Abstract
Composting and vermicomposting are sustainable strategies to transform organic wastes into organic amendments, valuable as potting media or soil conditioner. However, the negative aspects of these processes are emissions of greenhouse gases and odorous molecules and final product potentially containing toxic compounds. These negative aspects can be limited through the addition of organic, inorganic or biological additives to the composted or vermicomposted mixture. The aims of this review are (1) to present the main characteristics of composting and vermicomposting processes with and without additives, (2) to show the influence of additives on greenhouse gas emissions during waste degradation and (3) to report the effects of additives on the properties of the final products (heavy metal and nutrient contents), in view of their use as a soil conditioner or potting media. Finally, the feasibility and potential environmental benefits of co-composting and co-vermicomposting are discussed. Our results show that additives affect composting parameters such as temperature, pH and moisture and thus have an impact on the composting process. They may be used to reduce gas emissions and mobility of mineral ions. The various additives have contrasting effects on the quality of the final product and its impact on soil quality. The use of worms and additives seems to increase plant available nutrient contents, while decreasing N leaching, heavy metal mobility and composting time. Co-composting and co-vermicomposting strategies need to be locally optimised, involving the generated amendments in a circular economy to improve sustainability of agricultural systems.
Similar content being viewed by others

-
1. Introduction
-
2. Biological degradation processes and additives used for organic waste treatment
-
2.3 Types and sources of additives used during waste biological treatment
-
4. Influence of additives on gas emissions during composting
-
4.1 Odour emissions
-
4.2 GHG emissions
-
5.2 Influence of additives on nutrient contents and availability
-
6. Environmental perspectives and feasibility of co-composting
-
6.2 Reduction of GHG emissions and carbon sequestration potential of soil amended with co-compost
-
7. Conclusion
-
Acknowledgements
-
References
1 Introduction
The growing world population and the resulting higher consumption of goods and services has driven a rapid increase of organic wastes originating from households, industry and agriculture (Hoornweg et al. 2013). This situation generates serious environmental issues, calling for safe and sustainable strategies to treat these wastes, from their production to their recycling or elimination. On the other hand, the organic fraction of wastes represents a valuable organic resource, which could be recycled and transformed into nutrient-rich fertiliser and/or soil conditioner (Marshall and Farahbakhsh 2013; Bernstad et al. 2016; Calabi-Floody et al. 2017). Biological degradation during composting and vermicomposting is one of these strategies to transform organic wastes into organic amendments. These amendments can be applied on soils to increase soil carbon stocks and associated ecosystem services (Francou et al. 2005; Lashermes et al. 2009, Peltre et al. 2012; Bernstad et al. 2016) or used as potting media (Hashemimajd et al. 2004; Zaller 2007; Ceglie et al. 2015; Morales et al. 2016; Wang et al. 2017; Malińska et al. 2017). Compost application leads to improved soil structure, reduced erosion and increased water holding capacity (Diacono and Montemurro 2010). However, compost production is associated with greenhouse gas (GHG) emissions such as NO2, CH4 or CO2, contributing to global warming (Bernstad et al. 2016). In addition, immature composts have negative effects on plant germination and, more generally, on plant development (Bernal et al. 2009) and might cause environmental problems such as water pollution and odour emissions (Wang et al. 2016).
Composting is an aerobic process, performed at large scale in windrows or piles (Hobson et al. 2005) or at local scale with a home composter (Andersen et al. 2011). Composting is used to treat manures, green wastes or municipal solid wastes and their fractions (Goyal et al. 2005). Vermicomposting, involving the presence of worms, is used for similar purposes (Chan et al. 2011; Garg et al. 2006; Ngo et al. 2011).
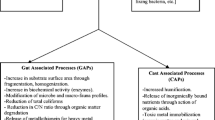
Various substrates might be added to waste during the composting process. They are either organic (Gabhane et al. 2012; Zhang et al. 2017), mineral (Wong et al. 1995; Himanen and Hänninen 2009; Wang et al. 2016), biological (Wakase et al. 2008; Jurado et al. 2015; Awasthi et al. 2017) or a mixture of substrates (Hayawin et al. 2014; Awasthi et al. 2018). Some added substrates are considered as bulking agents, when they only act on the physical structure of the compost (aeration), but most of the time, these substrates have also direct or indirect effects on other composting parameters and can be considered as additives (Villasenor et al. 2011). Additives are used to enhance the composting process by reducing leaching (Steiner et al. 2010) and gas emissions (McCrory and Hobbs 2001; Awasthi et al. 2016a, b), improving compost aeration or accelerating organic matter degradation (Sánchez-García et al. 2015) and improving nutrient content and availability in the final product (Gabhane et al. 2012; Morales et al. 2016). Only few studies assessed the effect of additives on vermicomposting (Wang et al. 2014; Barthod et al. 2016; Malińska et al. 2017) (Fig. 1).
Many studies were published in the last years proposing to use various additives during composting and vermicomposting of different feedstocks, but no critical review exists to compare their benefits. The main aims of this article are to present the different additives used during composting and vermicomposting and to evaluate their effect on the main composting parameters and on the quality of the end product in terms of their effect on soil functions and plant growth. The objective of this review is to give recommendations with regard to additive use during composting and vermicomposting. Finally, perspectives and feasibility of using additives during both processes will be discussed.
2 Biological degradation processes and additives used for organic waste treatment
2.1 The composting process
Composting is defined as a bio-oxidative process leading to organic matter mineralisation and transformation (Zucconi and de Bertoldi 1987). The end product is considered as stabilised and free of phytotoxic agents and pathogens (Wichuk and McCartney 2007). Composting typically consists of three phases: initial activation, followed by a thermophilic and a maturation phase (Fig. 2). The initial activation generally lasts 1–3 days during which simple organic compounds such as sugars are mineralised by microbial communities, producing CO2, NH3, organic acids and heat (Bernal et al. 2009). During this phase, the temperature in the composting pile increases. Thereafter, during the thermophilic phase, the temperature reaches a maximum. The optimum temperature range for composting is 40–65 °C (de Bertoldi et al. 1983), allowing to kill pathogens above 55 °C. During this phase, thermophilic microorganisms degrade fats, cellulose and lignin (Bernal et al. 2009). Finally, during the mesophilic phase or maturation, temperature slowly decreases due to reduced microbial activity resulting from a decrease of biodegradable compounds. Composting involves a succession of microbial communities (Insam and de Bertoli 2007; Mehta et al. 2014), according to the temperature of the composting pile. For instance, fungi are not present at a temperature above 60 °C, when bacteria predominate (Klamer and Bååth 1998). Thus, the relative abundance of specific microorganisms and temperature are good indicators of the compost evolution.
Temperature changes during composting and corresponding microbial communities and organic compounds. Stars indicate the phases when the removal of pathogen microorganisms takes place (after Insam and de Bertoli 2007). Mineral, organic as well as biological additives have been shown to stimulate the microbial activity, leading to an earlier start and a longer duration of the thermophilic phase as compared to regular composting
Biogenic CO2 emissions during composting derive from organic matter aerobic decomposition and CH4 oxidation by aerobic methanotrophic bacteria (Sánchez-García et al. 2015). CO2 is the most abundant gas emitted during composting (Haug 1993). CH4 emissions are mostly recorded during the initial and thermophilic phases (Beck-Friis et al. 2000), due to the formation of anaerobic spots. Nitrous oxide (N2O) emissions can occur during the whole process due to denitrification. NH3 emissions may also occur during composting, generally during the first 2 weeks. They decrease when the easily degradable materials are exhausted and the degradation rate decreases (Morand et al. 2005; Szanto et al. 2007).
The composting process and compost quality and maturity are controlled by several parameters influencing the microbial activity, such as the initial pH and C/N ratio of the feedstock, their particle size and their distribution, as well as the aeration and humidity of the compost pile (Bernal et al. 2009, Onwosi et al. 2017). The temperature profile during composting is influenced by the composition of the initial feedstock, aeration through pile turning, compost humidity and the addition of additives or bulking agents (Pasda et al. 2005). It influences compost duration, compost quality and maturity, but also compost sanitisation (Soobhany et al. 2017). CO2 and NH3 emissions, mainly produced during the thermophilic stage of organic matter degradation (Maulini-Duran et al. 2014; Pagans et al. 2006), are affected by the C/N ratio of the composted material, the maximum temperature reached during the process and the aeration of the pile (Pagans et al. 2006). The presence of anaerobic zones is responsible for N2O and CH4 emissions (Nasini et al. 2016).
Controlling compost parameters and influencing them with additives have major impacts in terms of compost quality (maturity, heavy metal contents, nutrient content and bioavailability) and environmental impacts of composting, such as production of greenhouse gases and other volatile compounds such as NH3, sulphur-containing compounds and volatile organic compounds (Maulini-Duran et al. 2014; Cerda et al. 2018).
2.2 The vermicomposting process
During vermicomposting, temperature ranges from 25 to 37 °C, depending on the worm species. Generally, worm activities and development are negatively affected by higher or lower temperatures. Four species are extensively used in vermicomposting facilities. They comprise two tropical species [Eudrilus eugeniae (Reinecke et al. 1992; Kurien and Ramasamy 2006; Hayawin et al. 2014) and Perionyx excavatus (Suthar and Singh 2008)] and two temperate ones [Eisenia andrei and Eisenia fetida (Kaushik and Garg 2003; Garg et al. 2006; Malińska et al. 2017; Alavi et al. 2017)]. These species have been classified as epigeic worms and correspond to worms, which mainly feed on fresh organic matter such as litter, manure and compost (Bouché 1977). Most vermicomposting facilities and most studies are using the worms Eisenia andrei and Eisenia fetida due to their high rate of consumption, digestion and assimilation of organic matter, adaptation to a wide range of environmental factors, short life cycles, high reproductive rates and endurance and resistance during handling (Domínguez and Edwards 2010).
Although pathogen removal has been recently demonstrated to occur during transit in the worm gut (Soobhany et al. 2017) as well as during composting, combining vermicomposting to composting is the preferred process, because it favours the removal of pathogen microorganisms and increases organic matter decomposition rate (Frederickson et al. 1997, 2007; Wang et al. 2014). Wastes are composted during a few weeks, until the end of the thermophilic phase. Then, after some days at high temperature, pre-mature compost is cooled by spreading it on vermicomposting beds (thin layers).
Vermicomposting produces GHG, similar to composting. Worm activity accelerates and enhances organic matter decomposition leading to higher CO2 emissions than those of regular composting (Chan et al. 2011; Nigussie et al. 2017). The emissions of N2O are either increased (Hobson et al. 2005) or decreased (Wang et al. 2014; Nigussie et al. 2017) compared to composting, probably depending on the feedstock materials.
As already noted for composting, vermicomposting parameters must be controlled because they influence the rate of decomposition, the quality of the end product and the amount of GHG emitted. In addition, for vermicomposting, worm mortality must be avoided, by regulating, in particular, the temperature and the moisture content. As for composting, the parameters controlling vermicomposting may be influenced by the addition of mineral, organic or biological substrates (Suthar 2009; Li et al. 2009; Malińska et al. 2017). For vermicomposting, the choice of additives is also driven by their possible negative impacts on worm development (Malińska et al. 2016).
2.3 Types and sources of additives used during biological waste treatment
A wide variety of additives, classified in three categories (mineral, organic or biological) may be used to enhance composting and vermicomposting processes and/or the quality of the final product (Bernal et al. 2009; Onwosi et al. 2017). A non-exhaustive list of the common additives is given in Table 1.
Biological additives refer to microorganisms, which are inoculated to a compost or vermicompost pile. These microorganisms are generally isolated from composts during the thermophilic phase, cultivated and sold as a commercial solution. Effective microbes and vertical transmitter bacteria are common commercial additives (Manu et al. 2017). Few manufacturers reveal the identity, fate and functions of the microbes present in the commercial solutions. However, in most of the commercial microbiological additives, Alcaligenes, Bacillus, Clostridium, Enterococcus and Lactobacillus microorganisms are present (Sasaki et al. 2006; Wakase et al. 2008). These microorganisms are involved in the ammonia assimilation during composting (Wakase et al. 2008) or in the decomposition of lignocellulose (Jurado et al. 2015).
