Abstract
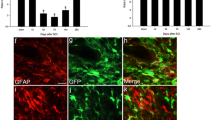
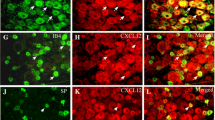
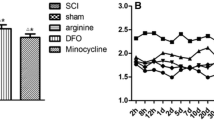
Spinal cord injury (SCI) causes not only sensorimotor and cognitive deficits, but frequently also severe chronic pain that is difficult to treat (SCI pain). We previously showed that hyperesthesia, as well as spontaneous pain induced by electrolytic lesions in the rat spinothalamic tract, is associated with increased spontaneous and sensory-evoked activity in the posterior thalamic nucleus (PO). We have also demonstrated that rodent impact SCI increases cell cycle activation (CCA) in the injury region and that post-traumatic treatment with cyclin dependent kinase inhibitors reduces lesion volume and motor dysfunction. Here we examined whether CCA contributes to neuronal hyperexcitability of PO and hyperpathia after rat contusion SCI, as well as to microglial and astroglial activation (gliopathy) that has been implicated in delayed SCI pain. Trauma caused enhanced pain sensitivity, which developed weeks after injury and was correlated with increased PO neuronal activity. Increased CCA was found at the thoracic spinal lesion site, the lumbar dorsal horn, and the PO. Increased microglial activation and cysteine–cysteine chemokine ligand 21 expression was also observed in the PO after SCI. In vitro, neurons co-cultured with activated microglia showed up-regulation of cyclin D1 and cysteine–cysteine chemokine ligand 21 expression. In vivo, post-injury treatment with a selective cyclin dependent kinase inhibitor (CR8) significantly reduced cell cycle protein induction, microglial activation, and neuronal activity in the PO nucleus, as well as limiting chronic SCI-induced hyperpathia. These results suggest a mechanistic role for CCA in the development of SCI pain, through effects mediated in part by the PO nucleus. Moreover, cell cycle modulation may provide an effective therapeutic strategy to improve reduce both hyperpathia and motor dysfunction after SCI.










Similar content being viewed by others
References
Persu C, Caun V, Dragomiriteanu I, Geavlete P. Urological management of the patient with traumatic spinal cord injury. J Med Life 2009;2:296-302.
Widerstrom-Noga EG, Felix ER, Cruz-Almeida Y, Turk DC. Psychosocial subgroups in persons with spinal cord injuries and chronic pain. Arch Phys Med Rehabil 2007;88:1628-1635.
Abramson CE, McBride KE, Konnyu KJ, Elliott SL. Sexual health outcome measures for individuals with a spinal cord injury: a systematic review. Spinal Cord 2008;46:320-324.
Stormer S, Gerner HJ, Gruninger W, et al. Chronic pain/dysaesthesiae in spinal cord injury patients: results of a multicentre study. Spinal Cord 1997;35:446-455.
Yezierski RP. Pain following spinal cord injury: pathophysiology and central mechanisms. Prog Brain Res 2000;129:429-449.
Defrin R, Ohry A, Blumen N, Urca G. Pain following spinal cord injury. Spinal Cord 2002;40:96-97.
Tasker RR. Meralgia paresthetica. J Neurosurg 1991;75:168.
Greenspan JD, Ohara S, Sarlani E, Lenz FA. Allodynia in patients with post-stroke central pain (CPSP) studied by statistical quantitative sensory testing within individuals. Pain 2004;109:357-366.
Baastrup C, Finnerup NB. Pharmacological management of neuropathic pain following spinal cord injury. CNS Drugs 2008;22:455-475.
Tasker RR, DeCarvalho GT, Dolan EJ. Intractable pain of spinal cord origin: clinical features and implications for surgery. J Neurosurg 1992;77:373-378.
Falci S, Best L, Bayles R, Lammertse D, Starnes C. Dorsal root entry zone microcoagulation for spinal cord injury-related central pain: operative intramedullary electrophysiological guidance and clinical outcome. J Neurosurg 2002;97:193-200.
