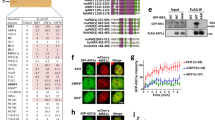
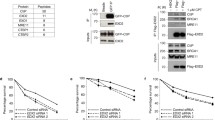
Abstract
To safeguard genome integrity, cells channel DNA double-strand breaks (DSBs) to repair pathway of either DNA end-joining or homology-directed repair. The MRE11-RAD50-NBS1 (MRN) complex functions in both pathways, teaming up with the right co-factors to ensure the correct repair of DSBs. The MRN complex is well known as a nuclease capable of resecting DNA in both endonucleolytic and exonucleolytic manners. At occupied DSB ends, DNA end resection is initiated by the “endonucleolytic cleavage followed by exonucleolytic digestion” action of MRN, with endonucleolytic cleavage specifically requiring phosphorylated CtIP being present. Previous studies by us and the other groups in the field help to uncover the mechanistic details of the MRN-CtIP nuclease ensemble functioning in DNA end resection and homologous recombination (HR), although many unclear parts still exist. Besides DSB repair, replication fork processing, R-loop, and transcription–replication conflict (TRC) resolution also have been suggested by accumulating studies to be relevant to MRN and CtIP. In this review, we will summarize current knowledge about the MRN-CtIP nuclease ensemble, its functions in DNA processing in various contexts that could generate genome instability, how this complex is regulated and its relevance to diseases like cancers.



Similar content being viewed by others
References
Anand, R., Jasrotia, A., Bundschuh, D., Howard, S. M., Ranjha, L., Stucki, M., & Cejka, P. (2019). NBS1 promotes the endonuclease activity of the MRE11-RAD50 complex by sensing CtIP phosphorylation. EMBO Journal. https://doi.org/10.15252/embj.2018101005
Anand, R., Ranjha, L., Cannavo, E., & Cejka, P. (2016). Phosphorylated CtIP functions as a Co-factor of the MRE11-RAD50-NBS1 endonuclease in DNA end resection. Molecular Cell, 64(5), 940–950. https://doi.org/10.1016/j.molcel.2016.10.017
Anderson, D. E., Trujillo, K. M., Sung, P., & Erickson, H. P. (2001). Structure of the Rad50 x Mre11 DNA repair complex from Saccharomyces cerevisiae by electron microscopy. Journal of Biological Chemistry, 276(40), 37027–37033. https://doi.org/10.1074/jbc.M106179200
Andres, S. N., Appel, C. D., Westmoreland, J. W., Williams, J. S., Nguyen, Y., Robertson, P. D., & Williams, R. S. (2015). Tetrameric Ctp1 coordinates DNA binding and DNA bridging in DNA double-strand-break repair. Nature Structural & Molecular Biology, 22(2), 158–166. https://doi.org/10.1038/nsmb.2945
Andres, S. N., Li, Z. M., Erie, D. A., & Williams, R. S. (2019). Ctp1 protein–DNA filaments promote DNA Bridging and DNA double-strand break repair. Journal of Biological Chemistry. https://doi.org/10.1074/jbc.RA118.006759
Atkinson, J., & McGlynn, P. (2009). Replication fork reversal and the maintenance of genome stability. Nucleic Acids Research, 37(11), 3475–3492. https://doi.org/10.1093/nar/gkp244
Bai, Y., Wang, W., Li, S., Zhan, J., Li, H., Zhao, M., & Wang, J. (2019). C1QBP promotes homologous recombination by stabilizing MRE11 and controlling the assembly and activation of MRE11/RAD50/NBS1 complex. Molecular Cell, 75(6), 1299–1314. https://doi.org/10.1016/j.molcel.2019.06.023
Bennett, L. G., Wilkie, A. M., Antonopoulou, E., Ceppi, I., Sanchez, A., Vernon, E. G., & Staples, C. J. (2020). MRNIP is a replication fork protection factor. Science Advances, 6(28), eaba5974. https://doi.org/10.1126/sciadv.aba5974