Organic additives cover a large variety of products: residual straws, mature composts, refuse from green waste compost screening, grass clippings, crushed hardwood materials, crushed wood pallets, bark and cornstalks (Doublet et al. 2011). When choosing organic additives, attention must be paid to the C/N ratio of the initial mixtures to ensure organic matter degradation and prevent N leaching during composting (Doublet et al. 2011). Furthermore, biochar, a highly aromatic pyrolysis product (Lehmann and Joseph 2015), has recently received great interest as highly stabilised organic additive for composting (Dias et al. 2010; Waqas et al. 2017) and vermicomposting (Malińska et al. 2017; Barthod et al. 2016). As the production of biochar was shown to yield highly aromatic materials with high stability when added to soil, biochar may enhance the carbon sequestration potential of composts and vermicomposts (Lehmann et al. 2006; Lehmann 2007), thus mitigating climate change. A great variety of biochars is produced, with different properties depending on the initial feedstock and pyrolysis temperature (Zhao et al. 2013). Generally higher temperatures lead to greater aromatisation of the material, decreasing its surface area, cation exchange capacity and content of volatile compounds (Lehmann et al. 2006).
The main inorganic or mineral additives are lime, clays or industrial wastes, for example red mud or fly ash. Red mud is a by-product of the industrial alumina production (Wang et al. 2008), and fly ash is a waste product of clean coal combustion used to mitigate gas emissions (e.g. used in power plants). Therefore, the main advantages of these alkaline materials are their high availability (Gomes et al. 2016) and their low cost as industrial wastes.
Minerals such as zeolite or Ca-bentonite became popular in the last decades due to their physical and chemical properties to adsorb heavy metals, in particular in soils and water purification systems (Lopez et al. 1998; Wang et al. 2016). Pure zeolite is easily synthesised by the slow crystallisation of a silica-alumina matrix. Finally, pure minerals such as clay, extracted from quarries or soils, are increasingly used with the aim of reducing greenhouse gas emissions during composting (Bolan et al. 2012; Barthod et al. 2016). These minerals are sometimes added during vermicomposting (Li et al. 2009; Hayawin et al. 2014; Barthod et al. 2016).
3 Effects of additives on the composting process
During composting, the naturally developing microbial community can be affected by additives, directly, when adding an inoculum, or indirectly, when adding organic or mineral materials inducing changes in the aeration, temperature, moisture content, pH, nutrient availability, etc.
3.1 Effects of additives on composting temperature profiles
The temperature profile is a good indicator of the microbial activity during composting (Haug 1993) and important to assess the removal of pathogens through high temperature (Gea et al. 2007). It can change due to the presence of additives. Mineral, organic as well as biological additives have been shown to stimulate the microbial activity, leading to an earlier start and a longer duration of the thermophilic phase as compared to regular composting (Fang and Wong 1999; Chen et al. 2010; Doublet et al. 2011; Himanen and Hänninen 2011; Gabhane et al. 2012; Jiang et al. 2015a; Morales et al. 2016). The duration of the thermophilic phase increased from 2 to 3 weeks following the addition of commercial products containing zeolite, kaolinite, chalk, ashes and sulfates (Himanen and Hänninen 2009) or biochar addition during biowaste and food waste composting, thereby shortening the composting process. For example, compost stability was already achieved after 50–60 days when amended with biochar (Waqas et al. 2017). Rapid temperature increases were observed following the addition of biochar as well as mineral and polymer additives (zeolite, jaggery and polyethylene glycol) during composting of animal manure, food waste and green waste (Chen et al. 2010; Czekała et al. 2016; Waqas et al. 2017; Venglovsky et al. 2005; Gabhane et al. 2012). Similar temperature profiles were recorded for compost with biological or organic additives (Manu et al. 2017; Nakasaki and Hirai 2017). The more rapid temperature increase with these additives may be due to the increase of microbial biomass and activity. However, some additives such as bentonite (Li et al. 2012), phosphogypsum and lime (Gabhane et al. 2012) do not change the temperature profile, suggesting that they do not affect microbial biomass.
3.2 Additives to stimulate microbial activity
The additives influence the microbial communities of composts through their effect on temperature, moisture and aeration of the pile (Fuchs 2009), which is due to their contents of nutrients and readily available forms of carbon. For example, adding jaggery increased the number of microorganisms and thus enhanced the enzymatic degradation of cellulose during composting of green wastes (Gabhane et al. 2012). Similar results were obtained with the addition of fish pond sediments, spent mushroom substrate and biochar (Zhang and Sun 2014; Zhang et al. 2017). The effect of biochars on microbial activity was probably due to their effect on microbial habitat and protection from grazers (Meng et al. 2013; Wei et al. 2014). Biochar, due to its porous structure, might also enhance microbial activity through moisture and aeration control (Waqas et al. 2017), which has an effect on compost temperature as discussed in the previous section. However, a biochar application rate higher than 20% is not recommended since it may hinder organic matter biodegradation (Liu et al. 2017; Xiao et al. 2017). Alkaline substrates such as fly ash and lime are also additives with high nutrient contents; however, they do not favour microbial activity due to their high pH (Fang et al. 1999). At high rates, fly ash addition inhibited phosphatase, β-glucosidase or dehydrogenase but did not affect urease activity, indicating that some stages of N-containing compound degradation were unchanged (Wong et al. 1997).
While mineral additives can change the microbial activity indirectly, microbial consortium addition directly changes the microbial community and activity of the compost. A commercial microbial additive changed the temperature profile of the composting process and the ammonia emissions due to the increase in the mesophilic and thermophilic bacteria compared to the regular composting without additives (Sasaki et al. 2006). The non-dominant microbes of commercial consortium influenced more the compost microbial composition than the dominant ones (Wakase et al. 2008). Isolation of microbial communities prepared by incubation mixtures of cow manure, soil and straw allowed for the selection of microbial colonies with specific degradation functions (Liu et al. 2011). These preparations also increased the duration of the thermophilic phase and were thus able to enhance the microbial diversity of compost (Liu et al. 2011).
Moreover, as discussed above, zeolite, biochar and polyethylene glycol increase microbial activity at doses mostly lower than 5%. Their addition may thus be a good way to reduce the length of the composting process.
3.3 Additives to improve aeration
An optimal aeration during composting is required as explained by Gao et al. (2010). A low aeration rate might lead to anaerobic conditions, while a high aeration rate might result in excessive cooling, thus preventing thermophilic conditions. The common way to enhance aeration and thus favour the microbial activity during composting is through mechanically turning the composted material (Chowdhury et al. 2014; Chen et al. 2015; Manu et al. 2017) and forced aeration through pipes (Ogunwande and Osunade 2011). These actions are not necessary in the case of vermicomposting due to worm activity. The costs of the pile turning or forced aeration may be reduced by the use of bulking agents, such as biochar, residual straws, woodchips or sawdust and crushed branches, since they enhance the natural aeration and porosity of the composting pile (Kulcu and Yaldiz 2007; Czekała et al. 2016). Due to their numerous pores and low moisture content, these bulking agents support the formation of inter- and intraparticle voids (Iqbal et al. 2010). Biochar addition can also improve compost aeration due to its porous structure (Waqas et al. 2017). Moreover, the biological oxidation rate is directly related to the surface area exposed to the microbial attack (de Bertoldi et al. 1983).
3.4 Additives to regulate moisture content
The moisture content during composting influences the oxygen uptake rate and thus the microbial activity and the degradation rate. An optimal moisture content hence decreases the compost maturation time (Iqbal et al. 2015 b). The optimal water content for organic matter biodegradation has been estimated within 50–70% (moisture content in wet basis: ratio of the weight of water to the weight of the material) (Richard et al. 2002). However, some organic wastes have higher or lower moisture content; for example, sewage sludge has a moisture content ranging from 80 to 90%. Such high humidity may favour anaerobic conditions and thus odour production during composting (Jolanun et al. 2008). The bulking agents commonly chosen to offset the high moisture content of organic wastes are fibrous materials (Miner et al. 2001, Eftoda and McCartney 2004, Doublet et al. 2011), which can absorb part of the leachate. Leachate absorption may be achieved by cornstalk, sawdust or spent mushroom substrate (Yang et al. 2013). Furthermore, Chang and Chen (2010) showed that increasing water absorption capacity by sawdust addition also resulted in higher degradation rate due to more air flow through the particles.
On the contrary, water loss during the first days of composting might delay the composting process and necessitates water sprinkling. The addition of materials with high water retention properties, such as clays, might limit water losses. Li et al. (2012) showed that the initial moisture decrease was buffered by the presence of bentonite, due to its swelling performance. During composting of green wastes, the water holding capacity was also increased by the addition of ash (Belyaeva and Haynes 2009) or phosphate rock (Zhang and Sun 2017). However, some additives such as eggshells had no influence on water holding capacity (Soares et al. 2017) and may even have a negative effect on the biological activity.
3.5 Additives to buffer pH
The pH varies during composting with a decrease during the early stages and an increase during the later stages (Onwosi et al. 2017), which affects microbial activities. Some additives are used to increase the pH and thus enhance the composting of acid feedstocks, such as food waste (Wong et al. 2009). The addition of an inoculum consortium was reported to result in a pH increase from 4.3 to 6.3 during food waste composting (Manu et al. 2017). This may be explained by the degradation of acids along with organic matter through enhanced biological activity. The use of bulking agents such as bagasse, paper, peanut shell, sawdust and Ca-bentonite might also increase the pH during composting (Iqbal et al. 2010; Wang et al. 2016), similar to fly ash, lime or red mud addition (Fang and Wong 1999; Gabhane et al. 2012; An et al. 2012; Chowdhury et al. 2016). However, such alkaline additives might inhibit the metabolic activity. For example, Wong et al. (1997) observed less thermophilic bacteria in the initial phase, when sludge was co-composted with 25% fly ash.
The increase of pH during the thermophilic phase was offset by using bamboo charcoal or zeolite, which was explained by the capacity of these additives to adsorb the ammonia produced by the mineralisation of organic nitrogen (Chen et al. 2010; Venglovsky et al. 2005). A lower pH may also decrease nitrogen loss by avoiding ammonia volatilisation, which occurs at high pH (Chen et al. 2010). Finally, the addition of elemental sulphur considerably decreased the pH during poultry manure composting (Mahimairaja et al. 1994), mainly due to the oxidation of elemental sulphur producing H2SO4, thus increasing the H+ ion concentration.
4 Influence of additives on gas emissions during composting
4.1 Odour emissions
Sulphur-containing compounds and NH3 are the main odorous gases emitted during composting. Emission of NH3 not only causes environmental pollution and odorous nuisances but also decreases the agronomic value of the compost (Ermolaev et al. 2015). We found no reference in the literature on vermicomposting and odour emissions, likely due to the fact that worm activities allow a continuous aeration of the composting pile, which limits the formation of anaerobic zones responsible for odour emissions. By improving oxygen transfer inside the composting pile of municipal wastes through the addition of rice straw (1:5), Shao et al. (2014) decreased the cumulative malodorous sulphur-containing gases. Similar results have been obtained after ash (Koivula et al. 2004) and biochar (Steiner et al. 2010; Hua et al. 2009; Khan et al. 2014) addition.
Another way to reduce the odour emissions is to trap the nitrogen excess and thus reduce the ammonia contents in the composting pile. Natural zeolite may be added for this purpose (Lefcourt and Meisinger 2001; Turan 2008; Wang et al. 2014; Awasthi et al. 2016b), the odour reduction being proportional to the amount of zeolite added. Zeolite addition on top of the pile may reduce ammonia emissions (Witter and Lopez-Real 1988; Bernal et al. 1993). Biochar was found to decrease ammonia losses when N-rich wastes were composted (Steiner et al. 2010). However, additives may also increase ammonia losses due to the enhancement of microbial activities (Waqas et al. 2017; Jiang et al. 2015b). By contrast, organic additives such as sawdust, cornstalk and spent mushroom substrates did not influence ammonia emissions (Yang et al. 2013; Yuan et al. 2015).
