Abstract
Introduction
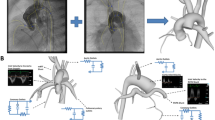
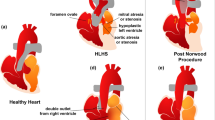
The hybrid Norwood (HN) is a relatively new first stage palliative procedure for neonates with hypoplastic left heart syndrome, in which a sustainable uni-ventricular circulation is established in a less invasive manner than with the standard Norwood procedure. A computational multiscale model of the circulation following the HN procedure was used to obtain detailed hemodynamics. Implementation of a reverse-BT shunt (RBTS), a synthetic bypass from the main pulmonary to the innominate artery placed to counteract aortic arch stenosis, and its effects on local and global hemodynamics were studied.
Methods
A post-op patient-derived anatomy of the HN procedure was utilized with varying degrees of distal arch obstruction, or stenosis, (nominal and 90% lumenal area reduction) and varying RBTS diameters (3.0, 3.5, 4.0 mm). A closed lumped parameter model (LPM) for the proximal and peripheral circulations was coupled to a 3D computational fluid dynamics (CFD) model in order to obtain converged flow fields for analysis.
Results
CFD analyses of patient-derived anatomic configurations demonstrated consistent trends of vascular bed perfusion, vorticity, oscillatory shear index and wall shear stress levels. In the models with severe stenosis, implementation of the RBTS resulted in a restoration of arterial perfusion to near-nominal levels regardless of the shunt diameter. Shunt flow velocity, vorticity, and overall wall shear stress levels decreased with increasing shunt diameter, while shunt flow and systemic oxygen delivery increased with increased shunt diameter. In the absence of distal arch stenosis, large (4.0 mm) grafts may risk thrombosis due to low velocities and flow patterns.
Conclusion
Among the three graft sizes, the best option seems to be the 3.5 mm RBTS which provides a more organized flow similar to that of the 3.0 mm configuration with lower levels of wall shear stress. As such, in the setting of this study and for comparable HN physiologies our results suggest that: (1) the 4.0 mm shunt is a generous shunt diameter choice that may be problematic particularly when implemented prophylactically in the absence of stenosis, and (2) the 3.5 mm shunt may be a more suitable alternative since it exhibits more favorable hemodynamics at lower levels of wall shear stress.

















Similar content being viewed by others
References
Alsoufi, B., J. Bennetts, S. Verma, and C. A. Caldarone. New developments in the treatment of hypoplastic left heart syndrome. Pediatrics 119:109–117, 2007.
Baba, K., O. Honjo, R. Chaturvedi, K. J. Lee, G. Van Arsdell, C. A. Caldarone, et al. “Reverse Blalock–Taussig shunt”: application in single ventricle hybrid palliation. J. Thorac. Cardiovasc. Surg. 146:352–357, 2013.
Baker, C. E., C. Corsini, D. Cosentino, G. Dubini, G. Pennati, F. Migliavacca, et al. Effects of pulmonary artery banding and retrograde aortic arch obstruction on the hybrid palliation of hypoplastic left heart syndrome. J. Thorac. Cardiovasc. Surg. 146:1341–1348, 2013.
Barnea, O., E. H. Austin, B. Richman, and W. P. Santamore. Balancing the circulation: theoretic optimization of pulmonary/systemic flow ratio in hypoplastic left heart syndrome. J. Am. Coll. Cardiol. 24:1376–1381, 1994.
Binns, R. L., D. N. Ku, M. T. Stewart, J. P. Ansley, and K. A. Coyle. Optimal graft diameter: effect of wall shear stress on vascular healing. J. Vasc. Surg. 10:326–337, 1989.
Bove, E. L., F. Migliavacca, M. R. de Leval, R. Balossino, G. Pennati, T. R. Lloyd, et al. Use of mathematic modeling to compare and predict hemodynamic effects of the modified Blalock–Taussig and right ventricle–pulmonary artery shunts for hypoplastic left heart syndrome. J. Thorac. Cardiovasc. Surg. 136:312–320, 2008.