Boisvert, F. M., Dery, U., Masson, J. Y., & Richard, S. (2005). Arginine methylation of MRE11 by PRMT1 is required for DNA damage checkpoint control. Genes and Development, 19(6), 671–676. https://doi.org/10.1101/gad.1279805
Broderick, R., Nieminuszczy, J., Baddock, H. T., Deshpande, R., Gileadi, O., Paull, T. T., & Niedzwiedz, W. (2016). EXD2 promotes homologous recombination by facilitating DNA end resection. Nature Cell Biology, 18(3), 271–280. https://doi.org/10.1038/ncb3303
Bryant, H. E., Petermann, E., Schultz, N., Jemth, A. S., Loseva, O., Issaeva, N., & Helleday, T. (2009). PARP is activated at stalled forks to mediate Mre11-dependent replication restart and recombination. EMBO Journal, 28(17), 2601–2615. https://doi.org/10.1038/emboj.2009.206
Cannan, W. J., & Pederson, D. S. (2016). Mechanisms and consequences of double-strand DNA break formation in chromatin. Journal of Cellular Physiology, 231(1), 3–14. https://doi.org/10.1002/jcp.25048
Cannavo, E., & Cejka, P. (2014). Sae2 promotes dsDNA endonuclease activity within Mre11-Rad50-Xrs2 to resect DNA breaks. Nature, 514(7520), 122–125. https://doi.org/10.1038/nature13771
Cannavo, E., Johnson, D., Andres, S. N., Kissling, V. M., Reinert, J. K., Garcia, V., & Cejka, P. (2018). Regulatory control of DNA end resection by Sae2 phosphorylation. Nature Communications, 9(1), 4016. https://doi.org/10.1038/s41467-018-06417-5
Casari, E., Rinaldi, C., Marsella, A., Gnugnoli, M., Colombo, C. V., Bonetti, D., & Longhese, M. P. (2019). Processing of DNA double-strand breaks by the MRX complex in a chromatin context. Frontiers in Molecular Biosciences, 6, 43. https://doi.org/10.3389/fmolb.2019.00043
Castillo-Villanueva, E., Ballesteros, G., Schmid, M., Hidalgo, P., Schreiner, S., Dobner, T., & Gonzalez, R. A. (2014). The Mre11 cellular protein is modified by conjugation of both SUMO-1 and SUMO-2/3 during adenovirus infection. ISRN Virology, 2014, 1–14. https://doi.org/10.1155/2014/989160
Chang, E. Y., Tsai, S., Aristizabal, M. J., Wells, J. P., Coulombe, Y., Busatto, F. F., & Stirling, P. C. (2019). MRE11-RAD50-NBS1 promotes Fanconi anemia R-loop suppression at transcription-replication conflicts. Nature Communications, 10(1), 4265. https://doi.org/10.1038/s41467-019-12271-w
Chen, L., Trujillo, K., Ramos, W., Sung, P., & Tomkinson, A. E. (2001). Promotion of Dnl4-catalyzed DNA end-joining by the Rad50/Mre11/Xrs2 and Hdf1/Hdf2 complexes. Molecular Cell, 8(5), 1105–1115. http://www.ncbi.nlm.nih.gov/pubmed/11741545
Choi, K. S., Kim, J. Y., Lim, S. K., Choi, Y. W., Kim, Y. H., Kang, S. Y., & Lim, I. K. (2012). TIS21(/BTG2/PC3) accelerates the repair of DNA double strand breaks by enhancing Mre11 methylation and blocking damage signal transfer to the Chk 2(T68)-p53(S20) pathway. DNA Repair (amst), 11(12), 965–975. https://doi.org/10.1016/j.dnarep.2012.09.009
Coquel, F., Silva, M. J., Techer, H., Zadorozhny, K., Sharma, S., Nieminuszczy, J., & Pasero, P. (2018). SAMHD1 acts at stalled replication forks to prevent interferon induction. Nature, 557(7703), 57–61. https://doi.org/10.1038/s41586-018-0050-1
Costantino, L., & Koshland, D. (2015). The Yin and Yang of R-loop biology. Current Opinion in Cell Biology, 34, 39–45. https://doi.org/10.1016/j.ceb.2015.04.008
