Abstract
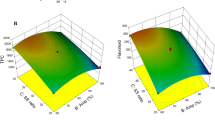
Banana peel is rich in phenolic compounds and is generally considered as waste. This study aimed to maximise recovery of phenolics from banana peel using water via microwave assisted extraction. The impact of various parameters including pH of solvent, sample to solvent ratio, irradiation time with/without cooling periods, and irradiation power were investigated individually. Following this, extraction conditions were further optimised using Response Surface Methodology. The results revealed that the extraction efficiency can be significantly improved by reducing the pH of water, increasing microwave power and time. However, cooling time during irradiation did not affect the extraction efficiency. Optimal conditions were identified at pH of 1, ratio of 2:100 g/mL, 6 min irradiation, and microwave power of 960 W. Under these optimal conditions, approximately 50.55 mg phenolics could be recovered from 1 g dried peel. These conditions are recommended for recovery of phenolic compounds from banana peel for further utilisation.



Similar content being viewed by others
References
Anal AK, Jaisanti S, Noomhorm A (2012) Enhanced yield of phenolic extracts from banana peels (Musa acuminata Colla AAA) and cinnamon barks (Cinnamomum varum) and their antioxidative potentials in fish oil. J Food Sci Technol 51:2632–2639. https://doi.org/10.1007/s13197-012-0793-x
Anhwange BA (2008) Chemical composition of Musa sapientum (banana) Peels. J Food Technol 6:263–266
Ballard TS, Mallikarjunan P, Zhou K, O’Keefe S (2010) Microwave-assisted extraction of phenolic antioxidant compounds from peanut skins. Food Chem 120:1185–1192. https://doi.org/10.1016/j.foodchem.2009.11.063
Bezerra MA, Santelli RE, Oliveira EP, Villar LS, Escaleira LA (2008) Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 76:965–977. https://doi.org/10.1016/j.talanta.2008.05.019
Chen VCP, Tsui K-L, Barton RR, Meckesheimer M (2006) A review on design, modeling and applications of computer experiments. IIE Trans 38:273–291. https://doi.org/10.1080/07408170500232495
Chirinos R, Rogez H, Campos D, Pedreschi R, Larondelle Y (2007) Optimization of extraction conditions of antioxidant phenolic compounds from mashua (Tropaeolum tuberosum Ruíz & Pavón) tubers. Sep Purif Technol 55:217–225. https://doi.org/10.1016/j.seppur.2006.12.005
Dahham SS, Mohamad T, Tabana YM, Majid A (2015) Antioxidant activities and anticancer screening of extracts from banana fruit (Musa sapientum). Acad J Cancer Res 8:28–34
Dahmoune F, Nayak B, Moussi K, Remini H, Madani K (2015) Optimization of microwave-assisted extraction of polyphenols from Myrtus communis L. leaves. Food Chem 166:585–595. https://doi.org/10.1016/j.foodchem.2014.06.066
FAOSTAT (2013) FAO statistical database (FAOSTAT). http://faostat.fao.org/site/567/DesktopDefault.aspx?PageID=567#ancor. Accessed 17 May 2016
Friedman M, Jürgens HS (2000) Effect of pH on the stability of plant phenolic compounds. J Agric Food Chem 48:2101–2110. https://doi.org/10.1021/jf990489j
González-Montelongo R, Gloria Lobo M, González M (2010) Antioxidant activity in banana peel extracts: testing extraction conditions and related bioactive compounds. Food Chem 119:1030–1039. https://doi.org/10.1016/j.foodchem.2009.08.012
Hayat K et al (2009) Optimized microwave-assisted extraction of phenolic acids from citrus mandarin peels and evaluation of antioxidant activity in vitro. Sep Purif Technol 70:63–70. https://doi.org/10.1016/j.seppur.2009.08.012
Kala HK, Mehta R, Sen KK, Tandey R, Mandal V (2016) Critical analysis of research trends and issues in microwave assisted extraction of phenolics: have we really done enough. TrAC Trends Anal Chem 85:140–152. https://doi.org/10.1016/j.trac.2016.09.007
Khayet M, Cojocaru C, Essalhi M (2011) Artificial neural network modeling and response surface methodology of desalination by reverse osmosis. J Membr Sci 368:202–214. https://doi.org/10.1016/j.memsci.2010.11.030
Kumar KPS, Bhowmik D, Duraivel S, Umadevi M (2012) Traditional and mecicinal uses of banana. J Pharmacogn Phytochem 1:53–63
Liazid A, Palma M, Brigui J, Barroso CG (2007) Investigation on phenolic compounds stability during microwave-assisted extraction. J Chromatogr A 1140:29–34. https://doi.org/10.1016/j.chroma.2006.11.040
Mohapatra D, Mishra S, Sutar N (2010) Banana and its by product utilisation. J Sci Ind Res 69:323–329
Nkhili E, Tomao V, El Hajji H, El Boustani ES, Chemat F, Dangles O (2009) Microwave-assisted water extraction of green tea polyphenols. Phytochem Anal 20:408–415. https://doi.org/10.1002/pca.1141
Palma M, Piñeiro Z, Barroso CG (2001) Stability of phenolic compounds during extraction with superheated solvents. J Chromatogr A 921:169–174. https://doi.org/10.1016/S0021-9673(01)00882-2
