Abstract
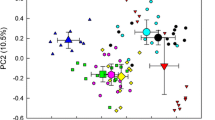
Little is known about how bacterial endophyte communities may vary from one tree to another of the same species. We sampled nine standardized replicates from the green stems of an ethnomedicinal tropical tree, Santiria apiculata, in a Malaysian rainforest, to investigate endophyte variation from one plant to another. DNA was extracted axenically from the inner stem, and PCR-amplified for the 16s rRNA gene, followed by Illumina HiSeq sequencing. For comparison, the rhizosphere soil of the same trees was also sampled, and the 16s rRNA genes sequenced. Santiria stems contained a diverse community of more than 1100 Operational Taxonomic Units (OTUs), which partitioned clearly into two community types. One community (here dubbed ‘rhizosphere-like’) resembled the rhizosphere soil in its major OTUs, while the other (‘rhizosphere-unlike’) was more distinct and dominated by the genus Bacillus. Nestedness analysis confirmed that both the rhizosphere-like and the rhizosphere-unlike stem communities tended to be a subset of the rhizosphere community. The β-nearest taxon index (βNTI) analysis revealed that the stem communities were dominated by stochastic processes, whereas deterministic processes with homogenizing selection dominated the rhizosphere soil. It appears that the endophyte community in Santiria may be dominated by colonization and priority effects, and it may derive from the rhizosphere community. The potential for alternate steady states within the endophyte community should be considered as a possible influence on plant growth and ecology.






Similar content being viewed by others
Availability of data and materials
References
Adebomojo AA, AbdulRahaman AA (2020) Surface sterilization of Ocimum seeds and tissues with biosynthesized nanosilver and its effects on callus induction. In IOP conference series: materials science and engineering, Vol. 805, No. 1. IOP Publishing, p 012024
Afzal I, Shinwari ZK, Sikandar S, Shahzad S (2019) Plant beneficial endophytic bacteria: mechanisms, diversity, host range and genetic determinants. Microbiol Res. https://doi.org/10.1016/j.micres.2019.02.001
Alina SO, Constantinscu F, Petruţa CC (2015) Biodiversity of Bacillus subtilis group and beneficial traits of Bacillus species useful in plant protection. Roman Biotechnol Lett 20(5):10737–10750
Andrić S, Meyer T, Ongena M (2020) Bacillus responses to plant-associated fungal and bacterial communities. Front Microbiol 10:10. https://doi.org/10.3389/fmicb.2020.01350
Aziz RK, Bartels D, Best AA, DeJongh M, Disz T, Edwards RA et al (2008) The RAST server: rapid annotations using subsystems technology. BMC Genom 9(1):75. https://doi.org/10.1186/1471-2164-9-75
Baba MS, Zin NM, Hassan ZAA, Latip J, Pethick F, Hunter IS et al (2015) In vivo antimalarial activity of the endophytic actinobacteria, Streptomyces SUK 10. J Microbiol 53(12):847–855. https://doi.org/10.1007/s12275-015-5076-6
Bakeri SA, Maidin MST, Masri MMM (2019) Soil bacterial biodiversity in development of secondary logged-over forest to oil palm plantation in mineral soil of Belaga, Sarawak. J Oil Palm Res 31(3):394–411. https://doi.org/10.21894/jopr.2019.0043
Beckers B, Op De Beeck M, Thijs S, Truyens S, Weyens N, Boerjan W, Vangronsveld J (2016) Performance of 16s rDNA primer pairs in the study of rhizosphere and endosphere bacterial microbiomes in metabarcoding studies. Front Microbiol. https://doi.org/10.3389/fmicb.2016.00650
Bhattacharyya PN, Jha DK (2012) Plant growth-promoting rhizobacteria (PGPR): emergence in agriculture. World J Microbiol Biotechnol 28(4):1327–1350. https://doi.org/10.1007/s11274-011-0979-9
Bhore S, Deivanai S, Bindusara A, Prabhakaran G (2014) Culturable bacterial endophytes isolated from Mangrove tree (Rhizophora apiculata Blume) enhance seedling growth in Rice. J Nat Sci Biol Med 5(2):437. https://doi.org/10.4103/0976-9668.136233
