Abstract
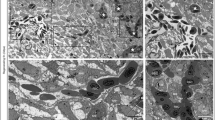
The rat aortic ring model has gained broad acceptance as an angiogenic assay. This system can be used to study the activity of angiogenic and anti-angiogenic factors, and investigate the molecular mechanisms of the angiogenic process. We describe here a thin prep modification of the aortic ring model, which significantly simplifies the procedure and allows staining of aortic outgrowths as whole mounts. Using this procedure, intact preparations of angiogenic outgrowths are successfully and reproducibly stained with endothelial cell (anti-CD-31 and -Tie2 antibodies, Griffonia Simplicifolia isolectin-B4) and smooth muscle cell (anti-α-smooth muscle actin antibody) markers. Combined use of double immunostaining and confocal microscopy allows concurrent visualization of endothelial and mural cells in the same cultures. Whole mount immunostains of rat aorta cultures are an effective way to rapidly characterize the cellular composition of the angiogenic outgrowths, and localize proteins implicated in the regulation of angiogenesis. This method should facilitate the work of the many vascular biologists that have adopted the rat aorta model as a tool to study angiogenesis and its mechanisms.
Similar content being viewed by others
References
Folkman J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med 1995; 1: 27–31.
Ferrara N, Alitalo K. Clinical applications ofangiogenic growth factors and their inhibitors. Nat Med 1999; 5: 1359–64.
Nelson MA, Passeri J, Frishman WH. Therapeutic angiogenesis: A new treatment modality for ischemic heart disease. Heart Dis 2000; 2: 314–25.
Folkman J. Tumor angiogenesis: Therapeutic implications. N Engl J Med 1971; 285: 1182–6.
Boehm T, Folkman J, Browder T et al. Antiangiogenic therapy of experimental cancer does not induce acquired drug resistance. Nature 1997; 390: 404–7.
Kim KJ, Li B, Winer J et al. Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 1993; 362: 841–4.
Saaristo A, Karpanen T, Alitalo K. Mechanisms of angiogenesis and their use in the inhibition of tumor growth and metastasis. Oncogene 2000; 19: 6122–9.
Bicknell R. Vascular targeting and the inhibition of angiogenesis. Ann Oncol 1994; 5 (Suppl 4): 45–50.
Billington DC. Angiogenesis and its inhibition: Potential new therapies in oncology and non-neoplastic diseases. Drug Des Discov 1991; 8: 3–35.
Aiello LP, Pierce EA, Foley ED et al. Suppression of retinal neovascularization in vivo by inhibition ofvascular endothelial growth factor (VEGF) using soluble VEGF-receptor chimeric proteins. Proc Natl Acad Sci USA 1995; 92: 10 457–61.
Freedman SB, Isner JM. Therapeutic angiogenesis for ischemic cardiovascular disease. J Mol Cell Cardiol 2001; 33: 379–93.
Jain RK, Schlenger K, Hockel M et al. Quantitative angiogenesis assays: Progress and problems. Nat Med 1997; 3: 1203–8.
Auerbach R, Auerbach W, Polakowski I. Assays for angiogenesis: A review. Pharmacol Ther 1991; 51: 1–11.
Nicosia RF, Ottinetti A. Growth of microvessels in serum-free matrix culture of rat aorta. A quantitative assay of angiogenesi s in vitro. Lab Invest 1990; 63: 115–22.
Nicosia RF. The rat aorta model of angiogenesis and its applications. In Mironov V, Little C, Sage H (eds): Microvascular Morphogenesis in vivo, in vitro and in mente.Boston: Birkhäuser 1998; 111–39.
Blacher S, Devy L, Burbridge MF et al. Improved quantification of angiogenesis in the rat aortic ring assay. Angiogenesis 2001; 4: 133–42.
Nicosia RF, Villaschi S. Rat aortic smooth muscle cells become pericytes during angiogenesis in vitro. Lab Invest 1995; 73: 658–66.
Nicosia RF, Tchao R, Leighton J. Histotypic angiogenesisin vitro: Light microscopic, ultrastructural, and radioautographic studies. In Vitro 1982; 18: 538–49.
Kruger EA, Duray PH, Price DK et al. Approaches to preclinical screening of antiangiog enic agents. Semin Oncol 2001; 28: 570–6.
Kruger EA, Duray PH, Tsokos MG et al. Endostatin inhibits microvessel formation in the ex vivo rat aortic ring angiogenesis assay. Biochem Biophys Res Commun 2000; 268: 183–91.
Malinda KM, Nomizu M, Chung M et al. Identification of laminin alpha1 and beta1 chain peptides active for endothelial cell adhesion, tube formation, and aortic sprouting. FASEB J 1999; 13: 53–62.
Huang L, Sankar S, Lin C et al. HCPTPA, a protein tyrosine phosphatase that regulates vascular endothelial growth factor receptor-mediated signal transduction and biological activity. J Biol Chem 1999; 274: 38,183–8.
Jaggers DC, Collins WP, Milligan SR. Potent inhibitory effects of steroids in anin vitro model ofangiogenesis. J Endocrinol 1996; 150: 457–64.
Belford DA, Gole GA, Rush RA. Localization of laminin to retinal vessels of the rat and mouse using whole mounts. Invest Ophthalmol Vis Sci 1987; 28: 1761–6.
D'Amato R, Wesolowski E, Smith LE. Microscopic visualization of the retina by angiography with high-molecular-weight fluorescein-labeled dextrans in the mouse. Microvasc Res 1993; 46: 135–42.
Nehls V, Denzer K, Drenckhahn D. Pericyte involvement in capillary sprouting during angiogenesis in situ. Cell Tissue Res 1992; 270: 469–74.
Chan-Ling T. Glial, vascular and neuronal cytogenesis in whole-mounted cat retina. Microsc Res Tech 1997; 36: 1–16.
Burbridge MF, West DC, Atassi G et al. The effect ofextracell ular pH on angiogenesis in vitro. Angiogenesis 1999; 3: 281–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, WH., Nicosia, R.F. The thin prep rat aortic ring assay: A modified method for the characterization of angiogenesis in whole mounts. Angiogenesis 5, 81–86 (2002). https://doi.org/10.1023/A:1021509004829
Issue Date:
DOI: https://doi.org/10.1023/A:1021509004829