Rintala DH, Holmes SA, Fiess RN, Courtade D, Loubser PG. Prevalence and characteristics of chronic pain in veterans with spinal cord injury. J Rehabil Res Dev 2005;42:573-584.
Masri R, Keller A. Chronic pain following spinal cord injury. Adv Exp Med Biol 2012;760:74-88.
Lenz FA, Kwan HC, Dostrovsky JO, Tasker RR. Characteristics of the bursting pattern of action potentials that occurs in the thalamus of patients with central pain. Brain Res 1989;496:357-360.
Yezierski RP. Spinal cord injury pain: spinal and supraspinal mechanisms. J Rehab Res Develop 2009;46:95-107.
Waxman SG, Hains BC. Fire and phantoms after spinal cord injury: Na+ channels and central pain. Trends Neurosci 2006;29:207-215.
Hubscher CH, Johnson RD. Chronic spinal cord injury induced changes in the responses of thalamic neurons. Exp Neurol 2006;197:177-188.
Gerke MB, Duggan AW, Xu L, Siddall PJ. Thalamic neuronal activity in rats with mechanical allodynia following contusive spinal cord injury. Neuroscience 2003;117:715-722.
Hains BC, Saab CY, Waxman SG. Changes in electrophysiological properties and sodium channel Nav1.3 expression in thalamic neurons after spinal cord injury. Brain 2005;128:2359-2371.
Hains BC, Saab CY, Waxman SG. Alterations in burst firing of thalamic VPL neurons and reversal by Na(v)1.3 antisense after spinal cord injury. J Neurophysiol 2006;95:3343-3352.
Zhao P, Waxman SG, Hains BC. Modulation of thalamic nociceptive processing after spinal cord injury through remote activation of thalamic microglia by cysteine cysteine chemokine ligand 21. J Neurosci 2007;27:8893-8902.
Poggio GF, Mountcastle VB. A study of the functional contributions of the lemniscal and spinothalamic systems to somatic sensibility. Central nervous mechanisms in pain. Bull Johns Hopkins Hosp 1960;106:266-316.
Zhang X, Giesler GJ, Jr. Response characteristics of spinothalamic tract neurons that project to the posterior thalamus in rats. J Neurophysiol 2005;93:2552-2564.
Masri R, Quiton RL, Lucas JM, Murray PD, Thompson SM, Keller A. Zona incerta: a role in central pain. J Neurophysiol 2009;102:181-191.
Popovich PG, Wei P, Stokes BT. Cellular inflammatory response after spinal cord injury in Sprague-Dawley and Lewis rats. J Comp Neurol 1997;377:443-464.
Schmitt AB, Buss A, Breuer S, et al. Major histocompatibility complex class II expression by activated microglia caudal to lesions of descending tracts in the human spinal cord is not associated with a T cell response. Acta Neuropathol 2000;100:528-536.
Crown ED, Ye Z, Johnson KM, Xu GY, McAdoo DJ, Hulsebosch CE. Increases in the activated forms of ERK 1/2, p38 MAPK, and CREB are correlated with the expression of at-level mechanical allodynia following spinal cord injury. Exp Neurol 2006;199:397-407.
Hains BC, Waxman SG. Activated microglia contribute to the maintenance of chronic pain after spinal cord injury. J Neurosci 2006;26:4308-4317.
Hulsebosch CE, Hains BC, Crown ED, Carlton SM. Mechanisms of chronic central neuropathic pain after spinal cord injury. Brain Res Rev 2009;60:202-213.
Wu J, Pajoohesh-Ganji A, Stoica BA, Dinizo M, Guanciale K, Faden AI. Delayed expression of cell cycle proteins contributes to astroglial scar formation and chronic inflammation after rat spinal cord contusion. J Neuroinflammation 2012;9:169.
Gwak YS, Kim HK, Kim HY, Leem JW. Bilateral hyperexcitability of thalamic VPL neurons following unilateral spinal injury in rats. J Physiol Sci 2010;60:59-66.
Hulsebosch CE. Gliopathy ensures persistent inflammation and chronic pain after spinal cord injury. Exp Neurol 2008;214:6-9.