de Jager, M., van Noort, J., van Gent, D. C., Dekker, C., Kanaar, R., & Wyman, C. (2001). Human Rad50/Mre11 is a flexible complex that can tether DNA ends. Molecular Cell, 8(5), 1129–1135. https://doi.org/10.1016/s1097-2765(01)00381-1
Delamarre, A., Barthe, A., de la Roche Saint-Andre, C., Luciano, P., Forey, R., Padioleau, I., & Lengronne, A. (2020). MRX increases chromatin accessibility at stalled replication forks to promote nascent DNA resection and cohesin loading. Molecular Cell, 77(2), 395–410. https://doi.org/10.1016/j.molcel.2019.10.029
Deshpande, R. A., Lee, J. H., Arora, S., & Paull, T. T. (2016). Nbs1 converts the human Mre11/Rad50 nuclease complex into an endo/exonuclease machine specific for protein-DNA adducts. Molecular Cell, 64(3), 593–606. https://doi.org/10.1016/j.molcel.2016.10.010
Falck, J., Forment, J. V., Coates, J., Mistrik, M., Lukas, J., Bartek, J., & Jackson, S. P. (2012). CDK targeting of NBS1 promotes DNA-end resection, replication restart and homologous recombination. EMBO Reports, 13(6), 561–568. https://doi.org/10.1038/embor.2012.58
Gao, M., Guo, G., Huang, J., Kloeber, J. A., Zhao, F., Deng, M., & Lou, Z. (2020). USP52 regulates DNA end resection and chemosensitivity through removing inhibitory ubiquitination from CtIP. Nature Communications, 11(1), 5362. https://doi.org/10.1038/s41467-020-19202-0
Garcia, V., Phelps, S. E., Gray, S., & Neale, M. J. (2011). Bidirectional resection of DNA double-strand breaks by Mre11 and Exo1. Nature, 479(7372), 241–244. https://doi.org/10.1038/nature10515
Ghodke, I., & Muniyappa, K. (2013). Processing of DNA double-stranded breaks and intermediates of recombination and repair by Saccharomyces cerevisiae Mre11 and its stimulation by Rad50, Xrs2, and Sae2 proteins. Journal of Biological Chemistry, 288(16), 11273–11286. https://doi.org/10.1074/jbc.M112.439315
Ha, G. H., Ji, J. H., Chae, S., Park, J., Kim, S., Lee, J. K., & Lee, C. W. (2019). Pellino1 regulates reversible ATM activation via NBS1 ubiquitination at DNA double-strand breaks. Nature Communications, 10(1), 1577. https://doi.org/10.1038/s41467-019-09641-9
Han, J., Wan, L., Jiang, G., Cao, L., Xia, F., Tian, T., & Huang, J. (2021). ATM controls the extent of DNA end resection by eliciting sequential posttranslational modifications of CtIP. Proceedings of the National Academy of Sciences, 118, 12. https://doi.org/10.1073/pnas.2022600118
He, Y. J., Meghani, K., Caron, M. C., Yang, C., Ronato, D. A., Bian, J., & Chowdhury, D. (2018). DYNLL1 binds to MRE11 to limit DNA end resection in BRCA1-deficient cells. Nature, 563(7732), 522–526. https://doi.org/10.1038/s41586-018-0670-5
Hopfner, K. P., Craig, L., Moncalian, G., Zinkel, R. A., Usui, T., Owen, B. A., & Tainer, J. A. (2002). The Rad50 zinc-hook is a structure joining Mre11 complexes in DNA recombination and repair. Nature, 418(6897), 562–566. https://doi.org/10.1038/nature00922
Hopfner, K. P., Karcher, A., Shin, D. S., Craig, L., Arthur, L. M., Carney, J. P., & Tainer, J. A. (2000). Structural biology of Rad50 ATPase: ATP-driven conformational control in DNA double-strand break repair and the ABC-ATPase superfamily. Cell, 101(7), 789–800. https://doi.org/10.1016/s0092-8674(00)80890-9
Huertas, P., & Jackson, S. P. (2009). Human CtIP mediates cell cycle control of DNA end resection and double strand break repair. Journal of Biological Chemistry, 284(14), 9558–9565. https://doi.org/10.1074/jbc.M808906200
