Abstract
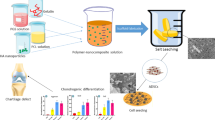
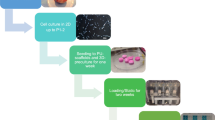
In this paper we report the differentiating properties of platelet-rich plasma releasates (PRPr) on human chondrocytes within elastomeric polycaprolactone triol–citrate (PCLT–CA) porous scaffold. Human-derived chondrocyte cellular content of glycosaminoglycans (GAGs) and total collagen were determined after seeding into PCLT–CA scaffold enriched with PRPr cells. Immunostaining and real time PCR was applied to evaluate the expression levels of chondrogenic and extracellular gene markers. Seeding of chondrocytes into PCLT–CA scaffold enriched with PRPr showed significant increase in total collagen and GAGs production compared with chondrocytes grown within control scaffold without PRPr cells. The mRNA levels of collagen II and SOX9 increased significantly while the upregulation in Cartilage Oligomeric Matrix Protein (COMP) expression was statistically insignificant. We also report the reduction of the expression levels of collagen I and III in chondrocytes as a consequence of proximity to PRPr cells within the scaffold. Interestingly, the pre-loading of PRPr caused an increase of expression levels of following extracellular matrix (ECM) proteins: fibronectin, laminin and integrin β over the period of 3 days. Overall, our results introduce the PCLT–CA elastomeric scaffold as a new system for cartilage tissue engineering. The method of PRPr cells loading prior to chondrocyte culture could be considered as a potential environment for cartilage tissue engineering as the differentiation and ECM formation is enhanced significantly.





Similar content being viewed by others
References
Snyder TN, Madhavan K, Intrator M, Dregalla RC, Park D. A fibrin/hyaluronic acid hydrogel for the delivery of mesenchymal stem cells and potential for articular cartilage repair. J Biol Eng. 2014;8:10.
Sandell LJ, Aigner T. Articular cartilage and changes in arthritis. An introduction: Cell biology of osteoarthritis. Arthritis Res. 2001;3:107–13.
Mobasheri A, Kalamegam G, Musumeci G, Batt ME. Chondrocyte and mesenchymal stem cell-based therapies for cartilage repair in osteoarthritis and related orthopaedic conditions. Maturitas. 2014;78:188–98.
Kang Y, Yang J, Khan S, Anissian L, Ameer GA. A new biodegradable polyester elastomer for cartilage tissue engineering. J Biomed Mater Res Part A. 2006;77:331–9.
Hildner F, Albrecht C, Gabriel C, Redl H, Van Griensven M. State of the art and future perspectives of articular cartilage regeneration: a focus on adipose-derived stem cells and platelet-derived products. J Tissue Eng Regen Med. 2011;5:e36–51.
Hao T, Wen N, Cao JK, Wang HB, Lu SH, Liu T, et al. The support of matrix accumulation and the promotion of sheep articular cartilage defects repair in vivo by chitosan hydrogels. Osteoarthr Cartil. 2010;18:257–65.
Whitaker MJ, Quirk RA, Howdle SM, Shakesheff KM. Growth factor release from tissue engineering scaffolds. J Pharm Pharmacol. 2001;53:1427–37.
Diaz-Gomez L, Alvarez-Lorenzo C, Concheiro A, Silva M, Dominguez F, Sheikh FA, et al. Biodegradable electrospun nanofibers coated with platelet-rich plasma for cell adhesion and proliferation. Mater Sci Eng C. 2014;40:180–8.
Shimojo AA, Perez AG, Galdames SE, Brissac IC, Santana MH. Stabilization of porous chitosan improves the performance of its association with platelet-rich plasma as a composite scaffold. Mater Sci Eng C. 2016;60:538–46.
Foster TE, Puskas BL, Mandelbaum BR, Gerhardt MB, Rodeo SA. Platelet-rich plasma: from basic science to clinical applications. Am J Sports Med. 2009;37:2259–72.
Demidova-Rice TN, Hamblin MR, Herman IM. Acute and impaired wound healing: pathophysiology and current methods for drug delivery, part 2: role of growth factors in normal and pathological wound healing: therapeutic potential and methods of delivery. Adv Skin Wound Care. 2012;25:349–70.
Tran RT, Yang J, Ameer GA. Citrate-based biomaterials and their applications in regenerative engineering. Annu Rev Mater Res. 2015;45:277–310.
Shirazi HS, Forooshani PK, Pingguan-Murphy B, Djordjevic, I. Processing and characterization of elastomeric polycaprolactone triol–citrate coatings for biomedical applications. Prog Org Coat. 2014;77:821–9.
