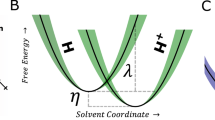
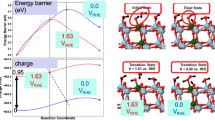
Abstract
The catalytic oxidation of methanol on Pt(111) has been addressed based on first-principles electronic structure calculations. The chemical environment corresponding to the conditions in heterogeneous and electro-catalysis has been taken into account in a grand-canonical approach. Furthermore, the aqueous electrolyte in electrocatalysis has been described in an implicit solvent model. Thus, we find characteristic differences between the methanol oxidation paths in heterogeneous and electro-catalysis. The presence of the aqueous electrolyte stabilizes reaction intermediates containing hydrophilic groups thus also influencing the selectivity in the methanol oxidation. In addition, adsorbed hydrogen on Pt(111) is shown to render the electro-oxidation of methanol less efficient.

The difference between methanol oxidation in heterogeneous and electro-catalysis has been studied theoretically from first principles employing a grand-canonical approach.





Similar content being viewed by others
References
R. Schlögl, The role of chemistry in the energy challenge. ChemSusChem. 3, 209 (2010)
C. Hartnig, P. Vassilev, M.T.M. Koper, Ab initio classical molecular dynamics studies of electrode reactions. Electrochim. Acta. 48, 3751 (2003)
C. Hartnig, E. Spohr, The role of water in the initial steps of methanol oxidation on Pt(111). Chem. Phys. 319, 185 (2005)
M.J. Janik, C.D. Taylor, M. Neurock, First principles analysis of the electrocatalytic oxidation of methanol and carbon monoxide. Top. Catal. 46, 306 (2007). doi:10.1007/s11244-007-9004-9
J.A. Keith, T. Jacob, Theoretical studies of potential-dependent and competing mechanisms of the electrocatalytic oxygen reduction reaction on Pt(111). Angew. Chem. Int. Ed. 49, 9521 (2010). doi:10.1002/anie.201004794
F. Calle-Vallejo, M.T. Koper, First-principles computational electrochemistry: achievements and challenges. Electrochim. Acta. 84(0), 3 (2012). doi:10.1016/j.electacta.2012.04.062
P. Quaino, E. Santos, H. Wolfschmidt, M. Montero, U. Stimming, Theory meets experiment: electrocatalysis of hydrogen oxidation/evolution at Pd-Au nanostructures. Catal. Today. 177, 55 (2011). doi:10.1016/j.cattod.2011.05.004
P. Quaino, N. Luque, G. Soldano, R. Nazmutdinov, E. Santos, T. Roman, A. Lundin, A. Groß, W. Schmickler, Solvated protons in density functional theory—few examples. Electrochim. Acta. 105, 248 (2013)
K. Chan, J.K. Nørskov, Electrochemical barriers made simple. J. Phys. Chem. Lett. 6(14), 2663 (2015). doi:10.1021/acs.jpclett.5b01043
A. Groß, F. Gossenberger, X. Lin, M. Naderian, S. Sakong, T. Roman, Water structures at metal electrodes studied by ab initio molecular dynamics simulations. J. Electrochem. Soc. 161(8), E3015 (2014). doi:10.1149/2.003408jes
T. Roman, F. Gossenberger, K. Forster-Tonigold, A. Groß, Halide adsorption on close-packed metal electrodes. Phys. Chem. Chem. Phys. 16, 13630 (2014). doi:10.1039/C4CP00237G
F. Gossenberger, T. Roman, K. Forster-Tonigold, A. Groß, Change of the work function of platinum electrodes induced by halide adsorption. Beilstein J. Nanotechnol. 5, 152 (2014). doi:10.3762/bjnano.5.15
N. G. Hörmann, M. Jäckle, F. Gossenberger, T. Roman, K. Forster-Tonigold, M. Naderian, S. Sakong, A. Groß, Some challenges in the first-principles modeling of structures and processes in electrochemical energy storage and transfer. J. Power Sources. 275, 531 (2015). doi:10.1016/j.jpowsour.2014.10.198