Chemical additives (a nitrogen electron acceptor, sodium nitrate and sodium nitrite or FeCl3) may be used to control odours related to sulphur-containing compounds by reducing the emissions of dimethyl sulphide and dimethyl disulphide, without altering the composting process (Yuan et al. 2015; Zang et al. 2017). A number of chemical additives may be used to reduce ammonia losses during composting, for example absorbent mixtures of magnesium hydroxide and phosphoric acid (Jeong and Hwang 2005; Ren et al. 2010), calcium superphosphate (Zhang et al. 2017) or other phosphate and magnesium salts (Jiang et al. 2016). Some specific additives such as dicyandiamide were used to inhibit the nitrification process in order to decrease the ammonia emissions (Jiang et al. 2016).
4.2 GHG emissions
The amounts of GHG released during composting of different initial feedstock with different additives are shown in Table 2. GHG emissions from co-composting processes have been largely studied, but few data are available for vermicomposting processes and even less for co-vermicomposting. Moreover, among GHG gases, only CH4 and N2O emissions during composting are taken into consideration in national GHG inventories, while CO2 emissions of biogenic origin are not accounted for (IPCC 2014). The amount of N2O and CH4 emitted during the composting process may depend on the feedstock and on the aeration conditions in the compost pile. Aeration in the compost pile may be enhanced by the addition of bulking agents or by mechanical turning. The proportion of added bulking agent and the turning frequency of the composting pile are thus important process parameters, which need to be controlled for regulating GHG emissions, in particular gaseous N losses (Szanto et al. 2007; Maulini-Duran et al. 2014; Morales et al. 2016).
N losses in the form of N2O can occur under aerobic conditions due to incomplete nitrification/denitrification or anaerobic conditions, when a lack of O2 leads to nitrate accumulation. Additives affect differently the N2O emissions according to the initial feedstock and the type of additive. For instance, mineral additives such as phosphogypsum (Hao et al. 2005; Luo et al. 2013) significantly reduced N2O emissions during manure composting, probably by increasing SO42- content of the compost or by altering the nitrification process. No effect was observed after the addition of woodchips and polyethylene tubes as bulking agents during municipal waste composting (Maulini-Duran et al. 2014), while sawdust addition significantly reduced N2O emissions during kitchen waste composting (Yang et al. 2013).
In a composting pile, CH4 emissions generally occur due to a lack of oxygen as a result of excessive moisture or presence of anaerobic zones (Amlinger et al. 2008). Contrary to N2O emissions, CH4 emissions appear to be more dependent on the properties of the additives than those of the initial feedstock. Thus, to reduce CH4 emissions, two types of additives might be applied, affecting directly or indirectly the carbon cycle: (1) biological additives in the form of methanotrophic bacteria (Luo et al. 2014) to enhance CH4 oxidation and thus reduce CH4 emissions and (2) organic additives as bulking agents to limit anaerobic zones (Yang et al. 2013; Maulini-Duran et al. 2014) or to trap emitted gases (Awasthi et al. 2016a). Physical properties of additives such as particle size may impact their efficiency for reducing CH4 emissions (Yang et al. 2013). For example, compact zones leading to even increased CH4 emissions may be formed after the addition of some specific organic bulking agents, such as sawdust and spent mushroom (Maulini-Duran et al. 2014), while small biochar particles may trap more gas than big ones due to increased surface area.
CO2 emissions may be impacted by the type of additive, in particular the decomposability of organic additives. These emissions are generally increased by the addition of readily decomposable organic materials (paper, straw, peat materials, etc.) (Mahimairaja et al. 1994) and decreased by the addition of organic materials rich in lignin or other slowly degrading organic compounds (Mahimairaja et al. 1994; Chowdhury et al. 2014). Biochar addition during composting has contradictory effects on CO2 emissions compared to regular compost, either increasing (Czekała et al. 2016; Wu et al. 2017) or decreasing them (Chowdhury et al. 2014). Such contradictory effects were also observed for vermicomposting (Barthod et al. 2016). Organic additives in the form of bulking materials, such as plastic or pumice, increase CO2 emissions through aeration improvement and microbial activity enhancement (Wu et al. 2015; Czekała et al. 2016). In conclusion, reducing CO2 emissions from composting without altering the biodegradation process appears difficult. Chemical and mineral additives have the potential to limit CO2 emissions, for example by trapping emitted CO2 with an adsorbent, such as red mud, or by protecting carbon from decomposition through associations with clays or amorphous hydroxyl-Al (Bolan et al. 2012; Haynes and Zhou 2015; Barthod et al. 2016).
In conclusion, the effect of additives on the composting process and on gas emissions might be contradictory. The additives must be chosen according to their possible positive and negative effects (Fig. 3), in view of developing amendments with specific properties.
5 Quality of co-compost for use as potting media or soil amendments: influence of additives on nutrient availability, metal availability and soil fertility
Soil health and functions, such as fertility, can be improved by compost and vermicompost amendment, through their effects on soil structure and porosity, soil microbial biomass and soil nutrient contents (Thangarajan et al. 2013). In addition to soil application, composts and vermicomposts are largely used as horticultural potting media, because they may replace peat, which is a limited resource (Hashemimajd et al. 2004; Zaller 2007). The presence of heavy metals or high content of nutrients in composts might cause important adverse effects on animal and human health (Senesil et al. 1999) and on plant growth. Therefore, the nutrient and heavy metal contents as well as their respective availability and mobility in the compost are important parameters to control. Additives can be used to improve the agronomic value of composts related to their nutrient availability or metal mobility.
5.1 Reduction of environmental hazards due to heavy metals
Depending on the initial feedstock, heavy metals might be present in the composted material (Swati and Hait 2017). Heavy metal contents are high in composts originating from animal manure or sewage sludge. During composting and vermicomposting, heavy metals can react with organic matter and their speciation might change. For instance, manure composts may contain high levels of Zn, Cd, Pb and Cu and their application on soils may lead to an excessive input of heavy metals (Chen et al. 2010). Using additives during composting is a way to reduce heavy metal availability, resulting in a marketable, safe material. Heavy metal mobility, in particular of Cu, Zn, Cd, Pb and Mn in water-soluble form, can be reduced by organic and mineral additives. Organic additives like bamboo charcoal, chestnut/leaf litter and biochar were used for this purpose (Chen et al. 2010 Guerra-Rodríguez et al. 2006; Lau et al. 2001). Mineral additives favorable to reduce heavy metal mobility are fly ash, Ca-bentonite, phosphate rock, lime or zeolite (Wang et al. 2016; Lu et al. 2014; Fang and Wong 1999; Wong et al. 2009; Nissen et al. 2000; Zorpas et al. 2000; Villasenor et al. 2011). Mixtures of organic and mineral additives in the form of lime and biochar were also effective for reducing heavy metal mobility in composts (Awasthi et al. 2016a). When using mineral additives during composting, the decrease of the labile fractions of heavy metals in manure or sewage sludge is mainly due to the formation of organo-metal complexes between additives and metals (Wang et al. 2013b) or a complexation of the metal ions (Lu et al. 2014). Lime and biochar limit heavy metal mobility also due to a pH effect.
Co-vermicomposting may be used to decrease heavy metal hazards, since worms act as bio-accumulators of heavy metals (He et al. 2016) or may change the interaction of heavy metals with organic matter (Li et al. 2009; Swati and Hait 2017). Organic additives, such as biochar, straw and sawdust, decreased the bioavailability of heavy metals to worms during vermicomposting (Malińska et al. 2017) and reduced the concentration of heavy metals in vermicomposts (He et al. 2016). During sewage sludge composting, worms improved the efficiency of additives such as fly ash or sawdust by increasing the exchangeable fraction of essential nutrients such as Zn and Cu and decreasing the soluble forms of non-essential elements such as Cr and Pb (Wang et al. 2013a). Mineral additives were found to decrease heavy metal content and availability in co-vermicomposts (Hayawin et al. 2014; He et al. 2016).
The application of co-compost to soil may also reduce soil environmental hazards related to heavy metal-polluted soils. Zhou et al. (2017) recommended the application of co-composts (obtained with red mud) on polluted soils to decrease the ecological risk of heavy metals by efficiently reducing soil heavy metal availability and increasing soil microbial biomass.
5.2 Influence of additives on nutrient contents and availability
The major possible negative effect of additives on composts or vermicomposts is a decrease of their nutrient content due to dilution (Banegas et al. 2007) or a decrease of nutrient availability due to adsorption on mineral additives. In some cases, the adsorption of excess nutrients from compost is beneficial to avoid environmental risks, such as eutrophication, after compost application to soil.
Mineral additives, such as coal fly ash, zeolite and alum, reduce compost-soluble P contents compared to regular composting of sewage sludge, due to the formation of insoluble phosphate compounds (Menon et al. 1993; Fang et al. 1999) or adsorption and formation of insoluble Al-phosphate intermediates (Lefcourt and Meisinger 2001; Belyaeva and Haynes 2012). Likewise, zeolite addition may retain soluble N (NH4+) (Li et al. 2008) and lead to a significant decrease in the C/N ratio and an increase in total nitrogen, total available phosphorous and total potassium (Hayawin et al. 2014).
Inversely, some mineral additives, such as phosphate rock (Nishanth and Biswas 2008; Billah and Bano 2015), lime and fly ash (Gabhane et al. 2012; Zhang et al. 2017) or medical stone (Wang et al. 2017), are used to increase nutrient contents of final composts made from nutrient-poor feedstocks. Nitrogen content increased in the presence of 2.5% bentonite during composting (Li et al. 2012) or with both bacteria (N2-fixing and P-solubilising) and phosphate rock during vermicomposting (Busato et al. 2012; Unuofin and Mnkeni 2014).
The use of organic additives in the form of branches, pallets and cornstalks during sludge composting had a positive effect on N availability in the final composts by enhancing the N organisation and limiting N losses by volatilisation during composting (Doublet et al. 2011). The effect of biochar on nutrient leaching from compost is contradictory (Chen et al. 2010; Iqbal et al. 2015a; Wu et al. 2017). Co-composting of food waste with biochar increased concentrations of NH4+ and NO3− (Waqas et al. 2017).
Worms may increase the bioavailability of N and P during vermicomposting with mineral additives. Unuofin and Mnkeni (2014) recommended to use a high worm density, up to 22.5 g kg−1, in order to maximise phosphorus availability in co-vermicompost with phosphate rock.
Co-composts used as potting media or soil amendment can improve fertility through two mechanisms: (1) because they are rich in nutrients when they are made with additives rich in available nutrients and/or (2) because they increase microbial biomass, which plays a pivotal role in nutrient cycling. In this way, soil application of co-composts obtained with phosphate rock and bacteria inoculum resulted in an increase of not only the soil microbial biomass but also the microbial biomass phosphorus, inducing high amounts of available phosphorus (Meena and Biswas 2014).
However, these effects are highly dependent on pedoclimatic conditions, which influence the availability of nutrients, such as P or N, and their leaching. For instance, in acidic soils, P ions may precipitate, thereby decreasing P availability (Hinsinger 2001), while nitrogen contents decrease when the pH falls below 5 (Lucas and Davis 1961). Thus, to improve the fertility of acid tropical soils, it was recommended to apply a co-compost of wood ash with high pH to retain Al and increase the bio-availability of exchangeable cations (Bougnom et al. 2009). The application of co-composts (rate and additives) should be optimised and adapted to the soil properties to improve soil health and avoid environmental risks (Roca-Pérez et al. 2009).
6 Environmental perspectives and feasibility of co-composting
6.1 Co-compost: a potential stimulator of plant growth
Few studies assessed the impact of co-composts on plant growth, and even less took place under field conditions (Chowdhury et al. 2016; Kuba et al. 2008). Moreover, to the best of our knowledge, none assessed the effect of co-vermicomposts on plants. The effect of co-composts on plants is generally tested by assessing the germination index, a biological indicator used to evaluate the toxicity and maturity of compost (Zucconi et al. 1981). A germination index higher than 50% indicates a phytotoxin-free compost. The presence of additives may influence this germination index, since additives have an effect on nutrient availability (see above). Chen et al. (2010) showed that the addition of bamboo charcoal and bamboo vinegar during pig manure composting increased this index up to 95%, suggesting that charcoal can be considered as a plant growth stimulator, while several studies concluded that the presence of mineral additives such as bentonite and alkaline materials inhibits plant growth (Fang and Wong 1999; Samaras et al. 2008; Li et al. 2012). Moreover, the nutrient uptake of plants can be changed by co-compost application. Zayed and Abdel-Motaal (2005) showed that co-compost with biological additives enhanced the plant phosphorous uptake and, in addition, decreased the growth of bacteria in the rhizosphere. Similarly, mineral additives can increase nutrient availability to plants. For example, phosphate rock-enriched compost supplied more phosphorous to seedling than a usual growing media (Mihreteab et al. 2015) and led to higher biomass yield, uptake and recoveries of P and K when prepared with additional waste mica (Nishanth and Biswas 2008). Moreover, the use of specific amendments able to absorb heavy metals limited metal transfers to plant (Nissen et al. 2000).