Bove JM, Garnier M (2002) Phloem-and xylem-restricted plant pathogenic bacteria. Plant Sci 163:1083–1098
Brader G, Compant S, Mitter B, Trognitz F, Sessitsch A (2014) Metabolic potential of endophytic bacteria. Curr Opin Biotechnol 27:30–37. https://doi.org/10.1016/j.copbio.2013.09.012
Cardoso VM, Campos FF, Santos ARO, Ottoni MHF, Rosa CA, Almeida VG, Grael CFF (2020) Biotechnological applications of the medicinal plant Pseudobrickellia brasiliensis and its isolated endophytic bacteria. J Appl Microbiol 129(4):926–934. https://doi.org/10.1111/jam.14666
Caulier S, Nannan C, Gillis A, Licciardi F, Bragard C, Mahillon J (2019) Overview of the antimicrobial compounds produced by members of the Bacillus subtilis group. Front Microbiol. https://doi.org/10.3389/fmicb.2019.00302
Chen K, Tian Z, He H, Long C, Jiang F (2020) Bacillus species as potential biocontrol agents against citrus diseases. Biol Control. https://doi.org/10.1016/j.biocontrol.2020.104419
Clarke KR, Ainsworth M (1993) A method of linking multivariate community structure to environmental variables. Mar Ecol Prog Ser 92:205–205
Compant S, Clément C, Sessitsch A (2010) Plant growth-promoting bacteria in the rhizo- and endosphere of plants: their role, colonization, mechanisms involved and prospects for utilization. Soil Biol Biochem 42(5):669–678. https://doi.org/10.1016/j.soilbio.2009.11.024
Coombs JT, Franco CMM (2003) Isolation and identification of Actinobacteria from surface-sterilized wheat roots. Appl Environ Microbiol 69(9):5603–5608. https://doi.org/10.1128/AEM.69.9.5603
Daniel J, Gleason JE, Cottenie K, Rooney RC (2019) Stochastic and deterministic processes drive wetland community assembly across a gradient of environmental filtering. Oikos. https://doi.org/10.1111/oik.05987
Dastogeer KMG, Tumpa FH, Sultana A, Akter MA, Chakraborty A (2020) Plant microbiome–an account of the factors that shape community composition and diversity. Curr Plant Biol. https://doi.org/10.1016/j.cpb.2020.100161
Dini-Andreote F, Stegen JC, van Elsas JD, Salles JF (2015) Disentangling mechanisms that mediate the balance between stochastic and deterministic processes in microbial succession. Proc Natl Acad Sci 112(11):E1326–E1332. https://doi.org/10.1073/pnas.1414261112
Dong C, Wang L, Li Q, Shang Q (2019) Bacterial communities in the rhizosphere, phyllosphere and endosphere of tomato plants. PLoS ONE 14(11):1–17. https://doi.org/10.1371/journal.pone.0223847
El-deeb B, Fayez K, Gherbawy Y (2012) Isolation and characterization of endophytic bacteria from Plectranthus tenuiflorus medicinal plant in Saudi Arabia desert and their antimicrobial activities. J Plant Interact. https://doi.org/10.1080/17429145.2012.680077
Emami S, Alikhani HA, Pourbabaei AA, Etesami H, Sarmadian F, Motessharezadeh B (2019) Effect of rhizospheric and endophytic bacteria with multiple plant growth promoting traits on wheat growth. Environ Sci Pollut Res 26(19):19804–19813. https://doi.org/10.1007/s11356-019-05284-x
Esty WW (1986) The efficiency of Good's nonparametric coverage estimator. Ann Stat 1257–1260
Frank A, Saldierna Guzmán J, Shay J (2017) Transmission of bacterial endophytes. Microorganisms 5(4):70. https://doi.org/10.3390/microorganisms5040070
Guo Y, Hou L, Zhang Z, Zhang J, Cheng J, Wei G, Lin Y (2019) Soil microbial diversity during 30 years of grassland restoration on the Loess Plateau, China: tight linkages with plant diversity. Land Degrad Dev 30(10):1172–1182. https://doi.org/10.1002/ldr.3300
Haruna E, Zin NM, Adams JM (2017) Draft genome sequence of an endophytic bacterium, Paenibacillus tyrfis strain SUK123, isolated from Santiria apiculata stem. Genom Data 14:44–46. https://doi.org/10.1016/j.gdata.2017.08.005