S. Sakong, M. Naderian, K. Mathew, R.G. Hennig, A. Groß, Density functional theory study of the electrochemical interface between a Pt electrode and an aqueous electrolyte using an implicit solvent method. J. Chem. Phys. 142(23), 234107 (2015). doi:10.1063/1.4922615
F. Gossenberger, T. Roman, A. Groß, Equilibrium coverage of halides on metal electrodes. Surf. Sci. 631, 17 (2015). doi:10.1016/j.susc.2014.01.021
S. Sakong, A. Groß, The importance of the electrochemical environment in the electro-oxidation of methanol on Pt(111). ACS Catal. 6, 5575 (2016). doi:10.1021/acscatal.6b00931
S. Sakong, K. Forster-Tonigold, A. Groß, The structure of water at a Pt(111) electrode and the potential of zero charge studied from first principles. J. Chem. Phys. 144(19), 194701 (2016). doi:10.1063/1.4948638
X. Lin, F. Evers, A. Groß, First-principles study of the structure of water layers on flat and stepped Pb electrodes. Beilstein J. Nanotechnol. 7, 533 (2016). doi:10.3762/bjnano.7.47
F. Gossenberger, T. Roman, A. Groß, Hydrogen and halide co-adsorption on Pt(111) in an electrochemical environment: A computational perspective. Electrochim. Acta. 216, 152 (2016). doi:10.1016/j.electacta.2016.08.117
M. Mehlhorn, S. Schnur, A. Groß, K. Morgenstern, Molecular-scale imaging of water near charged surfaces. ChemElectroChem. 1(2), 431 (2014). doi:10.1002/celc.201300063
S. Schnur, A. Groß, Properties of metal–water interfaces studied from first principles. J. Phys. 11(12), 125003 (2009). doi:10.1088/1367-2630/11/12/125003
S. Schnur, A. Groß, Challenges in the first-principles description of reactions in electrocatalysis. Catal. Today. 165(1), 129 (2011). doi:10.1016/j.cattod.2010.11.071
A. Ignaczak, R. Nazmutdinov, A. Goduljan, L.M. De Campos Pinto, F. Juarez, P. Quaino, E. Santos, W. Schmickler, A scenario for oxygen reduction in alkaline media. Nano Energy. 26, 558 (2016). doi:10.1016/j.nanoen.2016.06.001
C. Hartnig, M.T.M. Koper, Solvent reorganization in electron and ion transfer reactions near a smooth electrified surface: a molecular dynamics study. J. Am. Chem. Soc. 125, 9840 (2003)
K. Reuter, D. Frenkel, M. Scheffler, The steady state of heterogeneous catalysis, studied by first-principles statistical mechanics. Phys. Rev. Lett. 93, 116105 (2004)
K. Reuter, M. Scheffler, First-principles kinetic monte carlo simulations for heterogeneous catalysis: Application to the CO oxidation at RuO 2(110). Phys. Rev. B. 73, 045433 (2006)
J. Rogal, K. Reuter, M. Scheffler, CO oxidation at Pd(100): a first-principles constrained thermodynamics study. Phys. Rev. B. 75, 205433 (2007). doi:10.1103/PhysRevB.75.205433
C. Shi, H.A. Hansen, A.C. Lausche, J.K. Norskov, Trends in electrochemical co2 reduction activity for open and close-packed metal surfaces. Phys. Chem. Chem. Phys. 16, 4720 (2014). doi:10.1039/C3CP54822H
J.K. Nørskov, J. Rossmeisl, A. Logadottir, L. Lindqvist, J.R. Kitchin, T. Bligaard, H. Jónsson, Origin of the overpotential for oxygen reduction at a fuel-cell cathode. J. Phys. Chem. B. 108(46), 17886 (2004). doi:10.1021/jp047349j
E. Skulason, G.S. Karlberg, J. Rossmeisl, T. Bligaard, J. Greeley, H. Jonsson, J.K. Norskov, Density functional theory calculations for the hydrogen evolution reaction in an electrochemical double layer on the Pt(111) electrode. Phys. Chem. Chem. Phys. 9, 3241 (2007). doi:10.1039/B700099E
J.K. Nørskov, F. Abild-Pedersen, F. Studt, T. Bligaard, Density functional theory in surface chemistry and catalysis. Proc. Natl. Acad. Sci. 108(3), 937 (2011). doi:10.1073/pnas.1006652108
A.R. Leach. Molecular modelling: Principles and applications, 2nd edn. (Pearson, Harlow, 2001)
A. Roudgar, A. Groß, Water bilayer on the Pd/Au(111) overlayer system: coadsorption and electric field effects. Chem. Phys. Lett. 409, 157 (2005)
