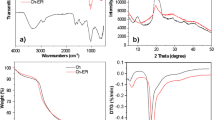
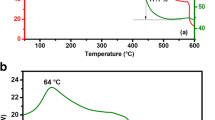
Abstract
The increase of pollution in aquifers by heavy metals in Mexico, because of industrialization and urbanization, have had negative effects for the environment and public health. This work aimed to evaluate the kinetic and equilibrium studies of Cr(VI) adsorption using glutaraldehyde-crosslinked chitosan beads in aqueous systems. Glutaraldehyde-crosslinked chitosan beads were prepared, and three different masses (3, 6, 9 g) were evaluated with 5 concentrations of Cr (VI) (20, 30, 50, 80, 100 mg/L). The adsorption mechanisms were studied through the adsorption isotherms of Langmuir and Freundlich, as well as the adsorption kinetics of pseudo-first order, pseudo-second order, and Elovich based on experimental data. Results showed that after 10 min. of exposure, the beads adsorbed more than 50% of the initial concentration of Cr(VI), and after 90 min. it was possible to remove up to 100% of Cr(VI) contained in the water. The adsorption kinetics of pseudo-second order and Elovich showed the best fits compared to pseudo-first order. Based on these results, glutaraldehyde-crosslinked chitosan beads could be proposed as a good alternative for the removal Cr(VI) from aqueous systems.








Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
References
Acosta-Arguello HA, Barraza-Yance CA, Albis-Arrieta AR (2017) Adsorción de cromo (VI) utilizando cáscara de yuca (Manihot esculenta) como bioadsorbente. Estudio Cinético Ingeniería y Desarrollo 35:8–76. https://doi.org/10.14482/inde.35.1.8943
Acosta-Ferreira S, Castillo OS, Madera-Santana JT, Mendoza-García DA, Núñez-Colín CA, Grijalva-Verdugo CG, Villa-Lerma AG, Morales-Vargas AT, Rodríguez-Núñez JR (2020) Production and physicochemical characterization of chitosan for the harvesting of wild microalgae consortia. Biotechnol Rep 28:e00554. https://doi.org/10.1016/j.btre.2020.e00554
Achal V, Pan X, Lee D-J, Kumari D, Zhang D (2013) Remediation of Cr(VI) from chromium slag by biocementation. Chemosphere 93:1352–1358. https://doi.org/10.1016/j.chemosphere.2013.08.008
Ainane T, Abourriche A, Kabbaj M, Elkouali M, Bennamara A, Charrauf M, Talbi M (2014) Removal of hexavalent chromium from aqueous solution by raw and chemically modified seaweed Bifurcaria bifurcata. J Mater Environ Sci 5:975–982
Altun T, Pehlivan E (2012) Removal of Cr(VI) from aqueous solutions by modified walnut shells. Food Chem 132:693–700. https://doi.org/10.1016/j.foodchem.2011.10.099
Barzchi S, Bahram M, Nouri S (2018) Equilibrium and kinetic studies on the removal of acid red-14 from aqueous solutions using PSMA. Iran J Sci Technol Trans Sci 42(1):203–208. https://doi.org/10.1007/s40995-018-0489-9
Bhatt R, Sreedhar B, Padmaja P (2015) Adsorption of chromium from aqueous solutions using crosslinked chitosan–diethylenetriaminepentaacetic acid. Int J Biol Macromol 74:458–466. https://doi.org/10.1016/j.ijbiomac.2014.12.041
Chatterjee S, Hadi A, Price B (2000) Simple linear regression. In regression analysis by example, Wiley (ed.), 5th edn, New York, USA, pp. 25–49
Chien SH, Clayton WR (1980) Application of Elovich equation to the kinetics of phosphate release and sorption in soils. Soil Sci Soc Am J 44:265–268. https://doi.org/10.2136/sssaj1980.03615995004400020013x