Haruna E, Zin NM, Kerfahi D, Adams JM (2018a) Extensive overlap of tropical rainforest bacterial endophytes between soil, plant parts, and plant species. Microb Ecol. https://doi.org/10.1007/s00248-017-1002-2
Haruna E, Zin NM, Kerfahi D, Adams JM (2018b) Extensive overlap of tropical rainforest bacterial endophytes between soil, plant parts, and plant species. Microb Ecol 75(1):88–103. https://doi.org/10.1007/s00248-017-1002-2
He R, Zeng J, Zhao D, Huang R, Yu Z, Wu QL (2020) Contrasting patterns in diversity and community assembly of Phragmites australis root-associated bacterial communities from different seasons. Appl Environ Microbiol. https://doi.org/10.1128/AEM.00379-20
Horner-Devine MC, Bohannan BJM (2006) Phylogenetic clustering and overdispersion in bacterial communities. Ecology 87(7 SUPPL.):S100–S108. https://doi.org/10.1890/0012-9658(2006)87[100:PCAOIB]2.0.CO;2
Hu W, Zhang Q, Tian T, Li D, Cheng G, Mu J, Wu Q (2015) Relative roles of deterministic and stochastic processes in driving the vertical distribution of bacterial communities in a permafrost core from the Qinghai-Tibet. PLoS ONE 10(12):e0145747. https://doi.org/10.1371/journal.pone.0145747
Huse SM, Welch DM, Morrison HG, Sogin ML (2010) Ironing out the wrinkles in the rare biosphere through improved OTU clustering. Environ Microbiol 12(7):1889–1898. https://doi.org/10.1111/j.1462-2920.2010.02193.x
Izzah N, Sarmin M, Yuan G, Tan A, Franco CMM, Edrada-ebel R et al (2013) Streptomyces kebangsaanensis sp. Nov. an endophytic actinomycete isolated from an ethnomedicinal plant, which produces phenazine-1-carboxylic acid. Int J Syst Evol Microbiol 3:3738. https://doi.org/10.1099/ijs.0.047878-0
Jackson CR, Randolph KC, Osborn SL, Tyler HL (2013) Culture dependent and independent analysis of bacterial communities associated with commercial salad leaf vegetables. BMC Microbiol 13(1):274. https://doi.org/10.1186/1471-2180-13-274
Jin H, Yang X-Y, Yan Z-Q, Liu Q, Li X-Z, Chen J-X et al (2014) Characterization of rhizosphere and endophytic bacterial communities from leaves, stems and roots of medicinal Stellera chamaejasme L. Syst Appl Microbiol 37(5):376–385. https://doi.org/10.1016/j.syapm.2014.05.001
Kerfahi D, Tripathi BM, Singh D, Kim H, Lee S, Lee J, Adams JM (2015) Effects of functionalized and raw multi-walled carbon nanotubes on soil bacterial community composition. PLoS ONE 10(3):e0123042. https://doi.org/10.1371/journal.pone.0123042
Kim BK, Lim Y-W, Kim M, Kim S, Chun J, Lee J-H, Jung Y (2007) EzTaxon: a web-based tool for the identification of prokaryotes based on 16S ribosomal RNA gene sequences. Int J Syst Evol Microbiol 57(10):2259–2261. https://doi.org/10.1099/ijs.0.64915-0
Köberl M, Schmidt R, Ramadan EM, Bauer R, Berg G (2013) The microbiome of medicinal plants: diversity and importance for plant growth, quality, and health. Front Microbiol 4:1–54. https://doi.org/10.3389/fmicb.2013.00400
Kogel K-H, Franken P, Hückelhoven R (2006) Endophyte or parasite—what decides? Curr Opin Plant Biol 9(4):358–363. https://doi.org/10.1016/j.pbi.2006.05.001
Kumar A, Droby S, Singh VK, Singh SK, White JF (2020) Entry, colonization, and distribution of endophytic microorganisms in plants. In: Microbial Endophytes. Woodhead Publishing, pp 1–33
Kusari S, Singh S, Jayabaskaran C (2014) Biotechnological potential of plant-associated endophytic fungi: hope versus hype. Trends Biotechnol 32(6):297–303. https://doi.org/10.1016/j.tibtech.2014.03.009
Kuźniar A, Włodarczyk K, Wolińska A (2019) Agricultural and other biotechnological applications resulting from trophic plant-endophyte interactions. Agronomy 9(12):779. https://doi.org/10.3390/agronomy9120779
Laforest-Lapointe I, Messier C, Kembel SW, Lindow S, Brandl M, Herre E et al (2016) Host species identity, site and time drive temperate tree phyllosphere bacterial community structure. Microbiome 4(1):27. https://doi.org/10.1186/s40168-016-0174-1