Di Giovanni S, Movsesyan V, Ahmed F, et al. Cell cycle inhibition provides neuroprotection and reduces glial proliferation and scar formation after traumatic brain injury. Proc Natl Acad Sci U S A 2005;102:8333-8338.
Wu J, Stoica BA, Faden AI. Cell cycle activation and spinal cord injury. Neurotherapeutics 2011;8:221-228.
Osuga H, Osuga S, Wang F, et al. Cyclin-dependent kinases as a therapeutic target for stroke. Proc Natl Acad Sci U S A 2000;97:10254-10259.
Park DS, Morris EJ, Bremner R, et al. Involvement of retinoblastoma family members and E2F/DP complexes in the death of neurons evoked by DNA damage. J Neurosci 2000;20:3104-3114.
Wang F, Corbett D, Osuga H, et al. Inhibition of cyclin-dependent kinases improves CA1 neuronal survival and behavioral performance after global ischemia in the rat. J Cereb Blood Flow Metab 2002;22:171-182.
Cernak I, Stoica B, Byrnes KR, Di Giovanni S, Faden AI. Role of the cell cycle in the pathobiology of central nervous system trauma. Cell Cycle 2005;4:1286-1293.
Byrnes KR, Stoica BA, Fricke S, Di Giovanni S, Faden AI. Cell cycle activation contributes to post-mitotic cell death and secondary damage after spinal cord injury. Brain 2007;130:2977-2992.
Tian DS, Dong Q, Pan DJ, et al. Attenuation of astrogliosis by suppressing of microglial proliferation with the cell cycle inhibitor olomoucine in rat spinal cord injury model. Brain Res 2007;1154:206-214.
Liu DZ, Ander BP, Sharp FR. Cell cycle inhibition without disruption of neurogenesis is a strategy for treatment of central nervous system diseases. Neurobiol Dis 2010;37:549-557.
Kabadi SV, Stoica BA, Byrnes KR, Hanscom M, Loane DJ, Faden AI. Selective CDK inhibitor limits neuroinflammation and progressive neurodegeneration after brain trauma. J Cereb Blood Flow Metab 2012;32:137-149.
Wu J, Stoica BA, Dinizo M, Pajoohesh-Ganji A, Piao C, Faden AI. Delayed cell cycle pathway modulation facilitates recovery after spinal cord injury. Cell Cycle 2012;11:1782-1795.
Tsuda M, Kohro Y, Yano T, et al. JAK-STAT3 pathway regulates spinal astrocyte proliferation and neuropathic pain maintenance in rats. Brain 2011;134:1127-1139.
Wang CH, Chou WY, Hung KS, et al. Intrathecal administration of roscovitine inhibits Cdk5 activity and attenuates formalin-induced nociceptive response in rats. Acta Pharmacol Sin 2005;26:46-50.
Yang YR, He Y, Zhang Y, et al. Activation of cyclin-dependent kinase 5 (Cdk5) in primary sensory and dorsal horn neurons by peripheral inflammation contributes to heat hyperalgesia. Pain 2007;127:109-120.
Yakovlev AG, Faden AI. Sequential expression of c-fos protooncogene, TNF-alpha, and dynorphin genes in spinal cord following experimental traumatic injury. Mol Chem Neuropathol 1994;23:179-190.
Basso DM, Beattie MS, Bresnahan JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 1995;12:1-21.
Gale K, Kerasidis H, Wrathall JR. Spinal cord contusion in the rat: behavioral analysis of functional neurologic impairment. Exp Neurol 1985;88:123-134.
Trageser JC, Burke KA, Masri R, Li Y, Sellers L, Keller A. State-dependent gating of sensory inputs by zona incerta. J Neurophysiol 2006;96:1456-1463.
Sceniak MP, Maciver MB. Cellular actions of urethane on rat visual cortical neurons in vitro. J Neurophysiol 2006;95:3865-3874.
Friedberg MH, Lee SM, Ebner FF. Modulation of receptive field properties of thalamic somatosensory neurons by the depth of anesthesia. J Neurophysiol 1999;81:2243-2252.