Correa-Murrieta MA, López-Cervantes J, Sánchez-Machado DI, Sánchez-Duarte RG, Rodríguez-Núñez JR, Núñez-Gastélum JA (2012) Fe(II) and Fe(III) adsorption by chitosan-tripolyphosphate beads: kinetic and equilibrium studies. J Water Supply Res Technol AQUA 61:331–341. https://doi.org/10.2166/aqua.2012.048
Dhal B, Abhilash DBP (2018) Mechanism elucidation and adsorbent characterization for removal of Cr(VI) by native fungal adsorbent. Sustain Environ Res 28:289–297. https://doi.org/10.1016/j.serj.2018.05.002
Díaz C, Jacinto C, Medina R, Navarro A, Cuizano N, Llanos B (2013) Estudio de la biosorción de Cr(VI) con quitosano cuaternario reticulado para su aplicación en biorremediación de aguas contaminadas. Rev Soc Quím Perú 79:304–308
Díaz de Apodaca DED, Villarán VMC, Río PF, Ramírez LA, Lorenzo IL (2007) Utilización de adsorbentes basados en quitosano y alginato sódico para la eliminación de iones metálicos: Cu2+, Pb2+, Cr3+ y Co2+. Rev Iberoam Polim 8:20–37
Dima JB, Sequeiros C, Zaritzky NE (2015) Hexavalent chromium removal in contaminated water using reticulated chitosan micro/nanoparticles from seafood processing wastes. Chemosphere 141:100–111. https://doi.org/10.1016/j.chemosphere.2015.06.030
Ho YS, Mckay G, Wase DAJ, Foster CF (2000) Study of the sorption of divalent metal ions onto peat. Adsorpt Sci Technol 18:639–650. https://doi.org/10.1260/0263617001493693
Hoffman B, Seitz D, Mencke A, Kokot A, Ziegler G (2009) Glutaraldehyde and oxidised dextran as crosslinker reagents for chitosan-based scaffolds for cartilage tissue engineering. J Mater Sci: Mater Med 20:1495–1503. https://doi.org/10.1007/s10856-009-3707-3
Huang C, Chen S, Pan JR (2000) Optimal condition for modification of chitosan: a biopolymer for coagulation of colloidal particles. Water Res 34:1057–1062. https://doi.org/10.1016/S0043-1354(99)00211-0
Kahu SS, Shekhawat A, Saravanan D, Jugade RM (2016) Two fold modified chitosan for enhanced adsorption of hexavalent chromium from simulated wastewater and industrial effluents. Carbohydr Polym 146:264–273. https://doi.org/10.1016/j.carbpol.2016.03.041
Karthik R, Meenakshi S (2014) Facile synthesis of cross linked-chitosan–grafted-polyaniline composite and its Cr(VI) uptake studies. Int J Biol Macromol 67:210–219. https://doi.org/10.1016/j.ijbiomac.2014.03.035
Kaya K, Pehlivan E, Schmit C, Bahadir M (2014) Use of modified wheat bran for the removal of chromium(VI) from aqueous solutions. Food Chem 158:112–117. https://doi.org/10.1016/j.foodchem.2014.02.107
Khalil TE, Elhusseiny AF, Ibrahim NM, El-dissouky (2021) Unexpected effect of magnetic nanoparticles on the performance of aqueous removal of toxic Cr(VI) using modified biopolymer chitosan. Int J Biol Macromol 170:768–779. https://doi.org/10.1016/j.ijbiomac.2020.12.188
Lagergren S (1898) About the theory of so-called adsorption of soluble substances. K Sven Vetenskapsaked Handlingar. Int J Environ Bioremediat Biodegrad 24:1–39. https://doi.org/10.12691/ijebb-4-2-4
Lu J, Xu K, Yang J, Hao Y, Cheng F (2017) Nano iron oxide impregnated in chitosan bead as a highly efficient sorbent for Cr(VI) removal from water. Carbohydr Polym 173:28–36. https://doi.org/10.1016/j.carbpol.2017.05.070
Madera-Santana TJ, Herrera-Méndez CH, Rodríguez-Núñez JR (2018) An overview of the chemical modifications of chitosan and their advantages. Green Mater 6:131–142. https://doi.org/10.1680/jgrma.18.00053
Madrid GL, Gracia LDC, Marrugo JL, Urango ID (2011) Genotoxicidad de metales pesados (Hg, Zn, Cu, Pb Y Cd) asociado a explotaciones mineras en pobladores de la cuenca del río San Jorge del departamento de Córdoba, Colombia. ACCB 23:103–111