Finally, the few field experiments carried out with co-composts showed that they might improve soil fertility and thus potential revegetation of degraded soil. Kuba et al. (2008) showed that a co-compost produced with 16% wood ash was more efficient than mineral and organic fertilisers for increasing plant cover during ski slope revegetation. Similarly, Chowdhury et al. (2016) observed that co-composts (biowastes with alkaline amendment) improved soil fertility when used to revegetate an urban landfill soil (Fig. 4).
6.2 Reduction of GHG emissions and carbon sequestration potential of soil amended with co-compost
The degree of degradation of compost organic matter is an important parameter to consider for their use as soil amendment (Francou et al. 2005). A stable organic matter applied to soil emits less carbon than an instable one (Bernal et al. 1998). Stability is related to compost chemical composition (Dignac et al. 2005). Since lignocellulose, a major component of green waste, is known to be slowly degradable, slowing the composting process (Jurado et al. 2015), stable phenolic compounds and complex aromatic compounds may be used as indicators of stability (Fukushima et al. 2009, Wang et al. 2017). Wei et al. (2007) and Manu et al. (2017) suggested that higher contents of aliphatic carbon chains and carboxyl and hydroxyl groups in the compost produced with biological additives compared to regular compost point to a more stable product. They also observed decreased contributions of cellulose, hemicellulose and lignin in the co-compost compared to the regular compost, indicating that organic matter transformation was facilitated by the presence of a microbial inoculum. Mineral additives may enhance organic matter degradation during composting. For example, Zorpas and Loizidou (2008) showed that the addition of sawdust and zeolite during sewage sludge composting increased the percentage of NaOH-extractable compounds in the final products and thus the number of oxygen-containing functional groups. Finally, organic additives in the form of biochar during poultry manure composting also increased the degree of transformation of the compost (Dias et al. 2010).
Soil can retain carbon for a long time, thus avoiding its mineralisation and release in the form of CO2 or CH4. Soil contains about three times more carbon than the atmosphere (Eglin et al. 2010), and carbon storage in soils might be one of the solutions to mitigate climate change (IPCC 2014; Lal et al. 2004). This storage may be favoured by agricultural practices adapted to soil and climate conditions (Dignac et al. 2017). Managing organic waste materials and returning their organic C to soil may increase the soils’ C sequestration potential in the sense of a circular economy (Chabbi et al. 2017). One strategy to increase soil carbon stocks is the transformation of organic waste through composting and the application of composts or vermicomposts to soils (Ngo et al. 2012). However, these organic amendments show a very fast degradation in soils due to the presence of large amounts of labile organic compounds, consumed by the soil microbial biomass, leading to increased carbon emissions. Favoino and Hogg (2008), using first-order decay kinetics, estimated that between 2 and 10% of the compost carbon applied to soil today would remain in the soil organic matter in a hundred years. However, this estimation depends largely on the type of soil (Busby et al. 2007), on the pedoclimatic conditions and on the type of compost (Peltre et al. 2012). For instance, the mineralisation process in temperate countries takes place rather slowly, while in tropical countries, the mineralisation generally leads to a rapid decline of the added compost. In Table 3, we estimated the amount of carbon potentially remaining in soil after application of various co-composts, based on the estimation of Favoino and Hogg (2008).
Reducing the amount of labile carbon through carbon stabilisation in composts and vermicomposts recently received attention (Bolan et al. 2012; Chowdhury et al. 2014; Barthod et al. 2016). To improve carbon stabilisation in the organic amendments, the addition of minerals, such as goethite, montmorillonite or industrial wastes, such as red mud, was used. Some authors observed that the presence of these minerals during composting allowed to increase the half-life of organic amendment carbon and thus increase soil carbon stocks without altering soil properties (Bolan et al. 2012; Chowdhury et al. 2016). The addition of biochar to vermicompost changed the biodegradation of vermicompost organic matter after addition to soil and might thus increase its potential for C sequestration (Ngo et al. 2013). However, little is known about the C stabilisation mechanisms occurring during composting with these minerals and even less is known about the mechanisms occurring during vermicomposting (Barthod et al. 2016). The main hypothesis proposed in these studies is that carbon from fresh organic matter used for composting and vermicomposting is stabilised through similar mechanisms as those occurring in soils. Further studies including the assessment of GHG emissions during composting and after application to soil are necessary to finally conclude on the global C balance.
6.3 Feasibility of co-composting at large scale: economical and practical aspects of co-composting and co-vermicomposting
Composting and vermicomposting processes are both suitable to produce valuable soil amendments, but their economic potential is dependent on the initial cost, the production volume, the initial quality of the feedstock and the end product prices at a particular location (Lim et al. 2016). Lim et al. (2016) estimated that a composting facility costs about 4.37 million USD annually (mainly related to initial investment and annual operation and maintenance costs) and generates 1.10 million USD of benefits per year. Since composting is a time-consuming process (Onwosi et al. 2017), using additives to shorten composting duration can reduce the costs.
Screening and re-using additives is not cost-effective (Villasenor et al. 2011). The additives must thus be cheap and efficient for producing a high-quality end product. For instance, the composting process can be improved by jaggery and polyethylene glycol, which are expensive (Gabhane et al. 2012), but also by bentonite (Li et al. 2012) or allophane (Bolan et al. 2012), which are cheap and abundant. In addition, the optimal additive to organic waste ratio must be optimised in order to reduce the cost of composting. For instance, compared to 3:1 proportions (sawdust/sludge), a 1:1 proportion was efficient and economic (Banegas et al. 2007). However, at low sawdust proportions, the final product had a lower quality, inducing, for example, a low germination index (Huang et al. 2004). Moreover, with low sawdust proportions, a longer composting process is required compared to high sawdust proportions.
Finally, while selecting additives, several trade-offs have to be considered. Taking into account the additive cost, spatial and temporal accessibility (e.g. agricultural residues collected seasonally) and abundance, the composting process with additives must be adapted to the region, the composting facility localisation and the season. Moreover, vermicomposting also generates revenue through excess worm biomass and improved end products as compared to a regular compost (Gajalakshmi and Abbasi 2002; Ngo et al. 2011; Doan et al. 2015). Thus, we suggest that the presence of worm, combined with an additive, will significantly improve the composting process by increasing nutrient availability, stabilising carbon, shortening compost duration and decreasing GHG emissions (Komakech et al. 2015). Therefore, this process may lead to higher return on investment and lower annual cost than a regular composting with or without additives (Fig. 4). However, to account for the economic advantages and disadvantages of the different composting procedures, a full economic analysis is needed. For example, while vermicomposting may reduce the cost of pile turning, the space required to treat a similar amount of organic wastes may be much higher than in traditional composting units, thus increasing costs. Moreover, vermicompost may not sanitise the wastes and additional composting may be required.
7 Conclusion
This review examined the effect of mineral, organic and biological additives during composting and vermicomposting procedures on the process parameters, the quality of the final products and their impact on soil ecosystem services (production of food and fibre through improved soil fertility, climate change mitigation through C storage in soil organic matter, etc.). Adding mineral, organic or biological materials during composting and vermicomposting affects composting parameters and may be used to (1) influence the key parameters of the composting process, such as aeration and porosity of the pile, and composting duration by manipulating the thermophilic period; (2) enhance the agronomic value of the final products by increasing nutrient contents and reducing metal mobility or increasing carbon stability; and (3) limit odour and GHG emissions. However, different additives have contrasting effects and costs and should therefore be used appropriately. After optimisation of co-composting processes, co-compost application to soil may improve soil health. On polluted soils, it appears relevant to use compost produced with an additive able to immobilise or reduce the availability of heavy metals, while on degraded soils, using a co-compost produced with organic or mineral additives rich in available nutrients might improve soil fertility. While regular composts are rapidly mineralised after soil application, co-compost application may increase the soil C stabilisation potential. However, negative effects of some co-composts on plant growth were also reported.
Further studies are needed to complete the information on the effects of additives:
-
The use of additives during composting leads to end products with contrasting properties, impacting differently the soil properties. Thus, in a view of an ecological application, more studies are needed to establish a direct link between co-compost properties, soil parameters and effects on plant growth, according to the species in order to improve soil health while avoiding environmental risks.
-
The quality and quantity of the additives should be considered in order to improve the cost-effectiveness of the system. The development of a cost-effective system based on co-composting or co-vermicomposting should be investigated. According to the available literature, vermicomposting is less expensive than composting and the final products generally show a higher quality than a regular compost. Therefore, improvement of the environmental impact of the vermicomposting systems should be assessed after the addition of organic, mineral or biological materials.
-
Ultimately, region-specific transformation processes should be designed depending on the availability of organic wastes and additives, taking into consideration the availability of organic wastes and additives as well as prevailing pedoclimatic conditions.
