Abstract
Plants of the genus Senna (Fabaceae) are valued for industrial and medicinal properties that have spurred crop improvement through breeding programs. However, there is very little chromosomal data for this genus, limiting the potential for cytogenetics and genomics research. Here, we used fluorescence in situ hybridization (FISH) to perform a comparative analysis of 10 Senna spp. karyotypes (2n = 28 except S. tora with 2n = 26). We detected differences in the distributions of rDNA gene clusters and Arabidopsis-type telomeric repeats (TTTAGGG)n. Only one 5S rDNA pair was detected in all species, but we observed more interspecies variation for 45S rDNA: four pairs in S. didymobotrya, S. spectabilis, and S. viarum; two pairs in S. candolleana; and one pair in S. bauhinioides, S. multiglandulosa, S. occidentalis, and S. tora. In both S. leandrii and S. sulfurea, 45S rDNA distribution was hemizygous. While all chromosomes possessed telomeric signals in the terminal region, this signal was also present in the interstitial regions of all S. tora chromosomes and, less obviously (fewer chromosomes, weaker signals), in S. occidentalis. These preliminary data demonstrate interspecific karyotype variation that reveals Senna genome dynamics. Our results facilitate cytogenetic mapping of other major repeats, thus contributing to an improved understanding of Senna genome structure and evolutionary history.


Similar content being viewed by others
References
Alfenito MR, Birchler JA (1993) Molecular characterization of a maize B chromosome centric sequence. Genetics 135:589–597
Ammiraju JS, Lu F, Sanyal A, Yu Y, Song X, Jiang N, Pontaroli AC, Rambo T, Currie J, Collura J et al (2008) Dynamic evolution of Oryza genomes is revealed by comparative genomic analysis of a genus-wide vertical data set. Plant Cell 20:3191–3209. https://doi.org/10.1105/tpc.108.063727
Baskin JM, Nan X, Baskin CC (1998) A comparative study of seed dormancy and germination in an annual and a perennial species of Senna (Fabaceae). Seed Sci Res 8:501–512. https://doi.org/10.1017/S0960258500004475
Belandres HR, Waminal NE, Hwang Y-J, Park B-S, Lee S-S, Huh JH, Kim HH (2015) FISH karyotype and GISH meiotic pairing analyses of a stable intergeneric hybrid × Brassicoraphanus line BB# 5. Korean J Hortic Sci Technol 33:83–92. https://doi.org/10.7235/hort.2015.14151
Bhat TA, Wani AA (2017) Chromosome structure and aberrations. Springer, New Dehli, pp 158–163. https://doi.org/10.1007/978-81-322-3673-3
Bir SS, Kumari S (1982) Karyotypic studies in Cassia Linn. from India. Proc Indian Natl Sci Acad 48:397–404
Chaulagain BP, Shakya SR (2002) Inconstancy in chromosome number in some species of Cassia L. found in Nepal. NJST 4:123–128
Chester M, Leitch AR, Soltis PS, Soltis DE (2010) Review of the application of modern cytogenetic methods (FISH/GISH) to the study of reticulation. (polyploidy/hybridisation). Genes 1:166–192. https://doi.org/10.3390/genes1020166
Devi J, Ko JM, Seo BB (2005) FISH and GISH: modern cytogenetic techniques. Indian J Biotechnol 4:307–315
Dover GA (1986) Molecular drive in multigene families: how biological novelties arise, spread and are assimilated. Trends Genet 2:159–165. https://doi.org/10.1016/0168-9525(86)90211-8
Dover GA (1989) Linkage disequilibrium and molecular drive in the rDNA gene family. Genet Soc Am 122:249–252
Dubcovsky J, Dvorák J (1995) Ribosomal RNA multigene loci: nomads of the Triticeae genomes. Genet 140:1367–1377
Elaine B, Miotto ST, Schifino-Wittmann MT, De Castro B (2005) Cytogenetics and cytotaxonomy of Brazilian species of Senna Mill. (Cassieae–Caesalpinioideae–Leguminosae). Caryologia 58:152–163. https://doi.org/10.1080/00087114.2005.10589445
Ferreira K, Torres GA, Sousa SM, Santos ACP (2010) Karyotype, meiotic behavior and pollen features of Senna occidentalis. Biologia 65:789–795. https://doi.org/10.2478/s11756-010-0080-0
Figueroa DM, Bass HW (2010) A historical and modern perspective on plant cytogenetics. Brief Funct Genom 9:95–102. https://doi.org/10.1093/bfgp/elp058