Moore D, McCabe G (2003) Introduction to the Practice of Statistics. WH Freeman and Company, London
Nthumbi RM, Ngila JC, Moodley B, Kindness A, Petrik L (2012) Application of chitosan/polyacrylamide nanofibres for removal of chromate and phosphate in water. Phys Chem Earth, Parts a/b/c 50–52:243–251. https://doi.org/10.1016/j.pce.2012.07.001
Núñez-Gastélum JA, Rodríguez-Núñez JR, de la Rosa LA, Días-Sánchez AG, Alvarez-Parrilla E, Martínez-Martínez A, Villa-Lerma A (2019a) Análisis de propiedades físicas y estructurales de las películas de quitosano-policaprolactona adicionadas con extractos de hojas de Moringa oleífera. Rev Mex Ing Quim 18:99–105
Núñez-Gastélum JA, Rodríguez-Núñez JR, de la Rosa LA, Díaz-Sánchez AG, Alvarez-Parrilla E, Martínez-Martínez A, Villa-Lerma G (2019b) Screening of the physical and structural properties of chitosan-polycaprolactone films added with Moringa oleifera leaf extract. Rev Mex Ing Quim 18(99):105
Omer AM, Khalifa RE, Hu Z, Zhang H, Liu C, Ouyang XK (2019) Fabrication of tetraethylenepentamine functionalized alginate beads for adsorptive removal of Cr(VI) from aqueous solutions. Int J Biol Macromol 125:1221–1231. https://doi.org/10.1016/j.ijbiomac.2018.09.097
Pinzón-Bedolla ML, Vera-Villamizar LE (2009) Modelamiento de la cinética de bioadsorción de Cr(III) usando cáscara de naranja. Dyna 76:95–106
Reyes-Gutiérrez LR, Romero-Guzmán ET, Olmos-Salinas MG, Rodríguez-Castillo R (2009) Chemical species of chromatite of an industrial landfill in the León valley, Guanajuato, México. Rev Mex De Cienc Geol 26:104–116
Rodríguez A, Pacheco N, Cárdenas J, Tovar J, Martínez VM, Acosta I (2017) Bioadsorción de cromo (VI) en solución acuosa por la biomasa de amaranto (Amaranthus caudatus). Av Cienc in 8:11–20
Rodríguez-Núñez JR, Madera-Santana TJ, Sánchez-Machado DI, López-Cervantes J, Soto VH (2014) Chitosan/hydrophilic plasticizer-based films: preparation, physicochemical and antimicrobial properties. J Polym Environ 22:41–51. https://doi.org/10.1007/s10924-013-0621-z
Sánchez-Duarte RG, López-Cervantes J, Sánchez-Machado DI, Correa-Murrieta MA, Núñez-Gastélum JA, Rodríguez-Núñez JR (2016) Chitosan-based adsorbents gels for the removal of tris-azo dye: isotherms and kinetics studies. Environ Eng Manag J 15:2469–2478. https://doi.org/10.1002/apj.1841
SEMARNAT, México. NOM-147-SEMARNAT/SSA1–2004 (2006). Que establece criterios para determinar las concentraciones de remediación de suelos contaminados por arsénico, bario, berilio, cadmio, cromo hexavalente, mercurio, níquel, plata, plomo, selenio, talio y/o vanadio. http://www.profepa.gob.mx/innovaportal/file/1392/1/nom-147-semarnat_ssa1-2004.pdf. (accessed 15/10/2019)
Soto EM, Nelsy PL (2017) Remoción de cromo hexavalente de aguas residuales con microorganismos adaptados a medios ricos en cromo. ACCB 29:49–57
Tagle ZD, Caldera OA, Rodríguez GJA (2017) Complejidad ambiental en el Bajío mexicano: implicaciones del proyecto civilizatorio vinculado al crecimiento económico. Reg Soc 68:193–221. https://doi.org/10.22198/rys.2017.68.a873
Tandon RK, Crisp PT, Ellis J, Baker RS (1984) Effect of pH on chromium(VI) species in solution. Talanta 31:227–228. https://doi.org/10.1016/0039-9140(84)80059-4
Tejada-Tovar C, Villabona-Ortiz A, Garcés-Jaraba L (2015) Adsorción de metales pesados en aguas residuales usando materiales de origen biológico. TecnoLógicas 18:109–123. https://doi.org/10.22430/22565337.209