West MJ, Slomianka L, Gundersen HJ. Unbiased stereological estimation of the total number of neurons in thesubdivisions of the rat hippocampus using the optical fractionator. Anat Rec 1991;231:482-497.
Soltys Z, Ziaja M, Pawlinski R, Setkowicz Z, Janeczko K. Morphology of reactive microglia in the injured cerebral cortex. Fractal analysis and complementary quantitative methods. J Neurosci Res 2001;63:90-97.
Byrnes KR, Loane DJ, Stoica BA, Zhang J, Faden AI. Delayed mGluR5 activation limits neuroinflammation and neurodegeneration after traumatic brain injury. J Neuroinflammation 2012;9:43.
Wu J, Kharebava G, Piao C, et al. Inhibition of E2F1/CDK1 pathway attenuates neuronal apoptosis in vitro and confers neuroprotection after spinal cord injury in vivo. PLoS One 2012;7:e42129.
Lee HJ, Wu J, Chung J, Wrathall JR. SOX2 expression is upregulated in adult spinal cord after contusion injury in both oligodendrocyte lineage and ependymal cells. J Neurosci Res 2013;91:196-210.
Wu J, Wrathall JR, Schachner M. Phosphatidylinositol 3-kinase/protein kinase Cdelta activation induces close homolog of adhesion molecule L1 (CHL1) expression in cultured astrocytes. Glia 2010;58:315-328.
Hubscher CH, Fell JD, Gupta DS. Sex and hormonal variations in the development of at-level allodynia in a rat chronic spinal cord injury model. Neurosci Lett 2010;477:153-156.
Hall BJ, Lally JE, Vukmanic EV, et al. Spinal cord injuries containing asymmetrical damage in the ventrolateral funiculus is associated with a higher incidence of at-level allodynia. J Pain 2010;11:864-875.
Whitt JL, Masri R, Pulimood NS, Keller A. Pathological activity in mediodorsal thalamus of rats with spinal cord injury pain. J Neurosci 2013;33:3915-3926.
Vierck CJ, Jr., Siddall P, Yezierski RP. Pain following spinal cord injury: animal models and mechanistic studies. Pain 2000;89:1-5.
Carlson SL, Parrish ME, Springer JE, Doty K, Dossett L. Acute inflammatory response in spinal cord following impact injury. Exp Neurol 1998;151:77-88.
Beck KD, Nguyen HX, Galvan MD, Salazar DL, Woodruff TM, Anderson AJ. Quantitative analysis of cellular inflammation after traumatic spinal cord injury: evidence for a multiphasic inflammatory response in the acute to chronic environment. Brain 2010;133:433-447.
Detloff MR, Fisher LC, McGaughy V, Longbrake EE, Popovich PG, Basso DM. Remote activation of microglia and pro-inflammatory cytokines predict the onset and severity of below-level neuropathic pain after spinal cord injury in rats. Exp Neurol 2008;212:337-347.
Ji RR, Kawasaki Y, Zhuang ZY, Wen YR, Decosterd I. Possible role of spinal astrocytes in maintaining chronic pain sensitization: review of current evidence with focus on bFGF/JNK pathway. Neuron Glia Biol 2006;2:259-269.
Gwak YS, Kang J, Unabia GC, Hulsebosch CE. Spatial and temporal activation of spinal glial cells: role of gliopathy in central neuropathic pain following spinal cord injury in rats. Exp Neurol 2012;234:362-372.
Hilton GD, Stoica BA, Byrnes KR, Faden AI. Roscovitine reduces neuronal loss, glial activation, and neurologic deficits after brain trauma. J Cereb Blood Flow Metab 2008;28:1845-1859.
Wang G, Thompson SM. Maladaptive homeostatic plasticity in a rodent model of central pain syndrome: thalamic hyperexcitability after spinothalamic tract lesions. J Neurosci 2008;28:11959-11969.
Koralek KA, Jensen KF, Killackey HP. Evidence for two complementary patterns of thalamic input to the rat somatosensory cortex. Brain Res 1988;463:346-351.