Li HY, Shen M, Zhou ZP, Li T, Wei YL, Lin LB (2012) Diversity and cold adaptation of endophytic fungi from five dominant plant species collected from the Baima Snow Mountain, Southwest China. Fungal Divers 54:79–86. https://doi.org/10.1007/s13225-012-0153-1
Liotti RG, da Silva Figueiredo MI, da Silva GF, de Mendonça EAF, Soares MA (2018) Diversity of cultivable bacterial endophytes in Paullinia cupana and their potential for plant growth promotion and phytopathogen control. Microbiol Res 207:8–18. https://doi.org/10.1016/j.micres.2017.10.011
Liu H, Carvalhais LC, Crawford M, Singh E, Dennis PG, Pieterse CMJ, Schenk PM (2017) Inner plant values: diversity, colonization and benefits from endophytic bacteria. Front Microbiol. https://doi.org/10.3389/fmicb.2017.02552
Lopes R, Tsui S, Gonçalves PJRO, de Queiroz MV (2018) A look into a multifunctional toolbox: endophytic Bacillus species provide broad and underexploited benefits for plants. World J Microbiol Biotechnol 10:10. https://doi.org/10.1007/s11274-018-2479-7
Masella AP, Bartram AK, Truszkowski JM, Brown DG, Neufeld JD (2012) PANDAseq: paired-end assembler for illumina sequences. BMC Bioinform 13(1):31. https://doi.org/10.1186/1471-2105-13-31
Matthews A, Pierce S, Hipperson H, Raymond B (2019) Rhizobacterial community assembly patterns vary between crop species. Front Microbiol 10:1–13. https://doi.org/10.3389/fmicb.2019.00581
McElrone AJ, Sherald JL, Forseth IN (2003) Interactive effects of water stress and xylem-limited bacterial infection on the water relations of a host vine. J Exp Bot 54(381):419–430. https://doi.org/10.1093/jxb/erg046
Miyambo T, Makhalanyane TP, Cowan DA, Valverde A (2016) Plants of the fynbos biome harbour host species-specific bacterial communities. FEMS Microbiol Lett 363(15):fnw22. https://doi.org/10.1093/femsle/fnw122
Morella NM, Weng FCH, Joubert PM, Jessica C, Lindow S, Koskella B (2020) Successive passaging of a plant-associated microbiome reveals robust habitat and host genotype-dependent selection. Proc Natl Acad Sci USA 117(2):1148–1159. https://doi.org/10.1073/pnas.1908600116
Mousa WK, Raizada MN (2013) The diversity of anti-microbial secondary metabolites produced by fungal endophytes: an interdisciplinary perspective. Front Microbiol 4:65. https://doi.org/10.3389/fmicb.2013.00065
Munir S, Li Y, He P, Huang M, He P, He P et al (2020) Core endophyte communities of different citrus varieties from citrus growing regions in China. Sci Rep 10(1):1–12. https://doi.org/10.1038/s41598-020-60350-6
Naik S, Shaanker RU, Ravikanth G, Dayanandan S (2019) How and why do endophytes produce plant secondary metabolites? Symbiosis 78(3):193–201. https://doi.org/10.1007/s13199-019-00614-6
Newton AC, Fitt BDL, Atkins SD, Walters DR, Daniell TJ, Huang YJ et al (2010) Pathogenesis, parasitism and mutualism in the trophic space of microbe-plant interactions. Trends Microbiol 18(8):365–373. https://doi.org/10.1016/j.tim.2010.06.002
Ogbe AA, Finnie JF, Van Staden J (2020) The role of endophytes in secondary metabolites accumulation in medicinal plants under abiotic stress. S Afr J Bot 134:126–134. https://doi.org/10.1016/j.sajb.2020.06.023
Pinto-Carbó M, Gademann K, Eberl L, Carlier A (2018) Leaf nodule symbiosis: function and transmission of obligate bacterial endophytes. Curr Opin Plant Biol 21:10. https://doi.org/10.1016/j.pbi.2018.01.001
Qin S, Chen HH, Zhao GZ, Li J, Zhu WY, Xu LH et al (2012) Abundant and diverse endophytic actinobacteria associated with medicinal plant Maytenus austroyunnanensis in Xishuangbanna tropical rainforest revealed by culture-dependent and culture-independent methods. Environ Microbiol Rep 4(5):522–531. https://doi.org/10.1111/j.1758-2229.2012.00357.x
