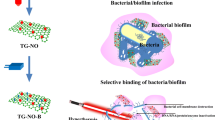
Abstract
Bacterial infections are grave threats to human health, particularly those caused by the most common Gram-positive bacteria. The massive administration of broad-spectrum antibiotics to treat various bacterial infections has led to the evolution and spread of drug resistance. As a universal antimicrobial technique unapt to induce drug resistance, photothermal therapy (PTT) is attracting extensive attention in recent years. However, its unspecific killing capability and side effects towards adjacent mammalian cells severely impede the practical applications. Herein, we proposed a metabolic engineering strategy to selectively inactivate Gram-positive bacteria by PTT. A bioorthogonal photothermal agent was prepared by the conjugation of IR-780 iodide and dibenzo-cyclooctyne (IR780-DBCO). Upon pre-metabolizing with 3-azido-D-alanine, Gram-positive bacteria rather than Gram-negative ones, such as Staphylococcus aureus and vancomycin-resistant Enterococcus faecalis (VRE), could be specifically tied up by the explosive IR780-DBCO via copper-free click chemistry. Thereafter, they spontaneously detonated under 15 min near-infrared light irradiation and inactivated nearly 100% Gram-positive bacteria in vitro. Moreover, superbug VRE-induced infection was significantly inhibited by this approach in a mouse skin wound model. This metabolic labelling-based photothermal ablation strategy specific to Gram-positive microbes would stimulate the development of precise anti-bacterial candidates for preclinical applications.
摘要
细菌感染, 尤其是最常见的革兰氏阳性菌引起的感染, 严重威胁着人类的生命健康. 抗生素是治疗细菌感染的首选药物, 但是它的大量使用造成了耐药性细菌的快速滋生和蔓延. 近年来, 光热抗菌疗法作为一种不易引起细菌耐药性的技术受到了广泛的关注. 然而, 非特异性的细菌杀伤和对邻近哺乳动物细胞的毒副作用严重限制了光热抗菌疗法的实际应用. 本文开发了一种代谢标记策略用于革兰氏阳性菌的选择性光热杀灭. 通过二苯并环辛炔(DBCO)和IR-780碘化物的共价连接,制备了生物正交的光热试剂IR780-DBCO. 革兰氏阳性菌或阴性菌经过3-叠氮基-D-丙氨酸预处理, 利用无铜点击化学反应实现了IR780-DBCO在金黄色葡萄球菌和耐万古霉素肠球菌等革兰氏阳性菌表面的选择性标记. 在15 min的近红外光照射下, 接近100%的革兰氏阳性菌被有效地杀灭. 此外, 该方法在小鼠皮肤伤口感染模型中也能够显著地抑制耐万古霉素肠球菌引起的感染. 我们提出的基于代谢标记的特异性光热杀灭革兰氏阳性菌的策略将会促进预临床研究中精准抗菌试剂的研发.
Article PDF
Similar content being viewed by others
References
Casadevall A, Pirofski LA. Microbiology: Ditch the term pathogen. Nature, 2014, 516: 165–166
Rohr JR, Barrett CB, Civitello DJ, et al. Emerging human infectious diseases and the links to global food production. Nat Sustain, 2019, 2: 445–456
Menichetti F. Current and emerging serious Gram-positive infections. Clin Microbiol Infect, 2005, 11: 22–28
Chung HJ, Reiner T, Budin G, et al. Ubiquitous detection of Gram-positive bacteria with bioorthogonal magnetofluorescent nanoparticles. ACS Nano, 2011, 5: 8834–8841
Rhee C, Dantes R, Epstein L, et al. Incidence and trends of sepsis in US hospitals using clinical vs claims data, 2009–2014. J Am Med Assoc, 2017, 318: 1241–1249
Fleischmann C, Scherag A, Adhikari NKJ, et al. Assessment of global incidence and mortality of hospital-treated sepsis. Current estimates and limitations. Am J Respir Crit Care Med, 2016, 193: 259–272
Kollef MH. Broad-spectrum antimicrobials and the treatment of serious bacterial infections: Getting it right up front. Clin Infect Dis, 2008, 47: S3–S13
Chellat MF, Raguž L, Riedl R. Targeting antibiotic resistance. Angew Chem Int Ed, 2016, 55: 6600–6626
Wang S, Xu M, Huang K, et al. Biocompatible metal-free organic phosphorescent nanoparticles for efficiently multidrug-resistant bacteria eradication. Sci China Mater, 2020, 63: 316–324
Levin-Reisman I, Ronin I, Gefen O, et al. Antibiotic tolerance facilitates the evolution of resistance. Science, 2017, 355: 826–830
Ma YX, Wang CY, Li YY, et al. Considerations and caveats in combating ESKAPE pathogens against nosocomial infections. Adv Sci, 2020, 7: 1901872
Yu F, Chen C, Yang G, et al. An acid-triggered porphyrin-based block copolymer for enhanced photodynamic antibacterial efficacy. Sci China Chem, 2021, 64: 459–466
Ye X, Feng T, Li L, et al. Theranostic platforms for specific discrimination and selective killing of bacteria. Acta Biomater, 2021, 125: 29–40
Ray PC, Khan SA, Singh AK, et al. Nanomaterials for targeted detection and photothermal killing of bacteria. Chem Soc Rev, 2012, 41: 3193–3209
Hsiao CW, Chen HL, Liao ZX, et al. Effective photothermal killing of pathogenic bacteria by using spatially tunable colloidal gels with nanolocalized heating sources. Adv Funct Mater, 2015, 25: 721–728