Jachimowicz, R. D., Beleggia, F., Isensee, J., Velpula, B. B., Goergens, J., Bustos, M. A., & Shiloh, Y. (2019). UBQLN4 represses homologous recombination and is overexpressed in aggressive tumors. Cell, 176(3), 505–519. https://doi.org/10.1016/j.cell.2018.11.024
Kilgas, S., Singh, A. N., Paillas, S., Then, C. K., Torrecilla, I., Nicholson, J., & Ramadan, K. (2021). p97/VCP inhibition causes excessive MRE11-dependent DNA end resection promoting cell killing after ionizing radiation. Cell Reports, 35(8), 109153. https://doi.org/10.1016/j.celrep.2021.109153
Kim, J. J., Lee, S. Y., Choi, J. H., Woo, H. G., Xhemalce, B., & Miller, K. M. (2020). PCAF-mediated histone acetylation promotes replication fork degradation by MRE11 and EXO1 in BRCA-deficient cells. Molecular Cell, 80(2), 327–344. https://doi.org/10.1016/j.molcel.2020.08.018
Lavin, M. F., Kozlov, S., Gatei, M., & Kijas, A. W. (2015). ATM-dependent phosphorylation of all three members of the MRN complex: from sensor to adaptor. Biomolecules, 5(4), 2877–2902. https://doi.org/10.3390/biom5042877
Lee, J. H., & Paull, T. T. (2004). Direct activation of the ATM protein kinase by the Mre11/Rad50/Nbs1 complex. Science, 304(5667), 93–96. https://doi.org/10.1126/science.1091496
Lee, L., Perez Oliva, A. B., Martinez-Balsalobre, E., Churikov, D., Peter, J., Rahmouni, D., & Lachaud, C. (2021). UFMylation of MRE11 is essential for telomere length maintenance and hematopoietic stem cell survival. Science Advance, 7(39), 7371. https://doi.org/10.1126/sciadv.abc7371
Lim, H. S., Kim, J. S., Park, Y. B., Gwon, G. H., & Cho, Y. (2011). Crystal structure of the Mre11-Rad50-ATPgammaS complex: Understanding the interplay between Mre11 and Rad50. Genes and Development, 25(10), 1091–1104. https://doi.org/10.1101/gad.2037811
Liu, H., Zhang, H., Wang, X., Tian, Q., Hu, Z., Peng, C., & Pei, H. (2015). The deubiquitylating enzyme USP4 cooperates with CtIP in DNA double-strand break end resection. Cell Reports, 13(1), 93–107. https://doi.org/10.1016/j.celrep.2015.08.056
Liu, S., Hua, Y., Wang, J., Li, L., Yuan, J., Zhang, B., & Kong, D. (2021). RNA polymerase III is required for the repair of DNA double-strand breaks by homologous recombination. Cell, 184(5), 1314–1329. https://doi.org/10.1016/j.cell.2021.01.048
Lou, J., Chen, H., Han, J., He, H., Huen, M. S. Y., Feng, X. H., & Huang, J. (2017). AUNIP/C1orf135 directs DNA double-strand breaks towards the homologous recombination repair pathway. Nature Communications, 8(1), 985. https://doi.org/10.1038/s41467-017-01151-w
Lyu, X., Lei, K. H., Biak Sang, P., Shiva, O., Chastain, M., Chi, P., & Chai, W. (2021). Human CST complex protects stalled replication forks by directly blocking MRE11 degradation of nascent-strand DNA. EMBO Journal, 40(2), e103654. https://doi.org/10.15252/embj.2019103654
Makharashvili, N., Arora, S., Yin, Y., Fu, Q., Wen, X., Lee, J. H., & Paull, T. T. (2018). Sae2/CtIP prevents R-loop accumulation in eukaryotic cells. eLife. https://doi.org/10.7554/eLife.42733
Manthey, K. C., Opiyo, S., Glanzer, J. G., Dimitrova, D., Elliott, J., & Oakley, G. G. (2007). NBS1 mediates ATR-dependent RPA hyperphosphorylation following replication-fork stall and collapse. Journal of Cell Science, 120(Pt 23), 4221–4229. https://doi.org/10.1242/jcs.004580
Michelini, F., Pitchiaya, S., Vitelli, V., Sharma, S., Gioia, U., Pessina, F., & d’Adda di Fagagna, F. (2017). Damage-induced lncRNAs control the DNA damage response through interaction with DDRNAs at individual double-strand breaks. Nature Cell Biology, 19(12), 1400–1411. https://doi.org/10.1038/ncb3643
