Abstract
Visual system pathway dysfunction has been postulated in migraineurs. We wanted to investigate if any difference exists interictally in visual attention and visual evoked habituation of frequently attacked migraineurs compared to the healthy control group. The effects of 3-month prophylactic migraine treatment on these parameters were also assessed. The migraineurs at headache-free interval (n = 52) and age, sex-matched healthy controls (n = 35) were compared by habituation response to 10 blocks of repetitive pattern-reversal visual stimuli (each block consisted 100 responses). The amplitude changes of 5th and 10th blocks were further compared with that of block 1 to assess the response of habituation (i.e., decrease) or potentiation (i.e., increase). The level of sustained visual attention was assessed by Cancellation test. Migraineurs were randomized to three different preventive treatments: propranolol 40 mg tid, flunarizine 5 mg bid, or topiramate 50 mg bid. After 3 months of preventive treatment, migraineurs data were compared with their baseline values. The groups did not differ by sex and age. In electrophysiological studies, the habituation ability observed in the healthy group was not observed in migraineurs. However, it was restored 3 months after preventive treatment. In migraineurs, compared to their baseline values, the distorted visual attention parameters also improved after treatment. All drugs were effective. The loss of habituation ability and low visual attention performance in migraineurs can be restored by migraine preventive treatment. This electrophysiological study accompanied by neuropsychological test may aid an objective and quantitative assessment tool for understanding migraine pathophysiology.

Similar content being viewed by others
References
Robbins MS, Lipton RB (2010) The epidemiology of primary headache disorders. Semin Neurol 30:107–119
Headache Classification Committee of the International Headache Society (IHS) (2013) The international classification of headache disorders, 3rd edition (beta version). Cephalalgia 33:629–808
Choi JY, Oh K, Kim BJ, Chung CS, Koh SB, Park KW (2009) Usefulness of a photophobia questionnaire in patients with migraine. Cephalalgia 29:953–959
Digre KB, Brennan KC (2012) Shedding light on photophobia. J Neuroophthalmol 32:68–81
Chronicle EP, Mulleners WM (1996) Visual system dysfunction in migraine: a review of clinical and psychophysical findings. Cephalalgia 16:525–535
Kowacs PA, Utiumi MA, Piovesan EJ (2015) The visual system in migraine: from the bench side to the office. Headache 55(Suppl 1):84–98
Martin H, del Rio MS, de Silanes CL, Alvarez-Linera J, Hernandez JA, Pareja JA (2011) Photoreactivity of the occipital cortex measured by functional magnetic resonance imaging-blood oxygenation level dependent in migraine patients and healthy volunteers: pathophysiological implications. Headache 51:1520–1528
Delange JM, Cutrer FM (2014) Our evolving understanding of migraine with aura. Curr Pain Headache Rep 18:453
Ambrosini A, Schoenen J (2003) The electrophysiology of migraine. Curr Opin Neurol 16:327–331
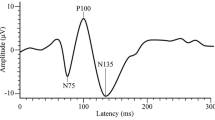
Sand T, Zhitniy N, White LR, Stovner LJ (2008) Visual evoked potential latency, amplitude and habituation in migraine: a longitudinal study. Clin Neurophysiol 119:1020–1027
Meyer JS, Thornby J, Crawford K, Rauch GM (2000) Reversible cognitive decline accompanies migraine and cluster headaches. Headache 40:638–646
Calandre EP, Bembibre J, Arnedo ML, Becerra D (2002) Cognitive disturbances and regional cerebral blood flow abnormalities in migraine patients: their relationship with the clinical manifestations of the illness. Cephalalgia 22:291–302
Suhr JA, Seng EK (2012) Neuropsychological functioning in migraine: clinical and research implications. Cephalalgia 32:39–54
Villa TR, Correa Moutran AR, Sobirai Diaz LA et al (2009) Visual attention in children with migraine: a controlled comparative study. Cephalalgia 29:631–634
Weintraub S, Mesulam MM (2000) Neuropsychological assessment of mental state. In: Mesulam MM (ed) Principles of behavioral and cognitive neurology. Oxford University Press, New York, pp 121–173
Headache Classification Subcommittee of the International Headache Society (2004) The international classification of headache disorders: 2nd edition. Cephalalgia 24(Suppl 1):1–160
Unal-Cevik I, Yildiz FG (2015) Visual snow in migraine with aura: further characterization by brain imaging, electrophysiology, and treatment-case report. Headache 55:1436–1441
Karakas S, Erdogan Bakar E, Dogutepe Dincer E (2006) Handbook of BILNOT battery: research and development of neuropsychological tests, 2nd edn. Eryilmaz Offset, Ankara
Stewart WF, Shechter A, Rasmussen BK (1994) Migraine prevalence. A review of population-based studies. Neurology 44:S17–S23
Burstein R, Noseda R, Borsook D (2015) Migraine: multiple processes, complex pathophysiology. J Neurosci 35:6619–6629