References
Alavi N, Daneshpajou M, Shirmardi M, Goudarzi G, Neisi A, Babaei AA (2017) Investigating the efficiency of co-composting and vermicomposting of vinasse with the mixture of cow manure wastes, bagasse, and natural zeolite. Waste Manag 69:17–126. https://doi.org/10.1016/j.wasman.2017.07.039
Alburquerque JA, Gonzálvez J, García D, Cegarra J (2006) Effects of bulking agent on the composting of ‘‘alperujo’’, the solid by-product of the two-phase centrifugation method for olive oil extraction. Process Biochem 41:127–132. https://doi.org/10.1016/j.procbio.2005.06.006
Amlinger F, Peyr S, Cuhls C (2008) Green house gas emissions from composting and mechanical biological treatment. Waste Manag Res 26:47–60. https://doi.org/10.1177/0734242X07088432
An CJ, Huang GH, Yao Y, Sun W, An K (2012) Performance of in-vessel composting of food waste in the presence of coal ash and uric acid. J Hazard Mater 203:38–45. https://doi.org/10.1016/j.jhazmat.2011.11.066
Andersen JK, Boldrin A, Christensen TH, Scheutz C (2011) Mass balances and life cycle inventory of home composting of organic waste. Waste Manag 31:1934–1942. https://doi.org/10.1016/j.wasman.2011.05.004
Awasthi MK, Wang Q, Huang H, Ren X, Lahori AH, Mahar A, Ali A, Shen F, Li R, Zhang Z (2016b) Influence of zeolite and lime as additives on greenhouse gas emissions and maturity evolution during sewage sludge composting. Bioresour Technol 216:172–181. https://doi.org/10.1016/j.biortech.2016.05.065
Awasthi MK, Wang Q, Huang H, Li R, Shen F, Lahori AH, Wang P, Guo D, Guo Z, Jiang S, Zhang Z (2016a) Effect of biochar amendment on greenhouse gas emission and bio-availability of heavy metals during sewage sludge co-composting. J Clean Prod 135:829–835. https://doi.org/10.1016/j.jclepro.2016.07.008
Awasthi MK, Selvam A, Lai KM, Wong JWC (2017) Critical evaluation of post-consumption food waste composting employing thermophilic bacterial consortium. Bioresour Technol 245:665–672. https://doi.org/10.1016/j.biortech.2017.09.014
Awasthi MK, Wang Q, Chen H, Awasthi SK, Wang M, Ren X, Zhao J, Zhang Z (2018) Beneficial effect of mixture of additives amendment on enzymatic activities, organic matter degradation and humification during biosolids co-composting. Bioresour Technol 247:138–146. https://doi.org/10.1016/j.biortech.2017.09.061
Banegas V, Moreno JL, Moreno JI, Garcia C, Leon G, Hernandez T (2007) Composting anaerobic and aerobic sewage sludges using two proportions of sawdust. Waste Manag 27:1317–1327. https://doi.org/10.1016/j.wasman.2006.09.008
Barthod J, Rumpel C, Paradelo R, Dignac M-F (2016) The effects of worms, clay and biochar on CO2 emissions during production and soil application of co-composts. Soil 2:673–683. https://doi.org/10.5194/soil-2-673-2016
Beck-Friis B, Pell M, Sonesson U, Jönsson H, Kirchmann H (2000) Formation and emission of N2O and CH4 from compost heaps of organic household waste. Environ Monit Assess 62:317–331. https://doi.org/10.1023/A:1006245227491
Belyaeva ON, Haynes RJ (2009) Chemical, microbial and physical properties of manufactured soils produced by co-composting municipal green waste with coal fly ash. Bioresour Technol 100:5203–5209. https://doi.org/10.1016/j.biortech.2009.05.032
Belyaeva ON, Haynes RJ (2012) Use of inorganic wastes as immobilizing agents for soluble P in green waste-based composts. Environ Sci Pollut Res Int 19:2138–2150. https://doi.org/10.1007/s11356-011-0713-z
Bernal MP, Lopez-Real JM, Scott KM (1993) Application of natural zeolites for the reduction of ammonia emissions during the composting of organic wastes in a laboratory composting simulator. Bioresour Technol 43:35–39. https://doi.org/10.1016/0960-8524(93)90079-Q
Bernal MP, Navarro AF, Sanchez-Monedero MA, Roig A, Cegarra J (1998) Influence of sewage sludge compost stability and maturity on carbon and nitrogen mineralization in soil. Soil Biol Biochem 30:305–313. https://doi.org/10.1016/S0038-0717(97)00129-6
Bernal MP, Alburquerque JA, Moral R (2009) Composting of animal manures and chemical criteria for compost maturity assessment. A review. Bioresour Technol 100:5444–5453. https://doi.org/10.1016/j.biortech.2008.11.027
Bernstad A, Schott S, Wenzel H, la Cour JJ (2016) Identification of decisive factors for greenhouse gas emissions in comparative life cycle assessments of food waste management—an analytical review. J Clean Prod 119:13–24. https://doi.org/10.1016/j.jclepro.2016.01.079
Billah M, Bano A (2015) Role of plant growth promoting rhizobacteria in modulating the efficiency of poultry litter composting with rock phosphate and its effect on growth and yield of wheat. Waste Manag Res 33:63–72. https://doi.org/10.1177/0734242X14559593
Bolan NS, Kunhikrishnan A, Choppala GK, Thangarajan R, Chung JW (2012) Stabilization of carbon in composts and biochars in relation to carbon sequestration and soil fertility. Sci Tot Environ 424:264–270. https://doi.org/10.1016/j.scitotenv.2012.02.061
Bouché MB (1977) Strategies lombriciennes. Ecol Bull:122–132
Bougnom BP, Mair J, Etoa FX, Insam H (2009) Composts with wood ash addition: a risk or a chance for ameliorating acid tropical soils? Geoderma 153:402–407. https://doi.org/10.1016/j.geoderma.2009.09.003
Busato JG, Lima LS, Aguiar NO, Canellas LP, Olivares FL (2012) Changes in labile phosphorus forms during maturation of vermicompost enriched with phosphorus-solubilizing and diazotrophic bacteria. Bioresour Technol 110:390–395. https://doi.org/10.1016/j.biortech.2012.01.126
Busby RR, Torbert HA, Gebhart DL (2007) Carbon and nitrogen mineralization of non-composted and composted municipal solid waste in sandy soils. Soil Biol Biochem 39:1277–1283. https://doi.org/10.1016/j.soilbio.2006.12.003
Chabbi A, Lehmann J, Ciais P, Loescher HW, Cotrufo MF, Don A, SanClements M, Schipper L, Six J, Smith P, Rumpel C (2017) Aligning agriculture and climate policy. Nat Clim Chang 7:307–309. https://doi.org/10.1038/nclimate3286
Calabi-Floody M, Medina J, Rumpel C, Condron LM, Hernandez M, Dumont M, Mora ML (2017) Smart fertilizers as a strategy for sustainable agriculture. Advances in Agronomy. https://doi.org/10.1016/bs.agron.2017.10.003
Ceglie FG, Bustamante MA, Ben Amara M, Tittarelli F (2015) The challenge of peat substitution in organic seedling production: optimization of growing media formulation through mixture design and response surface analysis. PLoS One 10(6):e0128600. https://doi.org/10.1371/journal.pone.0128600
Cerda A, Artola A, Font X, Barrena R, Gea T, Sánchez A (2018) Composting of food wastes: Status and challenges. Bioresour Technol 248A:57–67. https://doi.org/10.1016/j.biortech.2017.06.133
Chan YC, Sinha RK, Wang WJ (2011) Emission of greenhouse gases from home aerobic composting, anaerobic digestion and vermicomposting of household wastes in Brisbane (Australia). Waste Manag Res 29:540–548. https://doi.org/10.1177/0734242X10375587
Chang JI, Chen YJ (2010) Effects of bulking agents on food waste composting. Bioresour Technol 101:5917–5924. https://doi.org/10.1016/j.biortech.2010.02.042
Chen YX, Huang XD, Han ZY, Huang X, Hu B, Shi DZ, Wu WX (2010) Effects of bamboo charcoal and bamboo vinegar on nitrogen conservation and heavy metals immobility during pig manure composting. Chemosphere 78:1177–1181. https://doi.org/10.1016/j.chemosphere.2009.12.029
Chen Z, Zhang S, Wen Q, Zheng J (2015) Effect of aeration rate on composting of penicillin mycelial dreg. J Environ Sci 37:172–178. https://doi.org/10.1016/j.jes.2015.03.020
Chowdhury MA, de Neergaard A, Jensen LS (2014) Potential of aeration flow rate and bio-char addition to reduce greenhouse gas and ammonia emissions during manure composting. Chemosphere 97:16–25. https://doi.org/10.1016/j.chemosphere.2013.10.030
Chowdhury S, Bolan NS, Seshadri B, Kunhikrishnan A, Wijesekara H, Xu Y, Rumpel C (2016) Co-composting solid biowastes with alkaline materials to enhance carbon stabilization and revegetation potential. Environ Sci Poll Res 23:7099–7110. https://doi.org/10.1007/s11356-015-5411-9
Czekała W, Malińska K, Cáceres R, Janczak D, Dach J, Lewicki A (2016) Co-composting of poultry manure mixtures amended with biochar—the effect of biochar on temperature and C-CO2 emission. Bioresour Technol 200:921–927. https://doi.org/10.1016/j.biortech.2015.11.019
de Bertoldi MD, Vallini GE, Pera A (1983) The biology of composting: a review. Waste Manag Res 1:157–176. https://doi.org/10.1016/0734-242X(83)90055-1
Diacono M, Montemurro F (2010) Long-term effects of organic amendments on soil fertility. A review. Agron Sustain Develop 30:401–422. https://doi.org/10.1051/agro/2009040
Dias BO, Silva CA, Higashikawa FS, Roig A, Sánchez-Monedero MA (2010) Use of biochar as bulking agent for the composting of poultry manure: effect on organic matter degradation and humification. Bioresour Technol 101:1239–1246. https://doi.org/10.1016/j.biortech.2009.09.024
Dignac M-F, Houot S, Francou C, Derenne S (2005) Pyrolytic study of compost and waste organic matter. Org Geochem 36:1054–1071. https://doi.org/10.1016/j.orggeochem.2005.02.007
Dignac M-F, Derrien D, Barré P, Barot S, Cécillon L, Chenu C, Chevallier T, Freschet GT, Garnier P, Guenet B, Hedde M, Klumpp K, Lashermes G, Maron P-A, Nunan N, Roumet C, Basile-Doelsch I (2017) Increasing soil carbon storage: mechanisms, effects of agricultural practices and proxies. A review. Agron Sustain Dev 37:14. https://doi.org/10.1007/s13593-017-0421-2
Doan TT, Henry-des-Tureaux T, Rumpel C, Janeau JL, Jouquet P (2015) Impact of compost, vermicompost and biochar on soil fertility, maize yield and soil erosion in Northern Vietnam: a three year mesocosm experiment. Sci Tot Environ 514:147–154. https://doi.org/10.1016/j/scitotenv.2015.02.005
Domínguez J, Edwards CA (2010) Chapter 3. Biology and ecology of earthworm species used for vermicomposting. In: Edwards CA, Arancon NQ, Sherman R (eds) Vermiculture technology: earthworms, organic waste and environmental management. pp 25-37. https://doi.org/10.1201/b10453-4
Doublet J, Francou C, Poitrenaud M, Houot S (2011) Influence of bulking agents on organic matter evolution during sewage sludge composting; consequences on compost organic matter stability and N availability. Bioresour Technol 102:1298–1307. https://doi.org/10.1016/j.biortech.2010.08.065
Eglin T, Ciais P, Piao SL, Barre P, Bellassen V, Cadule P, Chenu C, Gasser T, Koven C, Reichstein M, Smith P (2010) Historical and future perspectives of global soil carbon response to climate and land-use changes. Tellus-B 62:700–718. https://doi.org/10.1111/j.1600-0889.2010.00499.x
Eftoda G, McCartney D (2004) Determining the critical bulking agent requirement for municipal biosolids composting. Compost Sci Util 12:208–218
Ermolaev E, Jarvis A, Sundberg C, Smars S, Pell M, Jönsson H (2015) Nitrous oxide and methane emissions from food waste composting at different temperatures. Waste Manag 46:113–119. https://doi.org/10.1016/j.wasman.2015.08.021
Fang M, Wong JWC, Ma KK, Wong MH (1999) Co-composting of sewage sludge and coal fly ash: nutrient transformations. Bioresour Technol 67:19–24
Fang M, Wong JWC (1999) Effects of lime amendment on availability of heavy metals and maturation in sewage sludge composting. Environ Poll 106:83–89
Favoino E, Hogg D (2008) The potential role of compost in reducing greenhouse gases. Waste Manag Res 26:61–69
Francou C, Poitrenaud M, Houot S (2005) Stabilization of organic matter during composting: influence of process and feedstocks. Compost Sci Util 13:72–83
Frederickson J, Butt KR, Morris RM, Daniel C (1997) Combining vermiculture with traditional green waste composting systems. Soil Biol Biochem 29:725–730. https://doi.org/10.1016/S0038-0717(96)00025-9
Frederickson J, Howell G, Hobson AM (2007) Effect of pre-composting and vermicomposting on compost characteristics. Europ Journal Soil Biol 43:S320–S326
Fuchs JG (2009) Interactions between beneficial and harmful microorganisms: from the composting process to compost application. In: Microbes at work: from wastes to resources. pp 213-229.