Korupalli C, Huang CC, Lin WC, et al. Acidity-triggered charge-convertible nanoparticles that can cause bacterium-specific aggregation in situ to enhance photothermal ablation of focal infection. Biomaterials, 2017, 116: 1–9
Chen Q, He S, Zhang F, et al. A versatile Pt-Ce6 nanoplatform as catalase nanozyme and NIR-II photothermal agent for enhanced PDT/PTT tumor therapy. Sci China Mater, 2021, 64: 510–530
Xu JW, Yao K, Xu ZK. Nanomaterials with a photothermal effect for antibacterial activities: An overview. Nanoscale, 2019, 11: 8680–8691
Zhao Y, Dai X, Wei X, et al. Near-infrared light-activated thermo-sensitive liposomes as efficient agents for photothermal and antibiotic synergistic therapy of bacterial biofilm. ACS Appl Mater Interfaces, 2018, 10: 14426–14437
Lei W, Ren K, Chen T, et al. Polydopamine nanocoating for effective photothermal killing of bacteria and fungus upon near-infrared irradiation. Adv Mater Interfaces, 2016, 3: 1600767
Wang H, Li X, Tse BWC, et al. Indocyanine green-incorporating nanoparticles for cancer theranostics. Theranostics, 2018, 8: 1227–1242
Cai Y, Si W, Huang W, et al. Organic dye based nanoparticles for cancer phototheranostics. Small, 2018, 14: 1704247
Wang H, Tang L, Liu Y, et al. In vivo targeting of metabolically labeled cancers with ultra-small silica nanoconjugates. Theranostics, 2016, 6: 1467–1476
Yuan Y, Xu S, Cheng X, et al. Bioorthogonal turn-on probe based on aggregation-induced emission characteristics for cancer cell imaging and ablation. Angew Chem Int Ed, 2016, 55: 6457–6461
Sun T, Yu SH, Zhao P, et al. One-step selective exoenzymatic labeling (SEEL) strategy for the biotinylation and identification of glycoproteins of living cells. J Am Chem Soc, 2016, 138: 11575–11582
Wang W, Lin L, Du Y, et al. Assessing the viability of transplanted gut microbiota by sequential tagging with D-amino acid-based metabolic probes. Nat Commun, 2019, 10: 1317
Wang W, Yang Q, Du Y, et al. Metabolic labeling of peptidoglycan with NIR-II dye enables in vivo imaging of gut microbiota. Angew Chem Int Ed, 2020, 59: 2628–2633
Hudak JE, Alvarez D, Skelly A, et al. Illuminating vital surface molecules of symbionts in health and disease. Nat Microbiol, 2017, 2: 1–8
Mao D, Hu F, Kenry F, et al. Metal-organic-framework-assisted in vivo bacterial metabolic labeling and precise antibacterial therapy. Adv Mater, 2018, 30: 1706831
Hu F, Qi G, Kenry G, et al. Visualization and in situ ablation of intracellular bacterial pathogens through metabolic labeling. Angew Chem Int Ed, 2020, 59: 9288–9292
Wu M, Qi G, Liu X, et al. Bio-orthogonal AIEgen for specific discrimination and elimination of bacterial pathogens via metabolic engineering. Chem Mater, 2020, 32: 858–865
Liang H, DeMeester KE, Hou CW, et al. Metabolic labelling of the carbohydrate core in bacterial peptidoglycan and its applications. Nat Commun, 2017, 8: 15015
Dumont A, Malleron A, Awwad M, et al. Click-mediated labeling of bacterial membranes through metabolic modification of the lipopoly-saccharide inner core. Angew Chem Int Ed, 2012, 51: 3143–3146
Shieh P, Sloan Siegrist M, Cullen AJ, et al. Imaging bacterial peptidoglycan with near-infrared fluorogenic azide probes. Proc Natl Acad Sci USA, 2014, 111: 5456–5461
Yan F, Duan W, Li Y, et al. NIR-laser-controlled drug release from DOX/IR-780-loaded temperature-sensitive-liposomes for chemo-photothermal synergistic tumor therapy. Theranostics, 2016, 6: 2337–2351
Qing G, Zhao X, Gong N, et al. Thermo-responsive triple-function nanotransporter for efficient chemo-photothermal therapy of multi-drug-resistant bacterial infection. Nat Commun, 2019, 10: 4336
Kuang Y, Zhang K, Cao Y, et al. Hydrophobic IR-780 dye encapsulated in cRGD-conjugated solid lipid nanoparticles for NIR imaging-guided photothermal therapy. ACS Appl Mater Interfaces, 2017, 9: 12217–12226
Kuru E, Hughes HV, Brown PJ, et al. In situ probing of newly synthesized peptidoglycan in live bacteria with fluorescent D-amino acids. Angew Chem Int Ed, 2012, 51: 12519–12523
Wu F, Zheng H, Wang W, et al. Rapid eradication of antibiotic-resistant bacteria and biofilms by MXene and near-infrared light through photothermal ablation. Sci China Mater, 2021, 64: 748–758
Zhi Z, Su Y, Xi Y, et al. Dual-functional polyethylene glycol-b-polyhexanide surface coating with in vitro and in vivo antimicrobial and antifouling activities. ACS Appl Mater Interfaces, 2017, 9: 10383–10397
Yang C, Hu F, Zhang X, et al. Combating bacterial infection by in situ self-assembly of AIEgen-peptide conjugate. Biomaterials, 2020, 244: 119972
