Abstract
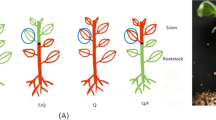
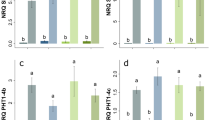
Grafting potato into Solanaceae plants has been used as an efficient method to solve such problems as a short flowering period, low fruit setting rate, and sterility in potato crossbreeding. However, the molecular mechanisms governing grafting-induced phenotypic variations have not been elucidated. In this study, one potato cultivar and wild Datura stramonium served as scion (StS) and rootstock (DsR), respectively. Phenotypic analysis showed that the fertility of StS was visibly improved in comparison with self-grafted potato (St). Comparative transcriptome profiling revealed that 1490 differentially expressed genes were identified in StS compared with St, and some of these genes appear to be involved in pathways related to genome damage and genotoxic stress response. We reported the comprehensive identification of mRNA movement between DsR and StS, indicating that 111 transcripts of DsR were delivered to StS. Conversely, the 1968 mRNAs were transported from StS to DsR. Based on GO analysis, some of these mobile transcripts had a biological role in regulating pollen tube development and fruit morphogenesis. This work provides abundant transcriptome profile data for potato scions and reveals that three key transcripts moving from rootstock to scion may be responsible for improving the fertility of potatoes.








Similar content being viewed by others
Abbreviations
- St:
-
Self-grafted potato
- StS:
-
Heterografted potato scion
- Ds:
-
Ungrafted D. stramonium
- DsR:
-
Heterografted D. stramonium rootstock
- TPS:
-
True potato seeds
References
Banerjee AK, Chatterjee M, Yu Y, Suh SG, Miller WA, Hannapel DJ (2006) Dynamics of a mobile RNA of potato involved in a long-distance signaling pathway. Plant Cell 18:3443–3457. https://doi.org/10.1105/tpc.106.042473
Cao L, Yu N, Li J, Qi Z, Wang D, Chen L (2016) Heritability and reversibility of DNA methylation induced by in vitro grafting between Brassica juncea and B. oleracea. Sci Rep 6:27233. https://doi.org/10.1038/srep27233
Corbesier L et al (2007) FT protein movement contributes to long-distance signaling in floral induction of Arabidopsis. Science 316:1030–1033. https://doi.org/10.1126/science.1141752
Gao X, Zhao S, Xu Q-L, Xiao J-X (2016) Transcriptome responses of grafted Citrus sinensis plants to inoculation with the arbuscular mycorrhizal fungus Glomus versiforme. Trees 30:1073–1082. https://doi.org/10.1007/s00468-015-1345-6
Grabherr MG et al (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652. https://doi.org/10.1038/nbt.1883
Gurdon C, Svab Z, Feng Y, Kumar D, Maliga P (2016) Cell-to-cell movement of mitochondria in plants. Proc Natl Acad Sci U S A 113:3395–3400. https://doi.org/10.1073/pnas.1518644113
Haywood V, Yu TS, Huang NC, Lucas WJ (2005) Phloem long-distance trafficking of Gibberellic acid-insensitive RNA regulates leaf development. Plant J 42:49–68. https://doi.org/10.1111/j.1365-313X.2005.02351.x
Hu C, Ham BK, El-Shabrawi HM, Alexander D, Zhang D, Ryals J, Lucas WJ (2016) Proteomics and metabolomics analyses reveal the cucurbit sieve tube system as a complex metabolic space. Plant J 87:442–454. https://doi.org/10.1111/tpj.13209
Jiang FZ et al (2016) The utility model relates to a method of potato cross breeding Chinese Patent
Kehr J, Kragler F (2018) Long distance RNA movement. New Phytol 218:29–40. https://doi.org/10.1111/nph.15025
Kim M, Canio W, Kessler S, Sinha N (2001) Developmental changes due to long-distance movement of a homeobox fusion transcript in tomato. Science 293:287–289. https://doi.org/10.1126/science.1059805
Lewsey MG et al (2016) Mobile small RNAs regulate genome-wide DNA methylation. Proc Natl Acad Sci U S A 113:E801-810. https://doi.org/10.1073/pnas.1515072113
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Method Enzymol 148C:350–382
Lin MK et al (2007) Flowering locus T protein may act as the long-distance florigenic signal in the cucurbits. Plant Cell 19:1488–1506. https://doi.org/10.1105/tpc.107.051920
Liu L, Chen X (2018) Intercellular and systemic trafficking of RNAs in plants. Nat Plants 4:869–878. https://doi.org/10.1038/s41477-018-0288-5
Lucas WJ, Yoo BC, Kragler F (2001) RNA as a long-distance information macromolecule in plants. Nat Rev Mol Cell Biol 2:849–857. https://doi.org/10.1038/35099096
Lucas WJ et al (2013) The plant vascular system: evolution, development and functions. J Integr Plant Biol 55:294–388. https://doi.org/10.1111/jipb.12041