Fukushima M, Yamamoto K, Ootsuka K, Komai T, Aramaki T, Ueda S, Horiya S (2009) Effects of the maturity of wood waste compost on the structural features of humic acids. Bioresour Technol 100:791–797
Gabhane J, William SP, Bidyadhar R, Bhilawe P, Anand D, Vaidya AN, Wate SR (2012) Additives aided composting of green waste: effects on organic matter degradation, compost maturity, and quality of the finished compost. Bioresour Technol 114:382–388
Gajalakshmi S, Abbasi SA (2002) Effect of the application of water hyacinth compost/vermicompost on the growth and flowering of Crossandra undulaefolia, and on several vegetables. Bioresour Technol 85:197–199. https://doi.org/10.1016/S0960-8524(02)00096-2
Gao M, Li B, Yu A, Liang F, Yang L, Sun Y (2010) The effect of aeration rate on forced-aeration composting of chicken manure and sawdust. Bioresour Technol 101:1899–1903. https://doi.org/10.1016/j.biortech.2009.10.027
Garg P, Gupta A, Satya S (2006) Vermicomposting of different types of waste using Eisenia foetida: a comparative study. Bioresour Technol 97:391–395. https://doi.org/10.1016/j.biortech.2005.03.009
Gea T, Barrena R, Artola A, Sánchez A (2007) Optimal bulking agent particle size and usage for heat retention and disinfection in domestic wastewater sludge composting. Waste Manag 27:1108–1116. https://doi.org/10.1016/j.wasman.2006.07.005
Gomes HI, Mayes WM, Rogerson M, Stewart DI, Burke IT (2016) Alkaline residues and the environment: a review of impacts, management practices and opportunities. Journal Cleaner Production 112:3571–3582. https://doi.org/10.1016/j.jclepro.2015.09.111
Goyal S, Dhull SK, Kapoor KK (2005) Chemical and biological changes during composting of different organic wastes and assessment of compost maturity. Bioresour Technol 96:1584–1591. https://doi.org/10.1016/j.biortech.2004.12.012
Guerra-Rodríguez E, Alonso J, Melgar MJ, Vázquez M (2006) Evaluation of heavy metal contents in co-composts of poultry manure with barley wastes or chestnut burr/leaf litter. Chemosphere 65:1801–1805. https://doi.org/10.1016/j.chemosphere.2006.04.023
Hayawin ZN et al (2014) Study on the effect of adding zeolite and other bio-adsorbance in enhancing the quality of the palm based vermicompost. App Mechan Mat 472:926–934. https://doi.org/10.4028/www.scientific.net/AMM.472.926
Hao X, Chang C, Larney FJ, Travis GR (2001) Greenhouse gas emissions during cattle feedlot manure composting. J Environ Qual 30:376–386. https://doi.org/10.1007/s10705-006-9083-1
Hao X, Larney FJ, Chang C, Travis GR, Nichol CK, Bremer E (2005) The effect of phosphogypsum on greenhouse gas emissions during cattle manure composting. J Environ Qual 34:774–781. https://doi.org/10.2134/jeq2004.0388
Hashemimajd K, Kalbasi M, Golchin A, Shariatmadari H (2004) Comparison of vermicompost and composts as potting media for growth of tomatoes. Journal Plant Nut 27:1107–1123. https://doi.org/10.1081/PLN-120037538
Haug RT (1993) The practical handbook of compost engineering. CRC Press, Boca Raton
Haynes RJ, Zhou YF (2015) Use of alum water treatment sludge to stabilize C and immobilize P and metals in composts. Environ Sci Pollut Res 22:13903–13914. https://doi.org/10.1007/s11356-015-4517-4
He X, Zhang Y, Shen M, Zeng G, Zhou M, Li M (2016) Effect of vermicomposting on concentration and speciation of heavy metals in sewage sludge with additive materials. Bioresour Technol 218:867–873. https://doi.org/10.1016/j.biortech.2016.07.045
Himanen M, Hänninen K (2009) Effect of commercial mineral-based additives on composting and compost quality. Waste Manag 29:2265–2273. https://doi.org/10.1016/j.wasman.2009.03.016
Himanen M, Hänninen K (2011) Composting of bio-waste, aerobic and anaerobic sludges—effect of feedstock on the process and quality of compost. Bioresour Technol 102:2842–2852. https://doi.org/10.1016/j.biortech.2010.10.059
Hinsinger P (2001) Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review. Plant Soil 237:173–195. https://doi.org/10.1023/A:1013351617532
Hobson AM, Frederickson J, Dise NB (2005) CH4 and N2O from mechanically turned windrow and vermicomposting systems following in-vessel pre-treatment. Waste Manag 25:345–352. https://doi.org/10.1016/j.wasman.2005.02.015
Hoornweg D, Bhada-Tata P, Kennedy C, (2013) Environment: waste production must peak this century. Nature 502:615–617. https://doi.org/10.1038/502615a
Hua L, Wu W, Liu Y, McBride MB, Chen Y (2009) Reduction of nitrogen loss and Cu and Zn mobility during sludge composting with bamboo charcoal amendment. Environ Sci Poll Res 16:1–9. https://doi.org/10.1007/s11356-008-0041-0
Huang GF, Wong JWC, Wu QT, Nagar BB (2004) Effect of C/N on composting of pig manure with sawdust. Waste Manag 24:805–813. https://doi.org/10.1016/j.wasman.2004.03.011
Insam H, de Bertoli M (2007) Chapter 3. Microbiology of the composting process. Waste Manag Series 8:25–48. https://doi.org/10.1016/S1478-7482(07)80006-6
IPCC (2014) Climate change 2014. Synthesis Report.
Iqbal MK, Shafiq T, Ahmed K (2010) Characterization of bulking agents and its effects on physical properties of compost. Bioresour Technol 101:1913–1919. https://doi.org/10.1016/j.biortech.2009.10.030
Iqbal MK, Nadeem A, Sherazi F, Khan RA (2015b) Optimization of process parameters for kitchen waste composting by response surface methodology. Int J Environ Sci Technol 12:1759–1768. https://doi.org/10.1007/s13762-014-0543-x
Iqbal H, Garcia-Perez M, Flury M (2015a) Effect of biochar on leaching of organic carbon, nitrogen, and phosphorus from compost in bioretention systems. Sci Tot Environ 521:37–45. https://doi.org/10.1016/j.scitotenv.2015.03.060
Jeong YK, Hwang SJ (2005) Optimum doses of Mg and P salts for precipitating ammonia into struvite crystals in aerobic composting. Bioresour Technol 96:1–6. https://doi.org/10.1016/j.biortech.2004.05.028
Jiang J, Liu X, Huang Y, Huang H (2015a) Inoculation with nitrogen turnover bacterial agent appropriately increasing nitrogen and promoting maturity in pig manure composting. Waste Manag 39:78–85. https://doi.org/10.1016/j.wasman.2015.02.025
Jiang T, Li G, Tang Q, Ma X, Wang G, Schuchardt F (2015b) Effects of aeration method and aeration rate on greenhouse gas emissions during composting of pig feces in pilot scale. J Environ Sci 31:124–132. https://doi.org/10.1016/j.jes.2014.12.005
Jiang T, Ma X, Tang Q, Li G, Schuchardt F (2016) Combined use of nitrification inhibitor and struvite crystallization to reduce the NH3 and N2O emissions during composting. Bioresour Technol 217:210–218. https://doi.org/10.1016/j.biortech.2016.01.089
Jolanun B, Towprayoon S, Chiemchaisri C (2008) Aeration improvement in fed batch composting of vegetable and fruit wastes. Environ Prog 27:250–256. https://doi.org/10.1002/ep.10259
Juarez MFD, Prähauser B, Walter A, Insam H, Franke-Whittle IH (2015) Co-composting of biowaste and wood ash, influence on a microbially driven-process. Waste Manag 46:155–164. https://doi.org/10.1016/j.wasman.2015.09.015
Jurado MM, Suarez-Estrella F, Lopez MJ, Vargas-Garcia MC, Lopez-Gonzalez JA, Moreno J (2015) Enhanced turnover of organic matter fractions by microbial stimulation during lignocellulosic waste composting. Bioresour Technol 186:15–24. https://doi.org/10.1016/j.biortech.2015.03.059
Kato K, Miura N (2008) Effect of matured compost as a bulking and inoculating agent on the microbial community and maturity of cattle manure compost. Bioresour Technol 99:3372–3380. https://doi.org/10.1016/j.biortech.2007.08.019
Kaushik P, Garg VK (2003) Vermicomposting of mixed solid textile mill sludge and cow dung with the epigeic earthworm Eisenia foetida. Bioresour Technol 90:311–316. https://doi.org/10.1016/S0960-8524(03)00146-9
Khan N, Clark I, Sánchez-Monedero MA, Shea S, Meier S, Bolan N (2014) Maturity indices in co-composting of chicken manure and sawdust with biochar. Bioresour Technol 168:245–251. https://doi.org/10.1016/j.biortech.2014.02.123
Klamer M, Bååth E (1998) Microbial community dynamics during composting of straw material studied using phospholipid fatty acid analysis. FEMS Microb Ecol 27:9–20. https://doi.org/10.1111/j.1574-6941.1998.tb00521.x
Koivula N, Räikkönen T, Urpilainen S, Ranta J, Hänninen K (2004) Ash in composting of source-separated catering waste. Bioresour Technol 93:291–299. https://doi.org/10.1016/j.biortech.2003.10.025
Komakech AJ, Sundberg C, Jönsson H, Vinnerås B (2015) Life cycle assessment of biodegradable waste treatment systems for sub-Saharan African cities. Resour Conserv Recycl 99:100–110. https://doi.org/10.1016/j.resconrec.2015.03.006
Kuba T, Tschöll A, Partl C, Meyer K, Insam H (2008) Wood ash admixture to organic wastes improves compost and its performance. Agri Ecosys Environ 127:43–49. https://doi.org/10.1016/j.agee.2008.02.012
Kulcu R, Yaldiz O (2007) Composting of goat manure and wheat straw using pine cones as a bulking agent. Bioresour Technol 98:2700–2704. https://doi.org/10.1016/j.biortech.2006.09.025
Kurien J, Ramasamy EV (2006) Vermicomposting of taro (Colocasia esculenta) with two epigeic earthworm species. Bioresour Technol 97:1324–1328. https://doi.org/10.1016/j.biortech.2005.05.018
Lal R, Griffin M, Apt J, Lave L, Morgan MG (2004) Managing soil carbon. Science 304:393. https://doi.org/10.1126/science.1093079
Lashermes G, Nicolardot B, Parnaudeau V, Thuriès L, Chaussod R, Guillotin ML, Linères M, Mary B, Metzger L, Morvan T, Tricaud A, Villette C, Houot S (2009) Indicator of potential residual carbon in soils after exogenous organic matter application. Eur J Soil Sci 60:297–310. https://doi.org/10.1111/j.1365-2389.2008.01110.x
Lau SSS, Fang M, Wong JWC (2001) Effects of composting process and fly ash amendment on phytotoxicity of sewage sludge. Arch Environ Contam Toxicol 40:184–191. https://doi.org/10.1007/s002440010162
Lefcourt AM, Meisinger JJ (2001) Effect of adding alum or zeolite to dairy slurry on ammonia volatilization and chemical composition. J Dairy Sci 84:1814–1821. https://doi.org/10.3168/jds.S0022-0302(01)74620-6
Lehmann J, Gaunt J, Rondon M (2006) Biochar sequestration in terrestrial ecosystems—a review. Mitig Adapt Strat Glob Change 11:395–419. https://doi.org/10.1007/s11027-005-9006-5
Lehmann J (2007) A handful of carbon. Nature 447:143–144. https://doi.org/10.1038/447143a
Lehmann J, Joseph S (Eds.) (2015) Biochar for environmental management: science, technology and implementation. Taylor and Francis.