Pan X, Niu G, Liu H (2003) Microwave-assisted extraction of tea polyphenols and tea caffeine from green tea leaves. Chem Eng Process 42:129–133. https://doi.org/10.1016/S0255-2701(02)00037-5
Pereira A, Maraschin M (2015) Banana (Musa spp) from peel to pulp: ethnopharmacology, source of bioactive compounds and its relevance for human health. J Ethnopharmacol 160:149–163. https://doi.org/10.1016/j.jep.2014.11.008
Singh B, Singh JP, Kaur A, Singh N (2016) Bioactive compounds in banana and their associated health benefits—a review. Food Chem 206:1–11. https://doi.org/10.1016/j.foodchem.2016.03.033
Singh B, Singh N, Thakur S, Kaur A (2017) Ultrasound assisted extraction of polyphenols and their distribution in whole mung bean, hull and cotyledon. J Food Sci Technol 54:921–932. https://doi.org/10.1007/s13197-016-2356-z
Sirajudin ZNM, Ahmed QU, Chowdhury AJK, Kamarudin EZ, Khan AV, Uddin ABMH, Musa N (2014) Antimicrobial activity of banana (Musa paradisiaca L.) peels against food borne pathogenic microbes. J Pure Appl Microbio 8:3627–3639
Song J, Li D, Liu C, Zhang Y (2011) Optimized microwave-assisted extraction of total phenolics (TP) from Ipomoea batatas leaves and its antioxidant activity. Innov Food Sci Emerg Technol 12:282–287. https://doi.org/10.1016/j.ifset.2011.03.001
Sparr Eskilsson C, Björklund E (2000) Analytical-scale microwave-assisted extraction. J Chromatogr A 902:227–250. https://doi.org/10.1016/S0021-9673(00)00921-3
Sulaiman SF, Yusoff NAM, Eldeen IM, Seow EM, Sajak AAB, Supriatno Ooi KL (2011) Correlation between total phenolic and mineral contents with antioxidant activity of eight Malaysian bananas (Musa sp.). J Food Compost Anal 24:1–10. https://doi.org/10.1016/j.jfca.2010.04.005
Thaipong K, Boonprakob U, Crosby K, Cisneros-Zevallos L, Byrne DH (2006) Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J Food Compost Anal 19:669–675. https://doi.org/10.1016/j.jfca.2006.01.003
Vatai T, Škerget M, Knez Ž (2009) Extraction of phenolic compounds from elder berry and different grape marc varieties using organic solvents and/or supercritical carbon dioxide. J Food Eng 90:246–254. https://doi.org/10.1016/j.jfoodeng.2008.06.028
Vetal MD, Chavan RS, Rathod VK (2014) Microwave assisted extraction of ursolic acid and oleanolic acid from Ocimum sanctum. Biotechnol Bioprocess Eng 19:720–726. https://doi.org/10.1007/s12257-013-0798-y
Vu HT, Scarlett CJ, Vuong QV (2016a) Effects of drying conditions on physicochemical and antioxidant properties of banana (Musa cavendish) Peels. Drying Technol 35(9):1141–1151. https://doi.org/10.1080/07373937.2016.1233884
Vu HT, Scarlett CJ, Vuong QV (2016b) Optimization of ultrasound-assisted extraction conditions for recovery of phenolic compounds and antioxidant capacity from banana (Musa cavendish) peel. J Food Process Preserv 41:e13148. https://doi.org/10.1111/jfpp.13148
Vuong QV, Hirun S, Roach PD, Bowyer MC, Phillips PA, Scarlett CJ (2013) Effect of extraction conditions on total phenolic compounds and antioxidant activities of Carica papaya leaf aqueous extracts. J Herb Med 3:104–111. https://doi.org/10.1016/j.hermed.2013.04.004
Wu H, Xu FH, Hao J, Yang Y, Wang X (2015) Antihyperglycemic activity of banana (Musa nana Lour.) peel and its active ingredients in alloxan-Induced diabetic mice. In 3rd International conference on material, mechanical and manufacturing engineering, pp 231–238
Zhang B, Yang R, Liu C-Z (2008) Microwave-assisted extraction of chlorogenic acid from flower buds of Lonicera japonica Thunb. Sep Purif Technol 62:480–483. https://doi.org/10.1016/j.seppur.2008.02.013
Zhang H-F, Yang X-H, Wang Y (2011) Microwave assisted extraction of secondary metabolites from plants: current status and future directions. Trends Food Sci Technol 22:672–688. https://doi.org/10.1016/j.tifs.2011.07.003
Acknowledgements
The authors would like to thank Dr Dipangkar Kundu (The University of Sydney) and Ms Rebecca Richmond (The University of Newcastle) for proof reading and insightful review, which has significantly contributed to the quality of the manuscript. We would also like to thank Dr Mark Tarleton and Assoc. Prof. Michael Bowyer for their advice concerning the chemistry in this project. The financial support for this study was from the Vietnamese Government through the Vietnam International Education Development-Ministry of Education and Training, and the University of Newcastle. The awarding of a VIED-TUIT scholarship to the first author is greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Vu, H.T., Scarlett, C.J. & Vuong, Q.V. Maximising recovery of phenolic compounds and antioxidant properties from banana peel using microwave assisted extraction and water. J Food Sci Technol 56, 1360–1370 (2019). https://doi.org/10.1007/s13197-019-03610-2
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-019-03610-2