A. Michaelides, Density functional theory simulations of water-metal interfaces: Waltzing waters, a novel 2D ice phase, and more. Appl. Phys. A. 85, 415 (2006)
D. Gunceler, K. Letchworth-Weaver, R. Sundararaman, K.A. Schwarz, T.A. Arias, The importance of nonlinear fluid response in joint density-functional theory studies of battery systems. Model. Simul. Mater. Sc. 21 (7), 074005 (2013). doi:10.1088/0965-0393/21/7/074005
K. Letchworth-Weaver, T.A. Arias, Joint density functional theory of the electrode-electrolyte interface: Application to fixed electrode potentials, interfacial capacitances, and potentials of zero charge. Phys. Rev. B. 86, 075140 (2012). doi:10.1103/PhysRevB.86.075140
S.A. Petrosyan, A.A. Rigos, T.A. Arias, Joint density-functional theory: ab initio study of cr2o3 surface chemistry in solution. J. Phys. Chem. B. 109(32), 15436 (2005). doi:10.1021/jp044822k
S.A. Petrosyan, J.F. Briere, D. Roundy, T.A. Arias, Joint density-functional theory for electronic structure of solvated systems. Phys. Rev. B. 75, 205105 (2007). doi:10.1103/PhysRevB.75.205105
A. Arico, V. Baglio, A.D. Blasi, E. Modica, P. Antonucci, V. Antonucci, Analysis of the high-temperature methanol oxidation behaviour at carbon-supported pt–ru catalysts. J. Electroanal. Chem. 557, 167 (2003). doi:10.1016/S0022-0728(03)00369-3
R. Reichert, J. Schnaidt, Z. Jusys, R.J. Behm, The influence of reactive side products on the electrooxidation of methanol - a combined in situ infrared spectroscopy and online mass spectrometry study. Phys. Chem. Chem. Phys. 16, 13780 (2014). doi:10.1039/C4CP01229A
G. Kresse, J. Furthmüller, Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B. 54, 11169 (1996). doi:10.1103/PhysRevB.54.11169
B. Hammer, L.B. Hansen, J.K. Nørskov, Improved adsorption energetics within density-functional theory using revised Perdew-Burke-Ernzerhof functionals. Phys. Rev. B. 59, 7413 (1999)
S. Grimme, J. Antony, S. Ehrlich, H. Krieg, A consistent and accurate ab initio parametrization of density functional dispersion correction (dft-d) for the 94 elements h-pu. J. Chem. Phys. 132(15), 154104 (2010). doi:10.1063/1.3382344
S. Grimme, Density functional theory with london dispersion corrections. Wiley Interdiscip. Rev. Comput. Mol. Sci. 1(2), 211 (2011). doi:10.1002/wcms.30
S. Grimme, A. Hansen, J.G. Brandenburg, C. Bannwarth, Dispersion-corrected mean-field electronic structure methods. Chem. Rev. 116(9), 5105 (2016). doi:10.1021/acs.chemrev.5b00533
K. Tonigold, A. Groß, Dispersive interactions in water bilayers at metallic surfaces: A comparison of the pbe and rpbe functional including semiempirical dispersion corrections. J. Comput. Chem. 33(6), 695 (2012). doi:10.1002/jcc.22900
K. Forster-Tonigold, A. Groß, Dispersion corrected rpbe studies of liquid water. J. Chem. Phys. 141, 064501 (2014). doi:10.1063/1.4892400
B. Koslowski, A. Tschetschetkin, N. Maurer, P. Ziemann, J. Kuc̆era, A. Groß, 4,4’-dithiodipyridine on Au(111): a combined STM, STS, and DFT study. J. Phys. Chem. C. 117, 20060 (2013)
K. Mathew, R. Sundararaman, K. Letchworth-Weaver, T.A. Arias, R.G. Hennig, Implicit solvation model for density-functional study of nanocrystal surfaces and reaction pathways. J. Chem. Phys. 140, 084106 (2014). doi:10.1063/1.4865107
P.E. Blöchl, Projector augmented-wave method. Phys. Rev. B. 50, 17953 (1994). doi:10.1103/PhysRevB.50.17953
G. Mercurio, E.R. McNellis, I. Martin, S. Hagen, F. Leyssner, S. Soubatch, J. Meyer, M. Wolf, P. Tegeder, F.S. Tautz, K. Reuter, Structure and energetics of azobenzene on ag(111): Benchmarking semiempirical dispersion correction approaches. Phys. Rev. Lett. 104, 036102 (2010). doi:10.1103/PhysRevLett.104.036102