Nothias F, Peschanski M, Besson JM. Somatotopic reciprocal connections between the somatosensory cortex and the thalamic Po nucleus in the rat. Brain Res 1988;447:169-174.
Chmielowska J, Carvell GE, Simons DJ. Spatial organization of thalamocortical and corticothalamic projection systems in the rat SmI barrel cortex. J Comp Neurol 1989;285:325-338.
Fabri M, Burton H. Topography of connections between primary somatosensory cortex and posterior complex in rat: a multiple fluorescent tracer study. Brain Res 1991;538:351-357.
Lu SM, Lin RC. Thalamic afferents of the rat barrel cortex: a light- and electron-microscopic study using Phaseolus vulgaris leucoagglutinin as an anterograde tracer. Somatosens Mot Res 1993;10:1-16.
Bureau I, von Saint Paul F, Svoboda K. Interdigitated paralemniscal and lemniscal pathways in the mouse barrel cortex. PLoS Biol 2006;4:e382.
Bushnell MC, Duncan GH, Hofbauer RK, Ha B, Chen JI, Carrier B. Pain perception: is there a role for primary somatosensory cortex? Proc Natl Acad Sci U S A 1999;96:7705-7709.
Coghill RC, Sang CN, Maisog JM, Iadarola MJ. Pain intensity processing within the human brain: a bilateral, distributed mechanism. J Neurophysiol 1999;82:1934-1943.
Apkarian AV, Bushnell MC, Treede RD, Zubieta JK. Human brain mechanisms of pain perception and regulation in health and disease. Eur J Pain 2005;9:463-484.
Moulton EA, Keaser ML, Gullapalli RP, Greenspan JD. Regional intensive and temporal patterns of functional MRI activation distinguishing noxious and innocuous contact heat. J Neurophysiol 2005;93:2183-2193.
Quiton RL, Masri R, Thompson SM, Keller A. Abnormal activity of primary somatosensory cortex in central pain syndrome. J Neurophysiol 2010;104:1717-1725.
Tator CH, Fehlings MG. Review of clinical trials of neuroprotection in acute spinal cord injury. Neurosurg Focus 1999;6:e8.
Dumont RJ, Okonkwo DO, Verma S, et al. Acute spinal cord injury, part I: pathophysiologic mechanisms. Clin Neuropharmacol 2001;24:254-264.
Di Giovanni S, Knoblach SM, Brandoli C, Aden SA, Hoffman EP, Faden AI. Gene profiling in spinal cord injury shows role of cell cycle in neuronal death. Ann Neurol 2003;53:454-468.
Byrnes KR, Faden AI. Role of cell cycle proteins in CNS injury. Neurochem Res 2007;32:1799-1807.
de Jong EK, Dijkstra IM, Hensens M, et al. Vesicle-mediated transport and release of CCL21 in endangered neurons: a possible explanation for microglia activation remote from a primary lesion. J Neurosci 2005;25:7548-7557.
Byrnes KR, Washington PM, Knoblach SM, Hoffman E, Faden AI. Delayed inflammatory mRNA and protein expression after spinal cord injury. J Neuroinflammation 2011;8:130.
Paxinos G, Watson C. The rat brain in stereotaxic coordinates (2nd edn). Academic Press, Inc., 1998, plate 29.
Acknowledgments
This study was supported by the National Institutes of Health Grants R01 NS054221 (AIF) and R01 NS066965 (AK). We thank Michael Dinizo, Kelsey Guanciale, Rainier Cabatbat, Katherine Cardiff, Marie Hanscom, Angela Pan, and Aicha Moutanni for expert technical support.
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article.
Conflict of Interest
The authors declare that they have no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Junfang Wu and Charles Raver contributed equally to this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 1287 kb)
Rights and permissions
About this article
Cite this article
Wu, J., Raver, C., Piao, C. et al. Cell Cycle Activation Contributes to Increased Neuronal Activity in the Posterior Thalamic Nucleus and Associated Chronic Hyperesthesia after Rat Spinal Cord Contusion. Neurotherapeutics 10, 520–538 (2013). https://doi.org/10.1007/s13311-013-0198-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13311-013-0198-1