Rothan HA, Djordjevic I, Bahrani H, Paydar M, Ibrahim F, Abd Rahmanh N, et al. Three-dimensional culture environment increases the efficacy of platelet rich plasma releasate in prompting skin fibroblast differentiation and extracellular matrix formation. Int J Med Sci. 2014;11:1029–38.
Caron MM, Emans PJ, Coolsen MM, Voss L, Surtel DA, Cremers A, et al. Redifferentiation of dedifferentiated human articular chondrocytes: Comparison of 2D and 3D cultures. Osteoarthr Cartil. 2012;20:1170–8.
Lin L, Shen Q, Xue T, Duan X, Fu X, Yu C. Sonic hedgehog improves redifferentiation of dedifferentiated chondrocytes for articular cartilage repair. PLoS One. 2014;9:e88550.
Xu F, Xu L, Wang Q, Ye Z, Zhou Y, Tan WS. 3D dynamic culture of rabbit articular chondrocytes encapsulated in alginate gel beads using spinner flasks for cartilage tissue regeneration. BioMed Res Int. 2014;2014:539789.
Bernardini G, Chellini F, Frediani B, Spreafico A, Santucci A. Human platelet releasates combined with polyglycolic acid scaffold promote chondrocyte differentiation and phenotypic maintenance. J Biosci. 2015;40:61–9.
Gigout A, Jolicoeur M, Buschmann MD. Low calcium levels in serum-free media maintain chondrocyte phenotype in monolayer culture and reduce chondrocyte aggregation in suspension culture. Osteoarthr Cartil. 2005;13:1012–24.
Yates KE, Allemann F, Glowacki J. Phenotypic analysis of bovine chondrocytes cultured in 3D collagen sponges: effect of serum substitutes. Cell Tissue Bank. 2005;6:45–54.
Tseng S, Reddi AH, Di Cesare PE. Cartilage oligomeric matrix protein (COMP): A biomarker of arthritis. Biomark Insights. 2009;4:33–44.
Hamid AA, Idrus RB, Saim AB, Sathappan S, Chua KH. Characterization of human adipose-derived stem cells and expression of chondrogenic genes during induction of cartilage differentiation. Clinics. 2012;67:099–106.
Tavella S, Bellese G, Castagnola P, Martin I, Piccini D, Doliana R, et al. Regulated expression of fibronectin, laminin and related integrin receptors during the early chondrocyte differentiation. J Cell Sci. 1997;110:2261–70.
Fukumoto T, Sanyal A, Fitzsimmons JS, O'Driscoll SW. Expression of β1 integrins during periosteal chondrogenesis. Osteoarthr Cartil. 2002;10:135–44.
Knudson W, Loeser RF. CD44 and integrin matrix receptors participate in cartilage homeostasis. Cell Mol Life Sci. 2002;59:36–44.
Sophia Fox AJ, Bedi A, Rodeo SA. The basic science of articular cartilage: structure, composition, and function. Sports Health A Multidiscip Approach. 2009;1:461–8.
Qiu H, Yang J, Kodali P, Koh J, Ameer GA. A citric acid-based hydroxyapatite composite for orthopedic implants. Biomaterials. 2006;27:5845–54.
Mirza EH, Pan-Pan C, Ibrahim W, Bin WMA, Djordjevic I, Pingguan-Murphy B. Chondroprotective effect of zinc oxide nanoparticles in conjunction with hypoxia on bovine cartilage-matrix synthesis. J Biomed Mater Res A. 2015;103:3554–63.
Zeimaran E, Pourshahrestani S, Pingguan-Murphy B, Kadri NA, Rothan HA, Yusof R, et al. Fabrication and characterization of poly(octanediol citrate)/gallium-containing bioglass microcomposite scaffolds. J Mater Sci. 2015;50:2189–201.
Acknowledgement
This project was funded by University of Malaya-Malaysia (RG377-15AFR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.
Ethical statement
This study was approved by the Medical Ethics Committee, Faculty of Medicine University of Malaya (DFOP 1213/0079 (L)).
Rights and permissions
About this article
Cite this article
Rothan, H.A., Mahmod, S.A., Djordjevic, I. et al. Polycaprolactone Triol–Citrate Scaffolds Enriched with Human Platelet Releasates Promote Chondrogenic Phenotype and Cartilage Extracellular Matrix Formation. Tissue Eng Regen Med 14, 93–101 (2017). https://doi.org/10.1007/s13770-017-0023-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-017-0023-8