Mimitou, E. P., & Symington, L. S. (2008). Sae2, Exo1 and Sgs1 collaborate in DNA double-strand break processing. Nature, 455(7214), 770–774. https://doi.org/10.1038/nature07312
Moreno-Herrero, F., de Jager, M., Dekker, N. H., Kanaar, R., Wyman, C., & Dekker, C. (2005). Mesoscale conformational changes in the DNA-repair complex Rad50/Mre11/Nbs1 upon binding DNA. Nature, 437(7057), 440–443. https://doi.org/10.1038/nature03927
Myler, L. R., Gallardo, I. F., Soniat, M. M., Deshpande, R. A., Gonzalez, X. B., Kim, Y., & Finkelstein, I. J. (2017). Single-molecule imaging reveals how Mre11-Rad50-Nbs1 initiates DNA break repair. Molecular Cell. https://doi.org/10.1016/j.molcel.2017.08.002
Nath, S., & Nagaraju, G. (2020). FANCJ helicase promotes DNA end resection by facilitating CtIP recruitment to DNA double-strand breaks. PLoS Genetics, 16(4), e1008701. https://doi.org/10.1371/journal.pgen.1008701
Ohle, C., Tesorero, R., Schermann, G., Dobrev, N., Sinning, I., & Fischer, T. (2016). Transient RNA-DNA hybrids are required for efficient double-strand break repair. Cell, 167(4), 1001–1013. https://doi.org/10.1016/j.cell.2016.10.001
Ouyang, J., Yadav, T., Zhang, J. M., Yang, H., Rheinbay, E., Guo, H., & Zou, L. (2021). RNA transcripts stimulate homologous recombination by forming DR-loops. Nature, 594(7862), 283–288. https://doi.org/10.1038/s41586-021-03538-8
Park, Y. B., Chae, J., Kim, Y. C., & Cho, Y. (2011). Crystal structure of human Mre11: Understanding tumorigenic mutations. Structure, 19(11), 1591–1602. https://doi.org/10.1016/j.str.2011.09.010
Paudyal, S. C., & You, Z. (2016). Sharpening the ends for repair: Mechanisms and regulation of DNA resection. Acta Biochimica Et Biophysica Sinica (shanghai), 48(7), 647–657. https://doi.org/10.1093/abbs/gmw043
Paull, T. T., & Gellert, M. (1998). The 3' to 5' exonuclease activity of Mre 11 facilitates repair of DNA double-strand breaks. Molecular Cell, 1(7), 969–979. http://www.ncbi.nlm.nih.gov/pubmed/9651580
Ranjha, L., Howard, S. M., & Cejka, P. (2018). Main steps in DNA double-strand break repair: An introduction to homologous recombination and related processes. Chromosoma, 127(2), 187–214. https://doi.org/10.1007/s00412-017-0658-1
Ray Chaudhuri, A., Callen, E., Ding, X., Gogola, E., Duarte, A. A., Lee, J. E., & Nussenzweig, A. (2016). Replication fork stability confers chemoresistance in BRCA-deficient cells. Nature, 535(7612), 382–387. https://doi.org/10.1038/nature18325
Salifou, K., Burnard, C., Basavarajaiah, P., Grasso, G., Helsmoortel, M., Mac, V., & Kiernan, R. (2021). Chromatin-associated MRN complex protects highly transcribing genes from genomic instability. Science Advance, 7, 21. https://doi.org/10.1126/sciadv.abb2947
Sanchez-Bailon, M. P., Choi, S. Y., Dufficy, E. R., Sharma, K., McNee, G. S., Gunnell, E., & Davies, C. C. (2021). Arginine methylation and ubiquitylation crosstalk controls DNA end-resection and homologous recombination repair. Nature Communications, 12(1), 6313. https://doi.org/10.1038/s41467-021-26413-6
Santos-Pereira, J. M., & Aguilera, A. (2015). R loops: New modulators of genome dynamics and function. Nature Reviews Genetics, 16(10), 583–597. https://doi.org/10.1038/nrg3961
