Abstract
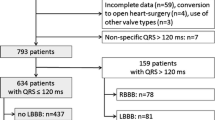
Despite a considerable improvement in TAVR devices and procedures, together with a reduction in procedural complications, the rate of conduction disturbances (CD) remained stable over the years. Indeed, the CD rate is still significantly higher than in surgical aortic valve replacement, and represents one of the main limitations to the expansion of TAVR to younger low-risk patients. The aim of the present study was to assess the incidence and predictors of CD in low-risk patients undergoing TAVR. Among 637 patients without preexisting CD who underwent TAVR, 116 (18.2%) were considered at low surgical risk. Up to 25% of low-risk patients presented with persistent CD at discharge. The pacemaker implantation rate was similar in the low-risk group compared to the intermediate-/high-risk group (8.7% vs 10.6%, p = 0.55). Moreover, the rate of new persistent left bundle branch block (LBBB) following TAVR was also similar between both groups (18.1% vs 22.1%, p = 0.34). At 1-year follow-up, LBBB was persistent in 62.5% of patients and 3 of them required a pacemaker implantation. Depth of valve implantation, baseline QRS duration and mean aortic transvalvular gradient were identified as independent predictors of CD in low-risk patients. Patients at low surgical risk showed an equivalent CD rate than intermediate-/high-risk patients. The depth of valve implantation was the main predictor of CD in low-risk patients undergoing TAVR. Baseline QRS duration and mean aortic transvalvular gradient were also associated with increased CD.
Similar content being viewed by others
References
Barker CM, Reardon MJ. Should TAVR replace surgery for aortic stenosis in low- to intermediate-risk patients? Can J Cardiol. 2017;33(9):1124–8.
Baumgartner H, Falk V, Bax JJ, et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2017;38(36):2739–91.
Leon MB, Smith CR, Mack MJ, et al. Transcatheter or surgical aortic-valve replacement in intermediate-risk patients. N Engl J Med. 2016;374(17):1609–20.
Popma JJ, Deeb GM, Yakubov SJ, et al. transcatheter aortic-valve replacement with a self-expanding valve in low-risk patients. N Engl J Med. 2019;380(18):1706–15.
Mack MJ, Leon MB, Thourani VH, et al. Transcatheter aortic-valve replacement with a balloon-expandable valve in low-risk patients. N Engl J Med. 2019;380(18):1695–705.
De Torres-Alba F, Kaleschke G, Diller G, et al. Changes in the pacemaker rate after transition from edwards SAPIEN XT to SAPIEN 3 transcatheter aortic valve implantation the critical role of valve implantation height. JACC Cardiovasc Interv. 2016;9(8):805–13.
Auffret V, Puri R, Urena M, et al. Conduction disturbances after transcatheter aortic valve replacement: current status and future perspectives. Circulation. 2017;136(11):1049–69.
Rodés-cabau J, Ellenbogen KA, Krahn AD, et al. Management of conduction disturbances associated with transcatheter aortic valve replacement. JACC Scientific Expert Panel. J Am Coll Cardiol. 2019;74(8):1086–106.
Kappetein AP, Head SJ, Généreux P, et al. Updated standardized endpoint definitions for transcatheter aortic valve implantation: the Valve Academic Research Consortium-2 consensus document. Eur Heart J. 2012;33(19):2403–18.
Kligfield P, Gettes LS, Bailey JJ, et al. Recommendations for the standardization and interpretation of the electrocardiogram: part I: the electrocardiogram and its technology: a scientific statement from the American Heart Association Electrocardiography and Arrhythmias Committee. Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society: endorsed by the International Society for Computerized Electrocardiology. Circulation. 2007;115(10):1306–24.
Epstein AE, DiMarco JP, Ellenbogen KA, et al. 2012 ACCF/AHA/HRS focused update incorporated into the ACCF/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guide. Circulation. 2013;127(3):e283–352.
Regueiro A, Abdul-Jawad Altisent O, Del Trigo M, et al. Impact of new-onset left bundle branch block and periprocedural permanent pacemaker implantation on clinical outcomes in patients undergoing transcatheter aortic valve replacement: a systematic review and meta-analysis. Circ Cardiovasc Interv. 2016;9(5).
Rodés-Cabau J, Urena M, Nombela-Franco L, et al. Arrhythmic burden as determined by ambulatory continuous cardiac monitoring in patients with new-onset persistent left bundle branch block following transcatheter aortic valve replacement: the MARE Study. JACC Cardiovasc Interv. 2018;11(15):1495–505.
Urena M, Webb JG, Tamburino C, et al. Permanent pacemaker implantation after transcatheter aortic valve implantation: impact on late clinical outcomes and left ventricular function. Circulation. 2014;129(11):1233–43.