Zhu Y, Xu C, Zhang N, et al. Polycationic synergistic antibacterial agents with multiple functional components for efficient anti-infective therapy. Adv Funct Mater, 2018, 28: 1706709
Chen Z, Ji H, Liu C, et al. A multinuclear metal complex based DNasemimetic artificial enzyme: Matrix cleavage for combating bacterial biofilms. Angew Chem Int Ed, 2016, 55: 10732–10736
Niu J, Sun Y, Wang F, et al. Photomodulated nanozyme used for a Gram-selective antimicrobial. Chem Mater, 2018, 30: 7027–7033
Sun Y, Zhao C, Niu J, et al. Colorimetric band-aids for point-of-care sensing and treating bacterial infection. ACS Cent Sci, 2020, 6: 207–212
Sayed SM, Xu KF, Jia HR, et al. Naphthalimide-based multifunctional AIEgens: Selective, fast, and wash-free fluorescence tracking and identification of Gram-positive bacteria. Anal Chim Acta, 2021, 1146: 41–52
Yang J, Gao G, Zhang X, et al. One-step synthesis of carbon dots with bacterial contact-enhanced fluorescence emission: Fast Gram-type identification and selective Gram-positive bacterial inactivation. Carbon, 2019, 146: 827–839
Zhang X, Chen X, Yang J, et al. Quaternized silicon nanoparticles with polarity-sensitive fluorescence for selectively imaging and killing Grampositive bacteria. Adv Funct Mater, 2016, 26: 5958–5970
Acknowledgements
This work was supported by the National Natural Science Foundation of China (52003222 and 21875189), Ningbo Natural Science Foundation (202003N4064), the Natural Science Foundation of Chongqing (cstc2020jcyj-msxmX0752), the Joint Research Funds of Department of Science & Technology of Shaanxi Province and Northwestern Polytechnical University (2020GXLH-Z-013), and the Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Contributions
Feng T, Li P, and Huang W conceived the idea and supervised the study; Feng T, Ye X, and Lu H performed the experiments and wrote the manuscript; Nie C, Zhang J, Yu L, and Jin H participated in the in vivo anti-infective assay. All authors contributed to the general discussion.
Corresponding authors
Additional information
Tao Feng received her BSc and MSc degrees from Jilin University in 2010 and 2013, and PhD degree from Nanyang Technological University in 2018. She is currently an associate professor at Northwestern Polytechnical University. Her research focuses on the design of fluorescent probes for selective discrimination and accurate killing of bacteria.
Hui Lu received his BSc degree from Anhui University of Technology in 2019. Currently, he is a graduate student at the Frontiers Science Center for Flexible Electronics, Northwestern Polytechnical University. His research interest is developing new phototheranostic agents to selectively inactivate bacteria.
Xiaoting Ye received her BSc degree from Nanjing Tech University in 2018 and MSc degree from the Frontiers Science Center for Flexible Electronics, Northwestern Polytechnical University in 2021. Her research focuses on preparing novel photothermal materials for specific killing of bacteria.
Peng Li received his BSc from Tianjin University in 2006 and PhD degree from Nanyang Technological University in 2013. He has been a professor of the Frontiers Science Center for Flexible Electronics at Northwestern Polytechnical University since 2018. His research interest is developing innovative antibacterial materials and strategies for infectious diseases.
Wei Huang received his BSc degree from Peking University in 1983, followed by MSc and PhD degrees from the same university. Then he did his postdoctoral research at the National University of Singapore, where he participated in the foundation of the Institute of Materials Research and Engineering in 1995. In 2011, he was elected as academician of the Chinese Academy of Sciences. Now, he is the chief scientist of the Frontiers Science Center for Flexible Electronics at Northwestern Polytechnical University. His research interests include polymer sciences, bioelectronics, nanoelectronics, and organic/plastic/flexible electronics.
Supplementary information
Experimental details and supporting data are available in the online version of the paper.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Feng, T., Lu, H., Ye, X. et al. Selective inactivation of Gram-positive bacteria in vitro and in vivo through metabolic labelling. Sci. China Mater. 65, 237–245 (2022). https://doi.org/10.1007/s40843-021-1735-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-021-1735-0