Caldarone, C. A., L. Benson, H. Holtby, J. Li, A. N. Redington, and G. S. Van Arsdell. Initial experience with hybrid palliation for neonates with single-ventricle physiology. Annals Thorac. Surg. 84:1294–1300, 2007.
Calderone, C. A., L. N. Benson, H. Holtby, and G. S. Van Arsdell. Main pulmonary artery to innominate artery shunt during hybrid palliation of hypoplastic left heart syndrome. J. Thorac. Cardiovasc. Surg. 130:e1–e2, 2005.
Ceballos, A. A coupled CFD-lumped parameter model of the human circulation: elucidating the hemodynamics of the hybrid Norwood palliative treatment and effects of the reverse blalock-taussig shunt placement and diameter. PhD Dissertation, University of Cental Florida, 2015.
Ceballos, A., I. R. Argueta-Morales, E. Divo, R. Osorio, C. A. Caldarone, A. J. Kassab, and W. M. DeCampli. Computational analysis of hybrid Norwood circulation with distal aortic arch obstruction and reverse Blalock–Taussig shunt. Annals Thorac. Surg. 94:1540–1550, 2012.
Ceballos, A., Divo, E., I. Argueta-Morales, C. Calderone, A. Kassab, and W. Decampli. A Multi-Scale CFD Analysis of the Hybrid Norwood Palliative Treatment for Hypoplastic Left Heart Syndrome: effect of the reverse Blalock-Taussig shunt diameter. ASME Paper IMECE 2013-66856, Proceedings of the 2013 International Congress and Exposition IMECE 2013, November 15–21, San Diego, California, USA.
Chandran, K. B., S. E. Rittgers, and A. P. Yoganathan. Biofluid Mechanics: The Human Circulation (2nd ed.). Boca Raton: CRC Press, 2012.
Corsini, C., G. Biglino, S. Schievano, T.-Y. Hsia, F. Migliavacca, G. Pennati, et al. The effect of modified Blalock–Taussig shunt size and coarctation severity on coronary perfusion after the Norwood operation. Ann. Thorac. Surg. 98:648–654, 2014.
Esmaily, M., B. Murtuza, T. Hsia, and A. Marsden. Simulations reveal adverse hemodynamics in patients with multiple systemic to pulmonary shunts. ASME J. Biomech. Eng. 137(3):031001-1–031001-12, 2015.
Galantowicz, M., J. P. Cheatham, A. Phillips, C. L. Cua, T. M. Hoffman, S. L. Hill, et al. Hybrid approach for hypoplastic left heart syndrome: intermediate results after the learning curve. Ann. Thorac. Surg. 85:2063–2071, 2008.
Ghista, D. N., and F. Kabinejadian. Coronary artery bypass grafting hemodynamics and anastomosis design: a biomedical engineering review. Biomed. Eng. Online 12:129, 2013.
Goubergrits, L., K. Affeld, E. Wellnhofer, T. Holmer, et al. Estimation of wall shear stress in bypass grafts with computational fluid dynamics method. Int. J. Artif. Organs 24:145–151, 2001.
Gutgesell, H. P., and J. Gibson. Management of hypoplastic left heart syndrome in the 1990s. Am. J. Cardiol. 89:842–846, 2002.
Hsia, T.-Y., D. Cosentino, C. Corsini, G. Pennati, G. Dubini, and F. Migliavacca. Use of mathematical modeling to compare and predict hemodynamic effects between hybrid and surgical Norwood palliations for hypoplastic left heart syndrome. Circulation 124:S204–S210, 2011.
Kroll, M. H., J. D. Hellums, L. V. McIntire, A. I. Schafer, and J. L. Moake. Platelets and shear stress. Blood 88:1525, 1996.