Li H, Xin H, Liang Y, Burns RT (2008) Reduction of ammonia emissions from stored laying hen manure through topical application of zeolite, Al+Clear, Ferix-3, or poultry litter treatment. J App Poultry Res 17:421–431. https://doi.org/10.3382/japr.2007-00076
Li L, Wu J, Tian G, Xu Z (2009) Effect of the transit through the gut of earthworm (Eisenia fetida) on fractionation of Cu and Zn in pig manure. J Hazard Mater 167:634–640. https://doi.org/10.1016/j.jhazmat.2009.01.013
Li R, Wang JJ, Zhang Z, Shen F, Zhang G, Qin R, Li X, Xiao R (2012) Nutrient transformations during composting of pig manure with bentonite. Bioresour Technol 121:362–368. https://doi.org/10.1016/j.biortech.2012.06.065
Lim SL, Lee LH, Wu TY (2016) Sustainability of using composting and vermicomposting technologies for organic solid waste biotransformation: recent overview, greenhouse gases emissions and economic analysis. Journal Cleaner Production 111:262–278. https://doi.org/10.1016/j.jclepro.2015.08.083
Liu J, Xu XH, Li HT, Xu Y (2011) Effect of microbiological inocula on chemical and physical properties and microbial community of cow manure compost. Biomass Energ 35:3433–3439. https://doi.org/10.1016/j.biombioe.2011.03.042
Liu N, Zhou J, Han L, Ma S, Sun X, Huang G (2017) Role and multi-scale characterization of bamboo biochar during poultry manure aerobic composting. Bioresour Technol 241:190–199. https://doi.org/10.1016/j.biortech.2017.03.144
Lopez E, Soto B, Arias M, Nunez A, Rubinos D, Barral MT (1998) Adsorbent properties of red mud and its use for wastewater treatment. Water Res 32:1314–1322. https://doi.org/10.1016/S0043-1354(97)00326-6
Lu D, Wang L, Yan B, Ou Y, Guan J, Bian Y, Zhang Y (2014) Speciation of Cu and Zn during composting of pig manure amended with rock phosphate. Waste Manag 34:1529–1536. https://doi.org/10.1016/j.wasman.2014.04.008
Lü DA, Yan BX, Wang LX, Deng ZQ, Zhang YB (2013) Changes in phosphorus fractions and nitrogen forms during composting of pig manure with rice straw. J Integr Agric 12:1855–1864. https://doi.org/10.1016/S2095-3119(13)60400-1
Lucas RE, Davis J (1961) Relationships between pH values of organic soils and availabilities of 12 plant nutrients. Soil Sci 92:177–182
Luo WH, Yuan J, Luo YM, Li GX, Nghiem LD, Price WE (2014) Effects of mixing and covering with mature compost on gaseous emissions during composting. Chemosphere 117:14–19. https://doi.org/10.1016/j.chemosphere.2014.05.043
Luo Y, Li G, Luo W, Schuchardt F, Jiang T, Xu D (2013) Effect of phosphogypsum and dicyandiamide as additives on NH3, N2O and CH4 emissions during composting. J Environ Sci 25:1338–1345
Mahimairaja S, Bolan NS, Hedley MJ, Macgregor AN (1994) Losses and transformation of nitrogen during composting of poultry manure with different amendments: an incubation experiment. Bioresour Technol 47:265–273. https://doi.org/10.1016/0960-8524(94)90190-2
Malińska K, Zabochnicka-Świątek M, Cáceres R, Marfà O (2016) The effect of precomposted sewage sludge mixture amended with biochar on the growth and reproduction of Eisenia fetida during laboratory vermicomposting. Ecol Eng 90:35–41. https://doi.org/10.1016/j.ecoleng.2016.01.042
Malińska K, Golanska M, Caceres R, Rorat A, Weisser P, Ślęzak E (2017) Biochar amendment for integrated composting and vermicomposting of sewage sludge—the effect of biochar on the activity of Eisenia fetida and the obtained vermicompost. Bioresour Technol 225:206–214. https://doi.org/10.1016/j.biortech.2016.11.049
Manu MK, Kumar R, Garg A (2017) Performance assessment of improved composting system for food waste with varying aeration and use of microbial inoculum. Bioresour Technol 234:167–177. https://doi.org/10.1016/j.biortech.2017.03.023
Marshall RE, Farahbakhsh K (2013) Systems approaches to integrated solid waste management in developing countries. Waste Manag 33:988–1003. https://doi.org/10.1016/j.wasman.2012.12.023
Maulini-Duran C, Artola A, Font X, Sánchez A (2014) Gaseous emissions in municipal wastes composting: effect of the bulking agent. Bioresour Technol 172:260–268. https://doi.org/10.1016/j.biortech.2014.09.041
McCrory DF, Hobbs PJ (2001) Additives to reduce ammonia and odor emissions from livestock wastes. J Environ Qual 30:345–355. https://doi.org/10.2134/jeq2001.302345x
Meena MD, Biswas DR (2014) Phosphorus and potassium transformations in soil amended with enriched compost and chemical fertilizers in a wheat–soybean cropping system. Com Soil Sci Plant Analysis 45:624–652. https://doi.org/10.1080/00103624.2013.867044
Mehta CM, Palni U, Franke-Whittle IH, Sharma AK (2014) Compost: its role, mechanism and impact on reducing soil-borne plant diseases. Waste Manag 34:607–622. https://doi.org/10.1016/j.wasman.2013.11.012
Meng J, Wang L, Liu X, Wu J, Brookes PC, Xu J (2013) Physicochemical properties of biochar produced from aerobically composted swine manure and its potential use as an environmental amendment. Bioresour Technol 142:641–646. https://doi.org/10.1016/j.biortech.2013.05.086
Menon MP, Sajwan KS, Ghuman GS, James J, Chandra K (1993) Fly ash-amended compost as a manure for agricultural crops. J Environ Sci Health Part A 28:2167–2182. https://doi.org/10.1080/10934529309375999
Mihreteab HT, Ceglie FG, Aly A, Tittarelli F (2015) Rock phosphate enriched compost as a growth media component for organic tomato (Solanum lycopersicum L.) seedlings production. Biol Agri Horti 32:7–20. https://doi.org/10.1080/01448765.2015.1016114
Miner FD Jr, Koenig RT, Miller BE (2001) The influence of bulking material type and volume on in-house composting in high-rise, caged layer facilities. Compost Sci Util 9:50–59. https://doi.org/10.1080/1065657X.2001.10702016
Morales AB, Bustamante MA, Marhuenda-Egea FC, Moral R, Ros M, Pascual JA (2016) Agri-food sludge management using different co-composting strategies: study of the added value of the composts obtained. J Clean Prod 121:186–197. https://doi.org/10.1016/j.jclepro.2016.02.012
Morand P, Peres G, Robin P, Yulipriyanto H, Baron S (2005) Gaseous emissions from composting bark/manure mixtures. Compost Sci Util 13:14–26. https://doi.org/10.1080/1065657X.2005.10702213
Morisaki N, Phae CG, Nakasaki K, Shoda M, Kubota H (1989) Nitrogen transformation during thermophilic composting. Journal Fermenta Bioeng 67:57–61. https://doi.org/10.1016/0922-338X(89)90087-1
Nakasaki K, Shoda M, Kubota H (1986) Effects of a bulking agent on the reaction rate of thermophilic sewage sludge composting. J Ferment Technol 64:539–544. https://doi.org/10.1016/0385-6380(86)90079-8
Nakasaki K, Hirai H (2017) Temperature control strategy to enhance the activity of yeast inoculated into compost raw material for accelerated composting. Waste Manag 65:29–36. https://doi.org/10.1016/j.wasman.2017.04.019
Nasini L, De Luca G, Ricci A, Ortolani F, Caselli A, Massaccesi L, Regni L, Gigliotti G, Proietti P (2016) Gas emissions during olive mill waste composting under static pile conditions. Int Biodeterior Biodegrad 107:70–76. https://doi.org/10.1016/j.ibiod.2015.11.001
Ngo P-T, Rumpel C, Dignac M-F, Billou D, Tran Duc T, Jouquet P (2011) Transformation of buffalo manure by composting or vermicomposting to rehabilitate degraded tropical soils. Ecol Engineer 37:269–276. https://doi.org/10.1016/j.ecoleng.2010.11.011
Ngo P-T, Rumpel C, Doan T-T, Jouquet P (2012) The effect of earthworms on carbon storage and soil organic matter composition in tropical soil amended with compost and vermicompost. Soil Biol Biochem 50:214–220. https://doi.org/10.1016/j.soilbio.2012.02.037
Ngo P-T et al (2013) Biological and chemical reactivity and phosphorus forms of buffalo manure compost, vermicompost and their mixture with biochar. Bioresour Technol 148:401–407. https://doi.org/10.1016/j.biortech.2013.08.098
Nigussie A, Bruun S, de Neergaard A, Kuyper TW (2017) Earthworms change the quantity and composition of dissolved organic carbon and reduce greenhouse gas emissions during composting. Waste Manag 62:43–51. https://doi.org/10.1016/j.wasman.2017.02.009
Nishanth D, Biswas DR (2008) Kinetics of phosphorus and potassium release from rock phosphate and waste mica enriched compost and their effect on yield and nutrient uptake by wheat (Triticum aestivum). Bioresour Technol 99:3342–3353. https://doi.org/10.1016/j.biortech.2007.08.025
Nissen LR, Lepp NW, Edwards R (2000) Synthetic zeolites as amendments for sewage sludge-based compost. Chemosphere 41:265–269. https://doi.org/10.1016/S0045-6535(99)00420-8
Ogunwande GA, Osunade JA (2011) Passive aeration composting of chicken litter: effects of aeration pipe orientation and perforation size on losses of compost elements. J Environ Manag 92:85–91. https://doi.org/10.1016/j.jenvman.2010.08.026
Oliveira SM, Ferrera AS (2014) Change in soil microbial and enzyme activities in response to the addition of rock-phosphate-enriched compost. Com Soil Sci Plant Analysis 21:2794–2803. https://doi.org/10.1080/00103624.2014.954286
Onwosi CO, Igbokwe VC, Odimba JN, Eke IE, Nwankwoala MO, Iroh IN, Ezeogu LI (2017) Composting technology in waste stabilization: on the methods, challenges and future prospects. J Environ Manag 190:140–157. https://doi.org/10.1016/j.jenvman.2016.12.051
Pagans E, Barrena R, Font X, Sánchez A (2006) Ammonia emissions from the composting of different organic wastes. Dependency on process temperature. Chemosphere 62:1534–1542. https://doi.org/10.1016/j.chemosphere.2005.06.044
Pasda N, Limtong P, Oliver R, Montange D, Panichsakpatana S (2005) Influence of bulking agents and microbial activator on thermophilic aerobic transformation of sewage sludge. Environ Technol 26:1127–1136. https://doi.org/10.1080/09593332608618481
Peltre C, Christensen BT, Dragon S, Icard C, Kätterer T, Houot S (2012) RothC simulation of carbon accumulation in soil after repeated application of widely different organic amendments. Soil Biol Biochem 52:49–60. https://doi.org/10.1016/j.soilbio.2012.03.023
Qiao L, Ho G (1997) The effects of clay amendment and composting on metal speciation in digested sludge. Water Res 31:951–964. https://doi.org/10.1016/S0043-1354(96)00290-4
Reinecke AJ, Viljoen SA, Saayman RJ (1992) The suitability of Eudrilus eugeniae, Perionyx excavatus and Eisenia fetida (Oligochaeta) for vermicomposting in Southern Africa in terms of their temperature requirements. Soil Biol Biochem 24:1295–1307. https://doi.org/10.1016/0038-0717(92)90109-B
Ren L, Schuchardt F, Shen Y, Li G, Li C (2010) Impact of struvite crystallization on nitrogen losses during composting of pig manure and cornstalk. Waste Manag 30:885–892. https://doi.org/10.1016/j.wasman.2009.08.006
Richard TL, Hamelers HVM, Veeken A, Silva T (2002) Moisture relationships in composting processes. Compost Sci Util 10:286–302. https://doi.org/10.1080/1065657X.2002.10702093
Roca-Pérez L, Martinez C, Marcilla P, Boluda R (2009) Composting rice straw with sewage sludge and compost effects on the soil–plant system. Chemosphere 75:781–787. https://doi.org/10.1016/j.chemosphere.2008.12.058
Samaras P, Papadimitriou CA, Haritou I, Zouboulis AI (2008) Investigation of sewage sludge stabilization potential by the addition of fly ash and lime. J Hazard Mater 154:1052–1059. https://doi.org/10.1016/j.jhazmat.2007.11.012
Sánchez-García M, Alburquerque JA, Sánchez-Monedero MA, Roig A, Cayuela ML (2015) Biochar accelerates organic matter degradation and enhances N mineralisation during composting of poultry manure without a relevant impact on gas emissions. Bioresour Technol 192:272–279. https://doi.org/10.1016/j.biortech.2015.05.003
Sasaki H, Kitazume O, Nonaka J, Hikosaka K, Otawa K, Itoh K, Nakai Y (2006) Effect of a commercial microbiological additive on beef manure compost in the composting process. Animal Sci J 77:545–548. https://doi.org/10.1111/j.1740-0929.2006.00384.x