Rajasekaran D, Palombo EA, Yeo TC, Ley DLS, Tu CL, Malherbe F, Grollo L (2013) Identification of traditional medicinal plant extracts with novel anti-influenza activity. PLoS ONE 8(11):1–15. https://doi.org/10.1371/journal.pone.0079293
Rana KL, Kour D, Kaur T, Sheikh I, Yadav AN, Kumar V et al (2020) Endophytic microbes from diverse wheat genotypes and their potential biotechnological applications in plant growth promotion and nutrient uptake. Proc Nat Aca Sci India Sect B Biol Sci 90(5):969–979. https://doi.org/10.1007/s40011-020-01168-0
Rice LB (2010) Progress and challenges in implementing the research on ESKAPE pathogens. Infect Control Hosp Epidemiol 31(S1):S7–S10. https://doi.org/10.1086/655995
Rodríguez-Gironés M, Santamaría L (2006) A new algorithm to calculate the nestedness temperature of presence–absence matrices. J Biogeogr 33(5):924–935. https://doi.org/10.1111/j.1365-2699.2006.01444.x
Sankaranarayanan K, Timofeeff MN, Spathis R, Lowenstein TK, Lum JK (2011) Ancient microbes from halite fluid inclusions: optimized surface sterilization and DNA extraction. PLoS ONE 6(6):1–8. https://doi.org/10.1371/journal.pone.0020683
Sarmin NIM, Zin NM, Tien NK, Sidik NM, Kaewkla O, Franco CM (2012) Ethnomedicinal plants as host of bioactive endophytic actinomycetes. Sains Malays 41(5):547–551
Saxena AK, Kumar M, Chakdar H, Anuroopa N, Bagyaraj DJ (2020) Bacillus species in soil as a natural resource for plant health and nutrition. J Appl Microbiol 128(6):1583–1594. https://doi.org/10.1111/jam.14506
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB et al (2009) Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75(23):7537–7541. https://doi.org/10.1128/AEM.01541-09
Schloss PD, Gevers D, Westcott SL (2011) Reducing the effects of PCR amplification and sequencing artifacts on 16s rRNA-based studies. PLoS ONE. https://doi.org/10.1371/journal.pone.0027310
Sessitsch A, Hardoim P, Döring J, Weilharter A, Krause A, Woyke T et al (2012) functional characteristics of an endophyte community colonizing rice roots as revealed by metagenomic analysis. Mol Plant Microbe Interact 25(1):28–36. https://doi.org/10.1094/MPMI-08-11-0204
Sharma A, Malhotra B, Kharkwal H, Kulkarni GT, Kaushik N (2020a) Therapeutic agents from endophytes harbored in Asian medicinal plants. Phytochem Rev. https://doi.org/10.1007/s11101-020-09683-8
Sharma S, Rani V, Saini R, Verma ML (2020b) Bioprospecting and biotechnological applications of microbial endophytes. Springer, Singapore, pp 191–228. https://doi.org/10.1007/978-981-15-2679-4_7
Stegen JC, Lin X, Konopka AE, Fredrickson JK (2012) Stochastic and deterministic assembly processes in subsurface microbial communities. ISME J 622(10):1653–1664. https://doi.org/10.1038/ismej.2012.22
Stein T (2005) Bacillus subtilis antibiotics: structures, syntheses and specific functions. Mol Microbiol 56(4):845–857. https://doi.org/10.1111/j.1365-2958.2005.04587.x
Strobel G, Daisy B (2003) Bioprospecting for microbial endophytes and their natural products. Microbiol Mol Biol Rev. https://doi.org/10.1128/MMBR.67.4.491-502.2003
Suryanarayanan TS (2013) Endophyte research: going beyond isolation and metabolite documentation. Fungal Ecol 6(6):561–568. https://doi.org/10.1016/j.funeco.2013.09.007
Tian B-Y, Cao Y, Zhang K-Q, Gaiero JR, Berg G, Hallmann J et al (2015) Metagenomic insights into communities, functions of endophytes, and their associates with infection by root-knot nematode, Meloidogyne incognita, in tomato roots. Sci Rep 5:17087. https://doi.org/10.1038/srep17087
Turner TR, James EK, Poole PS (2013) The plant microbiome. Genome Biol 14(6):209. https://doi.org/10.1186/gb-2013-14-6-209
Ullah A, Nisar M, Ali H, Hazrat A, Hayat K, Keerio AA et al (2019) Drought tolerance improvement in plants: an endophytic bacterial approach. Appl Microbiol Biotechnol. https://doi.org/10.1007/s00253-019-10045-4