Sohn, S. Y., & Hearing, P. (2012). Adenovirus regulates sumoylation of Mre11-Rad50-Nbs1 components through a paralog-specific mechanism. Journal of Virology, 86(18), 9656–9665. https://doi.org/10.1128/JVI.01273-12
Staples, C. J., Barone, G., Myers, K. N., Ganesh, A., Gibbs-Seymour, I., Patil, A. A., & Collis, S. J. (2016). MRNIP/C5orf45 interacts with the MRN complex and contributes to the DNA damage response. Cell Reports, 16(10), 2565–2575. https://doi.org/10.1016/j.celrep.2016.07.087
Studach, L., Wang, W. H., Weber, G., Tang, J., Hullinger, R. L., Malbrue, R., & Andrisani, O. (2010). Polo-like kinase 1 activated by the hepatitis B virus X protein attenuates both the DNA damage checkpoint and DNA repair resulting in partial polyploidy. Journal of Biological Chemistry, 285(39), 30282–30293. https://doi.org/10.1074/jbc.M109.093963
Syed, A., & Tainer, J. A. (2018). The MRE11-RAD50-NBS1 complex conducts the orchestration of damage signaling and outcomes to stress in DNA replication and repair. Annual Review of Biochemistry, 87, 263–294. https://doi.org/10.1146/annurev-biochem-062917-012415
Symington, L. S., & Gautier, J. (2011). Double-strand break end resection and repair pathway choice. Annual Review of Genetics, 45, 247–271. https://doi.org/10.1146/annurev-genet-110410-132435
Taylor, M. R., Spirek, M., Chaurasiya, K. R., Ward, J. D., Carzaniga, R., Yu, X., & Boulton, S. J. (2015). Rad51 paralogs remodel pre-synaptic Rad51 filaments to stimulate homologous recombination. Cell, 162(2), 271–286. https://doi.org/10.1016/j.cell.2015.06.015
Thomas, M., White, R. L., & Davis, R. W. (1976). Hybridization of RNA to double-stranded DNA: Formation of R-loops. Proceedings of the National Academy of Sciences of the United States of America, 73(7), 2294–2298. https://doi.org/10.1073/pnas.73.7.2294
Trujillo, K. M., Yuan, S. S., Lee, E. Y., & Sung, P. (1998). Nuclease activities in a complex of human recombination and DNA repair factors Rad50, Mre11, and p95. Journal of Biological Chemistry, 273(34), 21447–21450. http://www.ncbi.nlm.nih.gov/pubmed/9705271
Trujillo, K. M., Roh, D. H., Chen, L., Van Komen, S., Tomkinson, A., & Sung, P. (2003). Yeast Xrs2 binds DNA and helps target Rad50 and Mre11 to DNA ends. Journal of Biological Chemistry, 278(49), 48957–48964. https://doi.org/10.1074/jbc.M309877200
Trujillo, K. M., & Sung, P. (2001). DNA structure-specific nuclease activities in the Saccharomyces cerevisiae Rad50-Mre11 complex. Journal of Biological Chemistry, 276(38), 35458–35464. https://doi.org/10.1074/jbc.M105482200
Vadnais, C., Chen, R., Fraszczak, J., Yu, Z., Boulais, J., Pinder, J., & Moroy, T. (2018). GFI1 facilitates efficient DNA repair by regulating PRMT1 dependent methylation of MRE11 and 53BP1. Nature Communications, 9(1), 1418. https://doi.org/10.1038/s41467-018-03817-5
von Morgen, P., Burdova, K., Flower, T. G., O’Reilly, N. J., Boulton, S. J., Smerdon, S. J., & Horejsi, Z. (2017). MRE11 stability is regulated by CK2-dependent interaction with R2TP complex. Oncogene, 36(34), 4943–4950. https://doi.org/10.1038/onc.2017.99
Wang, W., Daley, J. M., Kwon, Y., Krasner, D. S., & Sung, P. (2017). Plasticity of the Mre11-Rad50-Xrs2-Sae2 nuclease ensemble in the processing of DNA-bound obstacles. Genes and Development, 31(23–24), 2331–2336. https://doi.org/10.1101/gad.307900.117