Senesil GS, Baldassarre G, Senesi N, Radina B (1999) Trace element inputs into soils by anthropogenic activities and implications for human health. Chemosphere 39:343–377. https://doi.org/10.1016/S0045-6535(99)00115-0
Shao LM, Zhang CY, Wu D, Lü F, Li TS, He PJ (2014) Effects of bulking agent addition on odorous compounds emissions during composting of OFMSW. Waste Manag 34:1381–1390. https://doi.org/10.1016/j.wasman.2014.04.016
Soares MA, Quina MJ, Reis MS, Quinta-Ferreira R (2017) Assessment of co-composting process with high load of an inorganic industrial waste. Waste Manag 59:80–89. https://doi.org/10.1016/j.wasman.2016.09.044
Soobhany N, Mohee R, Garg VK (2017) Inactivation of bacterial pathogenic load in compost against vermicompost of organic solid waste aiming to achieve sanitation goals: a review. Waste Manag 64:51–62. https://doi.org/10.1016/j.wasman.2017.03.003
Steiner C, Das KC, Melear N, Lakly D (2010) Reducing nitrogen loss during poultry litter composting using biochar. J Environ Qual 39:1236–1242. https://doi.org/10.2134/jeq2009.0337
Suthar S, Singh S (2008) Vermicomposting of domestic waste by using two epigeic earthworms (Perionyx excavatus and Perionyx sansibaricus). Int J Environ Sci Technol 5:99–106. https://doi.org/10.1007/BF03326002
Suthar S (2009) Vermicomposting of vegetable-market solid waste using Eisenia fetida: impact of bulking material on earthworm growth and decomposition rate. Ecol Engin 35:914–920. https://doi.org/10.1016/j.ecoleng.2008.12.019
Swati A, Hait S (2017) Fate and bioavailability of heavy metals during vermicomposting of various organic wastes—a review. Proc Safe Environ Protect 109:30–45. https://doi.org/10.1016/j.psep.2017.03.031
Szanto GL, Hamelers HVM, Rulkens WH, Veeken AHM (2007) NH3, N2O and CH4 emissions during passively aerated composting of straw-rich pig manure. Bioresour Technol 98:2659–2670. https://doi.org/10.1016/j.biortech.2006.09.021
Thangarajan R, Bolan NS, Tian G, Naidu R, Kunhikrishnan A (2013) Role of organic amendment application on greenhouse gas emission from soil. Sci Tot Environ 465:72–96. https://doi.org/10.1016/j.scitotenv.2013.01.031
Turan NG (2008) The effects of natural zeolite on salinity level of poultry litter compost. Bioresour Technol 99:2097–2101. https://doi.org/10.1016/j.biortech.2007.11.061
Turan NG, Ergun ON (2007) Ammonia uptake by natural zeolite in municipal solid waste compost. Environ Prog 26:149–156. https://doi.org/10.1002/ep.10198
Unmar G, Mohee R (2008) Assessing the effect of biodegradable and degradable plastics on the composting of green wastes and compost quality. Bioresour Technol 99:6738–6744. https://doi.org/10.1016/j.biortech.2008.01.016
Unuofin FO, Mnkeni PNS (2014) Optimization of Eisenia fetida stocking density for the bioconversion of rock phosphate enriched cow dung–waste paper mixtures. Waste Manag 34:2000–2006. https://doi.org/10.1016/j.wasman.2014.05.018
Venglovsky J, Sasakova N, Vargova M, Pacajova Z, Placha I, Petrovsky M, Harichova D (2005) Evolution of temperature and chemical parameters during composting of the pig slurry solid fraction amended with natural zeolite. Bioresour Technol 96:181–189. https://doi.org/10.1016/j.biortech.2004.05.006
Villasenor J, Rodriguez L, Fernandez FJ (2011) Composting domestic sewage sludge with natural zeolites in a rotary drum reactor. Bioresour Technol 102:1447–1454. https://doi.org/10.1016/j.biortech.2010.09.085
Wakase S, Sasaki H, Itoh K, Otawa K, Kitazume O, Nonaka J, Satoh M, Sasaki T, Nakai Y (2008) Investigation of the microbial community in a microbiological additive used in a manure composting process. Bioresour Technol 99:2687–2693. https://doi.org/10.1016/j.biortech.2007.04.040
Wang S, Ang HM, Tade MO (2008) Novel applications of red mud as coagulant, adsorbent and catalyst for environmentally benign processes. Chemosphere 72:1621–1635. https://doi.org/10.1016/j.chemosphere.2008.05.013
Wang L, Zhang Y, Lian J, Chao J, Gao Y, Yang F, Zhang L (2013a) Impact of fly ash and phosphatic rock on metal stabilization and bioavailability during sewage sludge vermicomposting. Bioresour Technol 136:281–287. https://doi.org/10.1016/j.biortech.2013.03.039
Wang L, Zheng Z, Zhang Y, Chao J, Gao Y, Luo X, Zhang J (2013b) Biostabilization enhancement of heavy metals during the vermiremediation of sewage sludge with passivant. J Hazard Mater 244:1–9. https://doi.org/10.1016/j.jhazmat.2012.11.036
Wang J, Hu Z, Xu X, Jiang X, Zheng B, Liu X, Pan X, Kardol P (2014) Emissions of ammonia and greenhouse gases during combined pre-composting and vermicomposting of duck manure. Waste Manag 34:1546–1552. https://doi.org/10.1016/j.wasman.2014.04.010
Wang Q, Li R, Cai H, Awasthi MK, Zhang Z, Wang JJ, Ali A, Amanullah M (2016) Improving pig manure composting efficiency employing Ca-bentonite. Ecol Eng 87:157–161. https://doi.org/10.1016/j.ecoleng.2015.11.032
Wang Q, Awasthi MK, Zhao J, Ren X, Li R, Wang Z, Wang M, Zhang Z (2017) Improvement of pig manure compost lignocellulose degradation, organic matter humification and compost quality with medical stone. Bioresour Technol 243:771–777. https://doi.org/10.1016/j.biortech.2017.07.021
Waqas M, Nizami AS, Aburiazaiza AS, Barakat MA, Ismail IMI, Rashid MI (2017) Optimization of food waste compost with the use of biochar. J Environ Manag (in press). https://doi.org/10.1016/j.jenvman.2017.06.015
Wei Z, Xi B, Zhao Y, Wang S, Liu H, Jiang Y (2007) Effect of inoculating microbes in municipal solid waste composting on characteristics of humic acid. Chemosphere 68:368–374. https://doi.org/10.1016/j.chemosphere.2006.12.067
Wei L, Shutao W, Jin Z, Tong X (2014) Biochar influences the microbial community structure during tomato stalk composting with chicken manure. Bioresour Technol 154:148–154. https://doi.org/10.1016/j.biortech.2013.12.022
Wichuk KM, McCartney D (2007) A review of the effectiveness of current time–temperature regulations on pathogen inactivation during composting. J Environ Engin Sci 6:573–586. https://doi.org/10.1139/S07-011
Witter E, Lopez-Real J (1988) Nitrogen losses during the composting of sewage sludge, and the effectiveness of clay soil, zeolite, and compost in adsorbing the volatilized ammonia. Biological Wastes 23:279–294. https://doi.org/10.1016/0269-7483(88)90016-X
Wong JWC, Li SWY, Wong MH (1995) Coal fly ash as a composting material for sewage sludge: effects on microbial activities. Environ Technol 16:527–537. https://doi.org/10.1080/09593331608616294
Wong JWC, Fang M, Li GX, Wong MH (1997) Feasibility of using coal ash residues as co-composting materials for sewage sludge. Environ Technol 18:563–568. https://doi.org/10.1080/09593331808616574
Wong JWC, Fung SO, Selvam A (2009) Coal fly ash and lime addition enhances the rate and efficiency of decomposition of food waste during composting. Bioresour Technol 100:3324–3331. https://doi.org/10.1016/j.biortech.2009.01.063
Wong JWC, Selvam A (2009) Reduction of indicator and pathogenic microorganisms in pig manure through fly ash and lime addition during alkaline stabilization. J Hazard Mater 169:882–889. https://doi.org/10.1016/j.jhazmat.2009.04.033
Wu C, Li W, Wang K, Li Y (2015) Usage of pumice as bulking agent in sewage sludge composting. Bioresour Technol 190:516–521. https://doi.org/10.1016/j.biortech.2015.03.104
Wu H, Lai C, Zeng G, Liang J, Chen J, Xu J, Dai J, Li X, Liu J, Chen M, Lu L, Hu L, Wan J (2017) The interactions of composting and biochar and their implications for soil amendment and pollution remediation: a review. Crit Rev Biotechnol 37:754–764. https://doi.org/10.1080/07388551.2016.1232696
Xiao R, Awasthi MK, Li R, Park J, Pensky SM, Wang Q, Wang JJ, Zhang Z (2017) Recent developments in biochar utilization as an additive in organic solid waste composting: a review. Bioresour Technol. https://doi.org/10.1016/j.biortech.2017.07.090
Yang F, Li GX, Yang QY, Luo WH (2013) Effect of bulking agents on maturity and gaseous emissions during kitchen waste composting. Chemosphere 93:1393–1399. https://doi.org/10.1016/j.chemosphere.2013.07.002
Yang F, Li G, Shi H, Wang Y (2015) Effects of phosphogypsum and superphosphate on compost maturity and gaseous emissions during kitchen waste composting. Waste Manag 36:70–76. https://doi.org/10.1016/j.wasman.2014.11.012
Yu H, Huang GH (2009) Effect of sodium acetate as a pH control amendment on the composting of food waste. Bioresour Technol 100:2005–2011. https://doi.org/10.1016/j.biortech.2008.10.007
Yuan J, Yang Q, Zhang Z, Li G, Luo W, Zhang D (2015) Use of additive and pretreatment to control odors in municipal kitchen waste during aerobic composting. J Environ Sci 37:83–90. https://doi.org/10.1016/j.jes.2015.03.028
Zaller JG (2007) Vermicompost in seedling potting media can affect germination, biomass allocation, yields and fruit quality of three tomato varieties. Eur J Soil Biol 43:S332–S336. https://doi.org/10.1016/j.ejsobi.2007.08.020
Zang B, Li S, Michel FC, Li G, Zhang D, Li Y (2017) Control of dimethyl sulfide and dimethyl disulfide odors during pig manure composting using nitrogen amendment. Bioresour Technol 224:419–427. https://doi.org/10.1016/j.biortech.2016.11.023
Zayed G, Abdel-Motaal H (2005) Bio-production of compost with low pH and high soluble phosphorus from sugar cane bagasse enriched with rock phosphate. World J Microbiol Biotechnol 21:747–752. https://doi.org/10.1007/s11274-004-5407-y
Zhang L, Sun X (2014) Changes in physical, chemical, and microbiological properties during the two-stage co-composting of green waste with spent mushroom compost and biochar. Bioresour Technol 171:274–284. https://doi.org/10.1016/j.biortech.2014.08.079
Zhang L, Sun X (2017) Addition of fish pond sediment and rock phosphate enhances the composting of green waste. Bioresour Technol 233:116–126. https://doi.org/10.1016/j.biortech.2017.02.073
Zhang D, Luo W, Yuan J, Li G, Luo Y (2017) Effects of woody peat and superphosphate on compost maturity and gaseous emissions during pig manure composting. Waste Manag 68:56–63. https://doi.org/10.1016/j.wasman.2017.05.042
Zhao L, Cao X, Mašek O, Zimmerman A (2013) Heterogeneity of biochar properties as a function of feedstock sources and production temperatures. J Hazard Mater 256:1–9. https://doi.org/10.1016/j.jhazmat.2013.04.015
Zhou R, Liu X, Luo L, Zhou Y, Wei J, Chen A, Tang L, Wu H, Deng Y, Wang Y (2017) Remediation of Cu, Pb, Zn and Cd-contaminated agricultural soil using a combined red mud and compost amendment. Int Biodeter Biodeg 118:73–81. https://doi.org/10.1016/j.ibiod.2017.01.023
Zorpas AA, Constantinides T, Vlyssides AG, Haralambous I, Loizidou M (2000) Heavy metal uptake by natural zeolite and metals partitioning in sewage sludge compost. Bioresour Technol 72:113–119. https://doi.org/10.1016/S0960-8524(99)00110-8
Zorpas AA, Loizidou M (2008) Sawdust and natural zeolite as a bulking agent for improving quality of a composting product from anaerobically stabilized sewage sludge. Bioresour Technol 99:7545–7552. https://doi.org/10.1016/j.biortech.2008.02.014
Zucconi F, Pera A, Forte M, de Bertoldi MD (1981) Evaluating toxicity of immature compost. Biocycle 22:54–57
Zucconi F, de Bertoldi MD (1987) Organic waste stabilization throughout composting and its compatibility with agricultural uses. In: Wise DL (ed) Global bioconversion. CRC, Boca Raton, pp 109–137
Acknowledgements
The authors wish to thank the Pierre and Marie Curie University for doctoral fellowship. Two anonymous reviewers are acknowledged for their constructive comments.
Funding
Financial support was provided by the CNRS under the framework EC2CO program (LOMBRICOM project) and by the ADEME under the framework of the DOSTE program (VERMISOL project).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Barthod, J., Rumpel, C. & Dignac, MF. Composting with additives to improve organic amendments. A review. Agron. Sustain. Dev. 38, 17 (2018). https://doi.org/10.1007/s13593-018-0491-9
Accepted:
Published:
DOI: https://doi.org/10.1007/s13593-018-0491-9