Freyman WA, Höhna S (2018) Cladogenetic and anagenetic models of chromosome number evolution: a Bayesian model averaging approach. Syst Biol 67:195–215. https://doi.org/10.1093/sysbio/syx065
Fuchs J, Brandes A, Schubert I (1995) Telomere sequence localization and karyotype evolution in higher plants. Plant Syst Evol 196:227–241. https://doi.org/10.1007/BF00982962
Guerra M (2008) Chromosome numbers in plant cytotaxonomy: concepts and implications. Cytogenet Genome Res 120:339–350. https://doi.org/10.1159/000121083
Hasterok R, Jenkins G, Langdon T, Jones RN, Maluszynska J (2001) Ribosomal DNA is an effective marker of Brassica chromosomes. Theor Appl Genet 103:486–490. https://doi.org/10.1007/s001220100653
He L, Liu J, Torres GA, Zhang H, Jiang J, Xie C (2013) Interstitial telomeric repeats are enriched in the centromeres of chromosomes in Solanum species. Chromosome Res 21:5–13
Heslop-Harrison JS (1991) The molecular cytogenetics of plants. J Cell Sci 100:5–21
Irwin H, Turner B (1960) Chromosomal relationships and taxonomic considerations in the genus Cassia. Am J Bot. https://doi.org/10.2307/2439612
Jiang J, Gill BS (2006) Current status and the future of fluorescence in situ hybridization (FISH) in plant genome research. Genome 49:1057–1068. https://doi.org/10.1139/g06-076
Koo D-H, Nam Y-W, Choi D, Bang J-W, De Jong H, Hur Y (2010) Molecular cytogenetic mapping of Cucumis sativus and C. melo using highly repetitive DNA sequences. Chromosome Res 18:325–336. https://doi.org/10.1007/s10577-010-9116-0
Lan T, Albert VA (2011) Dynamic distribution patterns of ribosomal DNA and chromosomal evolution in Paphiopedilum, a lady’s slipper orchid. BMC Plant Biol 11:126. https://doi.org/10.1186/1471-2229-11-126
Laxmikanta A, Pratap CP (2010) Validation of generic status of different taxa in the sub-tribe Cassiinae (Leguminosae: Caesalpinoidae) using RAPD, ISSR and AFLP markers. Int J Plant Physiol Biochem 2:18–28
Levan A, Fredga K, Sanberg AA (1964) Nomenclature for centromeric position on chromosomes. Hereditas 52:201–220. https://doi.org/10.1111/j.1601-5223.1964.tb01953.x
Liao D (1999) Concerted evolution: molecular mechanism and biological implications. Am J Hum Genet 64:24–30
Lim KB, Yang TJ, Hwang YJ, Kim JS, Park JY, Kwon SJ, Kim J, Choi BS, Lim MH, Jin M et al (2007) Characterization of the centromere and peri-centromere retrotransposons in Brassica rapa and their distribution in related Brassica species. Plant J 49:173–183. https://doi.org/10.1111/j.1365-313X.2006.02952.x
Maluszynska J, Heslop-Harrison J (1993) Physical mapping of rDNA loci in Brassica species. Genome 36:774–781
Mancia FH, Sohn S-H, Ahn YK, Kim D-S, Kim JS, Kwon Y-S, Kim C-W, Lee T-H, Hwang Y-J (2015) Distribution of various types of repetitive DNAs in Allium cepa L. based on dual color FISH. Hortic Environ Biotechnol 56:793–799. https://doi.org/10.1007/s13580-015-1100-3
Mantovani M, Abel S, Moreira-Filho O (2005) Conserved 5S and variable 45S rDNA chromosomal localisation revealed by FISH in Astyanax scabripinnis (Pisces, Characidae). Genetica 123:211–216. https://doi.org/10.1007/s10709-004-2281-3
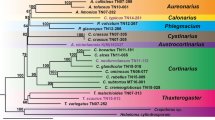
Marazzi B, Endress PK, de Queiroz LP, Conti E (2006) Phylogenetic relationships within Senna (Leguminosae, Cassiinae) based on three chloroplast DNA regions: patterns in the evolution of floral symmetry and extrafloral nectaries. Am J Bot 93:288–303
Martins C, Galetti PM (1999) Chromosomal localization of 5S rDNA genes in Leporinus fish (Anostomidae, Characiformes). Chromosome Res 7:363–367. https://doi.org/10.1023/A:1009216030316
Martins C, Wasko AP (2004) Organization and evolution of 5S ribosomal DNA in the fish genome. Focus Genom Res 289:335–363
Monkheang P, Sudmoon R, Tanee T, Noikotr K, Bletter N, Chaveerach A (2011) Species diversity, usages, molecular markers and barcode of medicinal Senna species (Fabaceae, Caesalpinioideae) in Thailand. J Med Plant Res 5:6173–6181. https://doi.org/10.5897/JMPR11.1075
Ohri D, Kumar A, Pal M (1986) Correlations between 2C DNA values and habit in Cassia (Leguminosae: Caesalpinioideae). Plant Syst Evol 153:223–227