Vakili M, Deng S, Li T, Wang W, Wang W, Yu G (2018) Novel crosslinked chitosan for enhanced adsorption of hexavalent chromium in acidic solution. Chem Eng J 347:782–790. https://doi.org/10.1016/j.cej.2018.04.181
Wang J, Chen C (2009) Biosorbents for heavy metals removal and their future. Biotechnol Adv 27:195–226. https://doi.org/10.1016/j.biotechadv.2008.11.002
Wang S, Qing M, Zhu ZH (2008) Characteristics of coal fly ash and adsorption application. Fuel 87:3469–3473. https://doi.org/10.1016/j.fuel.2008.05.022
Wu J, Zheng H, Zhang F, Jianxiong ZR, Xiang B (2019) Iron-carbon composite from carbonization of iron-crosslinked sodium alginate for Cr(VI) removal. Chem Eng J 365:21–29. https://doi.org/10.1016/j.cej.2019.01.009
Zeraatkar MA, Esmaeilkhanian E, Shakourian-Fard M (2019) Immobilizing magnetic glutaraldehyde cross-linked chitosan on graphene oxide and nitrogen-doped graphene oxide as well-dispersible adsorbents for chromate removal from aqueous solutions. Int J Biol Macromol 128:61–73. https://doi.org/10.1016/j.ijbiomac.2019.01.086
Zhang J, Hua P, Krebs P (2018) Influences of land use and antecedent dry-weather period on pollution level and ecological risk of heavy metals in road-deposited sediment. Environ Pollut 228:158–168. https://doi.org/10.1016/j.envpol.2017.05.029
Zhang W, Wang H, Hu X, Feng H, Xiong W, Guo W, Zhou J, Mosa A, Peng Y (2019) Multicavity triethylenetetramine-chitosan/alginate composite beads for enhanced Cr(VI) removal. J Clean Prod 231:733–745
Acknowledgements
The authors acknowledge the financial support from the grants CB 2015-252007-Z provided by the National Council of Science and Technology Mexico (CONACYT) and Cinvestav-LANNBIO for the support provided with the instrumental equipment.
Author Contribution’s
Sandra Torres-Badajoz and Eduardo López Ramírez: formally analyzed and investigated the study, Rodríguez-Núñez J. Rubén: helped in conceptualization, project administration, funding acquisition, roles/writing—original draft, Vicente Peña Caballero: wrote and edited the review and curated the data, Villa-Lerma Alma G.: wrote and edited the review and English language review, Madera-Santana J. Tomás: wrote and edited the review and helped in resources, María Guadalupe Rodríguez Carrillo: formally analyzed the study, and Castillo Omar S: conceptualized and supervised the study.
Funding
This project was supported by National Council of Science and Technology Mexico (CONACYT), CB 2015–252007-Z.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that there are no conflicts of interest.
Informed Consent Statement
All authors agree that this manuscript is submitted to the “Iranian Journal of Science and Technology, Transactions A: Science.”
Ethical Approval
All authors have been personally and actively involved in substantial work leading to the paper and will take public responsibility for its content. Also, this manuscript is not currently being considered for publication elsewhere.
Rights and permissions
About this article
Cite this article
Torres-Badajoz, S.G., Rodríguez-Núñez, J.R., López-Ramírez, E. et al. Kinetic and Equilibrium Studies of Cr(VI) Adsorption Using Glutaraldehyde-Crosslinked Chitosan Beads. Iran J Sci Technol Trans Sci 46, 147–160 (2022). https://doi.org/10.1007/s40995-021-01255-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40995-021-01255-5