van der Lelie D, Taghavi S, Monchy S, Schwender J, Miller L, Ferrieri R et al (2009) Poplar and its bacterial endophytes: coexistence and harmony. Crit Rev Plant Sci 28(5):346–358. https://doi.org/10.1080/07352680903241204
Vengatesh PK, Joe SS, Ramalashmi CK (2018) A potential surface sterilization technique and culture media for the isolation of endophytic bacteria from Acalypha indica and its antibacterial activity. J Med Plants Stud 6(1):181–184
Wani ZA, Ashraf N, Mohiuddin T, Riyaz-Ul-Hassan S (2015) Plant-endophyte symbiosis, an ecological perspective. Appl Microbiol Biotechnol 99(7):2955–2965. https://doi.org/10.1007/s00253-015-6487-3
White JF, Torres MS, Sullivan RF, Jabbour RE, Chen Q, Tadych M et al (2014) Microscopy research and technique: occurrence of Bacillus amyloliquefaciens as a systemic endophyte of vanilla orchids. Microsc Res Tech 11(77):875–885. https://doi.org/10.1002/jemt.22410
Yadav AN (2018) Biodiversity and biotechnological applications of host-specific endophytic fungi for sustainable agriculture and allied sectors. Acta Sci Microbiol 1(5):1–5. https://doi.org/10.31080/ASMI.2018.01.0044
Zhang X, Johnston ER, Liu W, Li L, Han X (2016) Environmental changes affect the assembly of soil bacterial community primarily by mediating stochastic processes. Glob Change Biol 22(1):198–207. https://doi.org/10.1111/gcb.13080
Zhang J, Zhang C, Yang J, Zhang R, Gao J, Zhao X et al (2019) Insights into endophytic bacterial community structures of seeds among various Oryza sativa L. rice genotypes. J Plant Growth Regul 38(1):93–102. https://doi.org/10.1007/s00344-018-9812-0
Zhou H-W, Li D-F, Tam NF-Y, Jiang X-T, Zhang H, Sheng H-F et al (2011) BIPES, a cost-effective high-throughput method for assessing microbial diversity. ISME J 5(4):741–749. https://doi.org/10.1038/ismej.2010.160
Zin NM, Sarmin NIM, Ghadin N, Basri DF, Sidik NM, Hess WM, Strobel GA (2007) Bioactive endophytic streptomycetes from the Malay Peninsula. FEMS Microbiol Lett 274(1):83–88. https://doi.org/10.1111/j.1574-6968.2007.00819.x
Zuo J, Zu M, Liu L, Song X, Yuan Y (2021) Composition and diversity of bacterial communities in the rhizosphere of the Chinese medicinal herb Dendrobium. BMC Plant Biol 21(1):127. https://doi.org/10.1186/s12870-021-02893-y
Acknowledgements
We are grateful to the University Kebangsaan University Forest Reserve and Herbarium staff. We thank Muhanna Al-Shabanni, Aishah Ismail, Radhiah Binti Khairon, and Nur Faizah Abu Bakar, for their assistance during sampling. This work was funded by the Universiti Kebangsaan Malaysia Research Grant (GUP 2015-042). E.H, N.M.Z and J.M.A design the work, E.H did the sampling, microbial DNA extraction. E.H, H.C, J.A.M analysed the work, manuscript written by all authors. Emmanuel Haruna was a Ph.D. scholar at UKM sponsored by the Tertiary Education TrustFund (TETFUND), Nigeria.
Funding
This work was funded by the Universiti Kebangsaan Malaysia Research Grant (GUP 2015–042).
Author information
Authors and Affiliations
Contributions
EH, NMZ and JA conceived the research questions and designed the project. The field sampling was done by EH, NMZ and JA. EH performed the lab experiments. EH and HC processed the sequences at different stages. EH, HC, JA did all the data analysis. EH, NMZ, and JA prepared all the draft versions and wrote the main manuscript text. HC contributed to preparing the draft and advised on the work. All of the authors reviewed, contributed to revisions and accepted the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Haruna, E., Zin, N.M., Cho, H. et al. Alternate states dominate the bacterial endophyte community of a tropical tree. Environmental Sustainability 4, 785–796 (2021). https://doi.org/10.1007/s42398-021-00198-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42398-021-00198-2