Laganà, K., R. Balossino, F. Migliavacca, G. Pennati, E. L. Bove, M. R. de Leval, et al. Multiscale modeling of the cardiovascular system: application to the study of pulmonary and coronary perfusions in the univentricular circulation. J. Biomech. 38:1129–1141, 2005.
Liu, J., Q. Sun, H. Hong, Y. Sun, J. Liu, Y. Qian, et al. Medical image-based hemodynamic analysis for modified Blalock–Taussig shunt. J. Mech. Med. Biol. 15(3):1550035, 2015.
Loth, F., S. A. Jones, C. K. Zarins, D. P. Giddens, R. F. Nassar, S. Glagov, et al. Relative contribution of wall shear stress and injury in experimental intimal thickening at PTFE end-to-side arterial anastomoses. J. Biomech. Eng. 124:44–51, 2002.
Malek, A. M., S. L. Alper, and S. Izumo. Hemodynamic shear stress and its role in atherosclerosis. J. Am. Med. Assoc. 282:2035–2042, 1999.
Migliavacca, F., K. Laganà, G. Pennati, M. R. de Leval, E. L. Bove, and G. Dubini. Global mathematical modelling of the Norwood circulation: a multiscale approach for the study of the pulmonary and coronary arterial perfusions. Cardiol. Young 14:71–76, 2004.
Migliavacca, F., G. Pennati, G. Dubini, R. Fumero, R. Pietrabissa, G. Urcelay, et al. Modeling of the Norwood circulation: effects of shunt size, vascular resistances, and heart rate. Am. J. Physiol. 280:H2076–H2086, 2001.
Moghadam, M. E., F. Migliavacca, I. E. Vignon-Clementel, T. Y. Hsia, A. L. Marsden, and M. C. H. Allianc. Optimization of shunt placement for the norwood surgery using multi-domain modeling. J. Biomech. Eng. Trans. ASME 134(5):051002, 2012.
Sheriff, J., D. Bluestein, G. Girdhar, and J. Jesty. High-shear stress sensitizes platelets to subsequent low-shear conditions. Ann. Biomed. Eng. 38(4):1442–1450, 2010.
Simaan, M. A., A. Ferreira, S. Chen, J. F. Antaki, and D. G. Galati. A dynamical state space representation and performance analysis of a feedback-controlled rotary left ventricular assist device. IEEE Trans. Controls Syst. Technol. 17:15–28, 2009.
Stoica, S. C., A. B. Philips, M. Egan, R. Rodeman, J. Chisolm, S. Hill, et al. The retrograde aortic arch in the hybrid approach to hypoplastic left heart syndrome. Ann. Thorac. Surg. 88:1939–1947, 2009.
Waite, L. Applied Biofluid Mechanics. New York: McGraw Hill Professional, 2007.
Waniewski, J., W. Kurowska, J. K. Mizerski, A. Trykozko, K. Nowiński, G. Brzezińska-Rajszys, et al. The effects of graft geometry on the patency of a systemic-to-pulmonary shunt: a computational fluid dynamics study. Artif. Organs 29:642–650, 2005.
FUNDING
Andres Ceballos received funding from the NIH NRSA predoctoral fellowship. Alain J. Kassab has received funding from the Orlando Health Foundation and from the American Heart Association under Grant No. 11GRNT7940011. William M. DeCampli has received funding from the American Heart Association under Grant No. 11GRNT7940011.
CONFLICT OF INTEREST
Ray Prather declares that he has no conflict of interest. Eduardo Divo declares that he has no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Dr. Ajit P. Yoganathan and Dr. Alison Marsden oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Ceballos, A., Prather, R., Divo, E. et al. Patient-Specific Multi-Scale Model Analysis of Hemodynamics Following the Hybrid Norwood Procedure for Hypoplastic Left Heart Syndrome: Effects of Reverse Blalock–Taussig Shunt Diameter. Cardiovasc Eng Tech 10, 136–154 (2019). https://doi.org/10.1007/s13239-018-00396-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-018-00396-w