Luo KR, Huang NC, Yu TS (2018) Selective targeting of mobile mRNAs to plasmodesmata for cell-to-cell movement. Plant Physiol 177:604–614. https://doi.org/10.1104/pp.18.00107
Mao X, Cai T, Olyarchuk JG, Wei L (2005) Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics 21:3787–3793. https://doi.org/10.1093/bioinformatics/bti430
Notaguchi M, Wolf S, Lucas WJ (2012) Phloem-mobile Aux/IAA transcripts target to the root tip and modify root architecture. J Integr Plant Biol 54:760–772. https://doi.org/10.1111/j.1744-7909.2012.01155.x
Omid A, Keilin T, Glass A, Leshkowitz D, Wolf S (2007) Characterization of phloem-sap transcription profile in melon plants. J Exp Bot 58:3645–3656. https://doi.org/10.1093/jxb/erm214
Qi LP et al (2015) The utility model relates to a method for solving the sterility of potato varieties as hybrid parent Chinese Patent
Thieme CJ et al (2015) Endogenous arabidopsis messenger RNAs transported to distant tissues. Nat Plants 1:15025. https://doi.org/10.1038/nplants.2015.25
Trapnell C, Pachter L, Salzberg SL (2009) TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 25:1105–1111. https://doi.org/10.1093/bioinformatics/btp120
Trapnell C et al (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–515. https://doi.org/10.1038/nbt.1621
Uthup TK, Karumamkandathil R, Ravindran M, Saha T (2018) Heterografting induced DNA methylation polymorphisms in Hevea brasiliensis. Planta. https://doi.org/10.1007/s00425-018-2918-6
Wang XK, Huang XL (2015) Principles and techniques of plant physiological and biochemical experiments. China Higher Education Press
Wang Y, Zhang WZ, Song LF, Zou JJ, Su Z, Wu WH (2008) Transcriptome analyses show changes in gene expression to accompany pollen germination and tube growth in Arabidopsis. Plant Physiol 148:1201–1211. https://doi.org/10.1104/pp.108.126375
Wang L, Feng Z, Wang X, Wang X, Zhang X (2010) DEGseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 26:136–138. https://doi.org/10.1093/bioinformatics/btp612
Wang SS, Liu ZZ, Sun C, Shi QH, Yao YX, You CX, Hao YJ (2012) Functional characterization of the apple MhGAI1 gene through ectopic expression and grafting experiments in tomatoes. J Plant Physiol 169:303–310. https://doi.org/10.1016/j.jplph.2011.09.012
Ye M et al (2018) Generation of self-compatible diploid potato by knockout of S-RNase. Nat Plants 4:651–654. https://doi.org/10.1038/s41477-018-0218-6
Young MD, Wakefield MJ, Smyth GK, Oshlack A (2010) Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol 11:R14. https://doi.org/10.1186/gb-2010-11-2-r14
Yu N, Cao L, Yuan L, Zhi X, Chen Y, Gan S, Chen L (2018) Maintenance of grafting-induced epigenetic variations in the asexual progeny of Brassica oleracea and B. juncea chimera. Plant J 96:22–38. https://doi.org/10.1111/tpj.14058
Zhang GH, Guo HC (2018) Effects of tomato and potato heterografting on photosynthesis, quality and yield of grafted parents. Hortic Environ Biotechnol 60:9–18. https://doi.org/10.1007/s13580-018-0096-x
Zhang GH, Mao ZC, Wang Q, Song J, Guo HC (2018) Comprehensive transcriptome profiling and phenotyping of rootstock and scion in a tomato/potato heterografting system. Physiol Plant 166:833–847. https://doi.org/10.1111/ppl.12858
Zhang C et al (2019) The genetic basis of inbreeding depression in potato. Nat Genet 51:374–378. https://doi.org/10.1038/s41588-018-0319-1
Zhang GH (2019) Phenotypic variation and genetic response of grafted parents in potato and tomato, D. stramonium heterograft system. Doctoral dissertation, pp 131–144. https://xueshu.baidu.com/scholarID/CN-BL82NF2J
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 31860402) and the National Potato Industry Technology System (No. CARS-09-P 15).
Funding
Funding is provided by the National Potato Industry Technology System (Grant No. No. CARS-09-P 15), National natural science foundation of china (Grant No. No. 31860402).
Author information
Authors and Affiliations
Contributions
HG and XN conceived and designed, GZ and JZ performed and completed the experiments, Transcriptome data analysis was conducted by JS and XG, GZ and XN wrote manuscript, HG obtained funding. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no financial or other relations that could lead to a conflict of interest.
Additional information
Communicated by Seon-In Yeom.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, G., Zhou, J., Song, J. et al. Grafting-induced transcriptome changes and long-distance mRNA movement in the potato/Datura stramonium heterograft system. Hortic. Environ. Biotechnol. 63, 229–238 (2022). https://doi.org/10.1007/s13580-021-00387-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-021-00387-2