier-Stocka, M. Cieślak-Golonka and M. Gaune-Escard, J. Therm. Anal. Cal., 72 (2003) 241.
L. Rycerz, M. Cieślak-Golonka, E. Ingier-Stocka and M. Gaune-Escard, J. Therm. Anal. Cal., 72 (2003) 231.
J. W. Brightwell, C. N. Buckley, G. Foxton, L. S. Miller and B. Ray, J. Mater. Sci. Lett., 1 (1982) 429.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wojakowska, A., Górniak, A., Wojakowski, A. et al. Studies of phase equilibria in the systems ZnCl2-AgCl and ZnBr2-AgBr. Journal of Thermal Analysis and Calorimetry 77, 41–47 (2004). https://doi.org/10.1023/B:JTAN.0000033186.52150.8d
Issue Date:
DOI: https://doi.org/10.1023/B:JTAN.0000033186.52150.8d