Chamandi C, Barbanti M, Munoz-Garcia A, et al. Long-term outcomes in patients with new permanent pacemaker implantation following transcatheter aortic valve replacement. JACC Cardiovasc Interv. 2018;11(3):301–10.
Søndergaard L, Steinbrüchel DA, Ihlemann N, et al. Two-year outcomes in patients with severe aortic valve stenosis randomized to transcatheter versus surgical aortic valve replacement: the all-comers nordic aortic valve intervention randomized clinical trial. Circ Cardiovasc Interv. 2016;9(6).
Popma JJ, Reardon MJ, Khabbaz K, et al. Early clinical outcomes after transcatheter aortic valve replacement using a novel self-expanding bioprosthesis in patients with severe aortic stenosis who are suboptimal for surgery: results of the evolut R U.S. Study. JACC Cardiovasc Interv. 2017;10(3):268–75.
Arai T, Lefèvre T, Hovasse T, et al. Comparison of Edwards SAPIEN 3 versus SAPIEN XT in transfemoral transcatheter aortic valve implantation: difference of valve selection in the real world. J Cardiol. 2017;69(3):565–9.
Herrmann HC, Thourani VH, Kodali SK, et al. One-year clinical outcomes with SAPIEN 3 transcatheter aortic valve replacement in high-risk and inoperable patients with severe aortic stenosis. Circulation. 2016;134(2):130–40.
Vahanian A, Urena M. The results of transcatheter aortic valve replacement continue to improve: the specific example of a self-expandable transcatheter heart valve in a real-life registry. JACC Cardiovasc Interv. 2017;10(20):2099–100.
Möllmann H, Hengstenberg C, Hilker M, et al. Real-world experience using the ACURATE neo prosthesis: 30-day outcomes of 1,000 patients enrolled in the SAVI TF registry. EuroIntervention. 2018;13(15):e1764–70.
Urena M, Mok M, Serra V, et al. Predictive factors and long-term clinical consequences of persistent left bundle branch block following transcatheter aortic valve implantation with a balloon-expandable valve. J Am Coll Cardiol. 2012;60(18):1743–52.
Mauri V, Reimann A, Stern D, et al. Predictors of permanent pacemaker implantation after transcatheter aortic valve replacement with the SAPIEN 3. JACC Cardiovasc Interv. 2016;9(21):2200–9.
Husser O, Pellegrini C, Kessler T, et al. Predictors of permanent pacemaker implantations and new-onset conduction abnormalities with the SAPIEN 3 balloon-expandable transcatheter heart valve. JACC Cardiovasc Interv. 2016;9(3):244–54.
Nazif TM, Dizon JM, Hahn RT, et al. Predictors and clinical outcomes of permanent pacemaker implantation after transcatheter aortic valve replacement: the PARTNER (Placement of AoRtic TraNscathetER Valves) trial and registry. JACC Cardiovasc Interv. 2015;8(1 Pt A):60–9.
Monteiro C, Ferrari ADL, Caramori PRA, et al. Permanent pacing after transcatheter aortic valve implantation: incidence, predictors and evolution of left ventricular function. Arq Bras Cardiol. 2017;109(6):550–9.
Gaede L, Kim WK, Liebetrau C, et al. Pacemaker implantation after TAVI: predictors of AV block persistence. Clin Res Cardiol. 2018;107(1):60–9.
Latsios G, Gerckens U, Buellesfeld L, et al. “Device landing zone” calcification, assessed by MSCT, as a predictive factor for pacemaker implantation after TAVI. Catheter Cardiovasc Interv. 2010;76(3):431–9.
Watanabe Y, Kozuma K, Hioki H, et al. Pre-existing right bundle branch block increases risk for death after transcatheter aortic valve replacement with a balloon-expandable valve. JACC Cardiovasc Interv. 2016;9(21):2210–6.
Hamdan A, Klempfner R, Konen E, et al. Inverse relationship between membranous septal length and the risk of atrioventricular block in patients undergoing transcatheter aortic valve implantation. JACC Cardiovasc Interv. 2015;8(9):1218–28.
Acknowledgements
This study will be included in a Medicine PhD thesis at the Autonomous University of Barcelona.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. All authors have reported that they have no relationships with industry to disclose.
Ethical approval
Our TAVR database and the present study were approved by the institutional research ethics committee and certify that the study was performed in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Informed consent was obtained from all patients included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zouari, F., Campelo-Parada, F., Matta, A. et al. Conduction disturbances in low-surgical-risk patients undergoing transcatheter aortic valve replacement with self-expandable or balloon-expandable valves. Cardiovasc Interv and Ther 36, 355–362 (2021). https://doi.org/10.1007/s12928-020-00687-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12928-020-00687-x