Pawar HA, Lalitha K (2014) Isolation, purification and characterization of galactomannans as an excipient from Senna tora seeds. Int J Biol Macromol 65:167–175. https://doi.org/10.1016/j.ijbiomac.2014.01.026
Pita M, Orellana J, Martinez-Rodriguez P, Martinez-Ramirez A, Fernandez-Calvin B, Bella JL (2014) FISH methods in cytogenetic studies. Methods Mol Biol 1094:109–135. https://doi.org/10.1007/978-1-62703-706-8_10
Presting GG, Frary A, Pillen K, Tanksley SD (1996) Telomere-homologous sequences occur near the centromeres of many tomato chromosomes. Mol Gen Genet 251:526–531
Rahman MO, Rahman MZ, Begum A (2013) Numerical taxonomy of the genus Senna mill from Bangladesh. Bangladesh J Plant Taxon 20:77
Resende K, Prado C, Davide L, Torres G (2014) Polyploidy and apomixis in accessions of Senna rugosa (G. Don) HS Irwin & Barneby. Turk J Biol 38:510–515. https://doi.org/10.3906/biy-1312-66
Rice A, Glick L, Abadi S, Einhorn M, Kopelman NM, Salman-Minkov A, Mayzel J, Chay O, Mayrose I (2015) The chromosome counts database (CCDB)—a community resource of plant chromosome numbers. New Phytol 206:19–26. https://doi.org/10.1111/nph.13191
Roa F, Guerra M (2012) Distribution of 45S rDNA sites in chromosomes of plants: structural and evolutionary implications. BMC Evol Biol 12:225. https://doi.org/10.1186/1471-2148-12-225
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Sharma S, Raina S (2005) Organization and evolution of highly repeated satellite DNA sequences in plant chromosomes. Cytogenet Genom Res 109:15–26. https://doi.org/10.1159/000082377
Singh S, Singh SK, Yadav A (2013) A review on Cassia species: pharmacological, traditional and medicinal aspects in various countries. Am J Phytomed Clin Ther 1:291–312
Tartof KD (1988) Unequal crossing over then and now. Genetics 120:1–6
Tessadori F, van Driel R, Fransz P (2004) Cytogenetics as a tool to study gene regulation. Trends Plant Sci 9:147–153. https://doi.org/10.1016/j.tplants.2004.01.008
Tripathi V, Goswami S (2011) Generic relationship among Cassia L., Senna Mill. and Chamaecrista Moench using RAPD markers. Int J Biodivers Conserv 3:92–100
Tucker SC (1996) Trends in evolution of floral ontogeny in Cassia sensu stricto, Senna, and Chamaecrista (Leguminosae: Caesalpinioideae: Cassieae: Cassiinae): a study in convergence. Am J Bot 83:687–711. https://doi.org/10.2307/2445846
Uchida W, Matsunaga S, Sugiyama R, Kawano S (2002) Interstitial telomere-like repeats in the Arabidopsis thaliana genome. Genes Genet Syst 77:6–67
Vrana J, Simkova H, Kubalakova M, Cihalikova J, Dolezel J (2012) Flow cytometric chromosome sorting in plants: the next generation. Methods 57:331–337. https://doi.org/10.1016/j.ymeth.2012.03.006
Waminal NE, Kim HH (2012) Dual-color FISH karyotype and rDNA distribution analyses on four Cucurbitaceae species. Hortic Environ Biotechnol 53:49–56. https://doi.org/10.1007/s13580-012-0105-4
Waminal NE, Kim HH (2015) FISH karyotype analysis of four wild Cucurbitaceae species using 5S and 45S rDNA probes and the emergence of new polyploids in Trichosanthes kirilowii Maxim. Korean J Hortic Sci Technol 33:869–876. https://doi.org/10.7235/hort.2015.15101
Waminal NE, Perumal S, Lee J, Kim HH, Yang T-J (2016) Repeat evolution in Brassica rapa (AA), B. oleracea (CC), and B. napus (AACC) genomes. Plant Breed Biotechnol 4:107–122. https://doi.org/10.9787/PBB.2016.4.2.107
Waminal NE, Pellerin RJ, Kim NS, Jayakodi M, Park JY, Yang TJ, Kim HH (2018) Rapid and efficient FISH using pre-labeled oligomer probes. Sci Rep 8:8224
Watanabe K, King RM, Yahara T, Ito M, Yokoyama J, Suzuki T, Crawford DJ (1995) Chromosomal cytology and evolution in Eupatorieae (Asteraceae). Ann Mo Bot Gard 82:581–592. https://doi.org/10.2307/2399838
Acknowledgments
This study was funded by a Grant from the National Research Foundation of Korea (NRF 2017R1A2B2004778).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pellerin, R.J., Waminal, N.E. & Kim, H.H. FISH mapping of rDNA and telomeric repeats in 10 Senna species. Hortic. Environ. Biotechnol. 60, 253–260 (2019). https://doi.org/10.1007/s13580-018-0115-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-018-0115-y