Wang, W., Daley, J. M., Kwon, Y., Xue, X., Krasner, D. S., Miller, A. S., & Sung, P. (2018). A DNA nick at Ku-blocked double-strand break ends serves as an entry site for exonuclease 1 (Exo1) or Sgs1-Dna2 in long-range DNA end resection. Journal of Biological Chemistry, 293(44), 17061–17069. https://doi.org/10.1074/jbc.RA118.004769
Wang, Z., Gong, Y., Peng, B., Shi, R., Fan, D., Zhao, H., & Xu, X. (2019). MRE11 UFMylation promotes ATM activation. Nucleic Acids Research, 47(8), 4124–4135. https://doi.org/10.1093/nar/gkz110
Wijnhoven, P., Konietzny, R., Blackford, A. N., Travers, J., Kessler, B. M., Nishi, R., & Jackson, S. P. (2015). USP4 auto-deubiquitylation promotes homologous recombination. Molecular Cell, 60(3), 362–373. https://doi.org/10.1016/j.molcel.2015.09.019
Wilkinson, O. J., Martin-Gonzalez, A., Kang, H., Northall, S. J., Wigley, D. B., Moreno-Herrero, F., & Dillingham, M. S. (2019). CtIP forms a tetrameric dumbbell-shaped particle which bridges complex DNA end structures for double-strand break repair. eLife. https://doi.org/10.7554/eLife.42129
Williams, R. S., Dodson, G. E., Limbo, O., Yamada, Y., Williams, J. S., Guenther, G., & Tainer, J. A. (2009). Nbs1 flexibly tethers Ctp1 and Mre11-Rad50 to coordinate DNA double-strand break processing and repair. Cell, 139(1), 87–99. https://doi.org/10.1016/j.cell.2009.07.033
Williams, R. S., Moncalian, G., Williams, J. S., Yamada, Y., Limbo, O., Shin, D. S., & Tainer, J. A. (2008). Mre11 dimers coordinate DNA end bridging and nuclease processing in double-strand-break repair. Cell, 135(1), 97–109. https://doi.org/10.1016/j.cell.2008.08.017
Wu, J., Zhang, X., Zhang, L., Wu, C. Y., Rezaeian, A. H., Chan, C. H., & Lin, H. K. (2012). Skp2 E3 ligase integrates ATM activation and homologous recombination repair by ubiquitinating NBS1. Molecular Cell, 46(3), 351–361. https://doi.org/10.1016/j.molcel.2012.02.018
Yu, X., Fu, S., Lai, M., Baer, R., & Chen, J. (2006). BRCA1 ubiquitinates its phosphorylation-dependent binding partner CtIP. Genes and Development, 20(13), 1721–1726. https://doi.org/10.1101/gad.1431006
Yuan, Z., Zhang, X., Sengupta, N., Lane, W. S., & Seto, E. (2007). SIRT1 regulates the function of the Nijmegen breakage syndrome protein. Molecular Cell, 27(1), 149–162. https://doi.org/10.1016/j.molcel.2007.05.029
Zhao, W., Steinfeld, J. B., Liang, F., Chen, X., Maranon, D. G., Jian Ma, C., & Sung, P. (2017). BRCA1-BARD1 promotes RAD51-mediated homologous DNA pairing. Nature, 550(7676), 360–365. https://doi.org/10.1038/nature24060
Zhao, W., Vaithiyalingam, S., San Filippo, J., Maranon, D. G., Jimenez-Sainz, J., Fontenay, G. V., & Sung, P. (2015). Promotion of BRCA2-dependent homologous recombination by DSS1 via RPA targeting and DNA mimicry. Molecular Cell, 59(2), 176–187. https://doi.org/10.1016/j.molcel.2015.05.032
Zhao, W., Wiese, C., Kwon, Y., Hromas, R., & Sung, P. (2019). The BRCA Tumor Suppressor Network in Chromosome Damage Repair by Homologous Recombination. Annual Review of Biochemistry. https://doi.org/10.1146/annurev-biochem-013118-111058
Acknowledgements
This review was supported by grants from the National Natural Science Foundation of China (81972608 and 82172951).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, T., Zhou, Z., Yang, H. et al. MRE11-RAD50-NBS1-CtIP: one key nuclease ensemble functions in the maintenance of genome stability. GENOME INSTAB. DIS. 3, 123–135 (2022). https://doi.org/10.1007/s42764-022-00065-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42764-022-00065-2