Coppola G, Pierelli F, Schoenen J (2007) Is the cerebral cortex hyperexcitable or hyperresponsive in migraine? Cephalalgia 27:1427–1439
Coppola G, Ambrosini A, Di CL et al (2007) Interictal abnormalities of gamma band activity in visual evoked responses in migraine: an indication of thalamocortical dysrhythmia? Cephalalgia 27:1360–1367
De Tommaso M, Ambrosini A, Brighina F et al (2014) Altered processing of sensory stimuli in patients with migraine. Nat Rev Neurol 10:144–155
Hodkinson DJ, Wilcox SL, Veggeberg R et al (2016) increased amplitude of thalamocortical low-frequency oscillations in patients with migraine. J Neurosci 36:8026–8036
Schmitz N, Admiraal-Behloul F, Arkink EB et al (2008) Attack frequency and disease duration as indicators for brain damage in migraine. Headache 48:1044–1055
Terwindt GM, Ferrari MD, Tijhuis M, Groenen SM, Picavet HS, Launer LJ (2000) The impact of migraine on quality of life in the general population: the GEM study. Neurology 55:624–629
Mangun GR, Hillyard SA (1991) Modulations of sensory-evoked brain potentials indicate changes in perceptual processing during visual-spatial priming. J Exp Psychol Hum Percept Perform 17:1057–1074
Mickleborough MJ, Chapman CM, Toma AS, Chan JH, Truong G, Handy TC (2013) Interictal neurocognitive processing of visual stimuli in migraine: evidence from event-related potentials. PLoS One 8:e80920
Annovazzi P, Colombo B, Bernasconi L, Schiatti E, Comi G, Leocani L (2004) Cortical function abnormalities in migraine: neurophysiological and neuropsychological evidence from reaction times and event-related potentials to the Stroop test. Neurol Sci 25(Suppl 3):S285–S287
Sukhodolsky DG, Leckman JF, Rothenberger A, Scahill L (2007) The role of abnormal neural oscillations in the pathophysiology of co-occurring Tourette syndrome and attention-deficit/hyperactivity disorder. Eur Child Adolesc Psychiatry 16(Suppl 1):51–59
Villa TR, Agessi LM, Moutran AR, Gabbai AA, Carvalho DS (2016) Visual attention in children with migraine: the importance of prophylaxis. J Child Neurol 31(5):569–572. doi:10.1177/0883073815601498
Mulleners WM, McCrory DC, Linde M (2015) Antiepileptics in migraine prophylaxis: an updated Cochrane review. Cephalalgia 35:51–62
Vecsei L, Majlath Z, Szok D, Csati A, Tajti J (2015) Drug safety and tolerability in prophylactic migraine treatment. Expert Opin Drug Saf 14:667–681
Thompson RF, Spencer WA (1966) Habituation: a model phenomenon for the study of neuronal substrates of behavior. Psychol Rev 73:16–43
Sappey-Marinier D, Calabrese G, Fein G, Hugg JW, Biggins C, Weiner MW (1992) Effect of photic stimulation on human visual cortex lactate and phosphates using 1H and 31P magnetic resonance spectroscopy. J Cereb Blood Flow Metab 12:584–592
Schoenen J, Wang W, Albert A, Delwaide PJ (1995) Potentiation instead of habituation characterizes visual evoked potentials in migraine patients between attacks. Eur J Neurol 2:115–122
Omland PM, Uglem M, Hagen K, Linde M, Tronvik E, Sand T (2016) Visual evoked potentials in migraine: is the “neurophysiological hallmark” concept still valid? Clin Neurophysiol 127:810–816
Rauschel V, Ruscheweyh R, Krafczyk S, Straube A (2016) Test-retest reliability of visual-evoked potential habituation. Cephalalgia 36:831–839
Ambrosini A, Coppola G, Iezzi E, Pierelli F, Schoenen J (2016) Reliability and repeatability of testing visual evoked potential habituation in migraine: a blinded case-control study. Cephalalgia. doi:10.1177/0333102416648656
Gerwig M, Niehaus L, Stude P, Katsarava Z, Diener HC (2012) Beta-blocker migraine prophylaxis affects the excitability of the visual cortex as revealed by transcranial magnetic stimulation. J Headache Pain 13:83–89
Ye Q, Yan LY, Xue LJ et al (2011) Flunarizine blocks voltage-gated Na(+) and Ca(2+) currents in cultured rat cortical neurons: a possible locus of action in the prevention of migraine. Neurosci Lett 487:394–399
De Tommaso M, Guido M, Sardaro M et al (2008) Effects of topiramate and levetiracetam vs placebo on habituation of contingent negative variation in migraine patients. Neurosci Lett 442:81–85
Chen WT, Wang SJ, Fuh JL et al (2012) Visual cortex excitability and plasticity associated with remission from chronic to episodic migraine. Cephalalgia 32:537–543
Acknowledgements
The authors report no conflict of interest and funding related with this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors report no conflict of interest related with this manuscript.
Ethical Approval
The study was approved by the Local Ethical Commmitte.
Informed consent
Informed consent was obtained from participants.
Rights and permissions
About this article
Cite this article
Ince, F., Erdogan-Bakar, E. & Unal-Cevik, I. Preventive drugs restore visual evoked habituation and attention in migraineurs. Acta Neurol Belg 117, 523–530 (2017). https://doi.org/10.1007/s13760-017-0749-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-017-0749-z