M. Fishman, H.L. Zhuang, K. Mathew, W. Dirschka, R.G. Hennig, Accuracy of exchange-correlation functionals and effect of solvation on the surface energy of copper. Phys. Rev. B. 87, 245402 (2013). doi:10.1103/PhysRevB.87.245402
O. Andreussi, I. Dabo, N. Marzari, Revised self-consistent continuum solvation in electronic-structure calculations. J. Chem. Phys. 136(6), 064102 (2012). doi:10.1063/1.3676407
O. Andreussi, N. Marzari, Electrostatics of solvated systems in periodic boundary conditions. Phys. Rev. B. 90, 245101 (2014). doi:10.1103/PhysRevB.90.245101
S. Günther, L. Zhou, M. Hävecker, A. Knop-Gericke, E. Kleimenov, R. Schlögl, R. Imbihl, Adsorbate coverages, surface reactivity in methanol oxidation over Cu(110) A in situ photoelectron spectroscopy study. J. Chem. Phys. 125, 114709 (2006)
S. Sakong, A. Groß, Density functional theory study of the partial oxidation of methanol on copper surfaces. J. Catal. 231, 420 (2005). doi:10.1016/j.jcat.2005.02.009
S. Sakong, A. Groß, Total oxidation of methanol on Cu(110): a density functional theory study. J. Phys. Chem. A. 111, 8814 (2007). doi:10.1021/jp072773g
W. Greiner, L. Neise, H. Stöcker. Thermodynamik und Statistische Mechanik (Harri Deutsch, Frankfurt am Main, 1987)
S. Sakong, Y.A. Du, P. Kratzer, Interface defects and impurities at the growth zone of Au-catalyzed GaAs nanowire from first principles. Phys. Status Solidi Rapid Res. Lett. 7(10), 882 (2013). doi:10.1002/pssr.201307210
K. Reuter, M. Scheffler, Composition, structure, and stability of RuO2(110) as a function of oxygen pressure. Phys. Rev. B. 65, 035406 (2001). doi:10.1103/PhysRevB.65.035406
T. Iwasita, Electrocatalysis of methanol oxidation. Electrochim. Acta. 47(22–23), 3663 (2002). doi:10.1016/S0013-4686(02)00336-5
A.B. Anderson, H.A. Asiri, Reversible potentials for steps in methanol and formic acid oxidation to CO 2; adsorption energies of intermediates on the ideal electrocatalyst for methanol oxidation and CO 2 reduction. Phys. Chem. Chem. Phys. 16, 10587 (2014). doi:10.1039/C3CP54837F
F. Calle-Vallejo, J. Tymoczko, V. Colic, Q.H. Vu, M.D. Pohl, K. Morgenstern, D. Loffreda, P. Sautet, W. Schuhmann, A.S. Bandarenka, Finding optimal surface sites on heterogeneous catalysts by counting nearest neighbors. Science. 350(6257), 185 (2015). doi:10.1126/science.aab3501
J. Greeley, M. Mavrikakis, A first-principles study of methanol decomposition on Pt(111). J. Am. Chem. Soc. 124, 7193 (2002)
J. Greeley, M. Mavrikakis, Competitive paths for methanol decomposition on Pt(111). J. Am. Chem. Soc. 126, 3910 (2004). doi:10.1021/ja037700z
S.K. Desai, M. Neurock, K. Kourtakis, A periodic density functional theory study of the dehydrogenation of methanol over Pt(111). J. Phys. Chem. B. 106(10), 2559 (2002). doi:10.1021/jp0132984
D. Cao, G.Q. Lu, A. Wieckowski, S.A. Wasileski, M. Neurock, Mechanisms of methanol decomposition on platinum: a combined experimental and ab initio approach. J. Phys. Chem. B. 109(23), 11622 (2005). doi:10.1021/jp0501188
C.D. Taylor, S.A. Wasileski, J.S. Filhol, M. Neurock, First principles reaction modeling of the electrochemical interface: Consideration and calculation of a tunable surface potential from atomic and electronic structure. Phys. Rev. B. 73, 165402 (2006). doi:10.1103/PhysRevB.73.165402
H.F. Wang, Z.P. Liu, Formic acid oxidation at Pt/H 2O interface from periodic DFT calculations integrated with a continuum solvation model. J. Phys. Chem. C. 113(40), 17502 (2009). doi:10.1021/jp9059888
Y.H. Fang, Z.P. Liu, Tafel kinetics of electrocatalytic reactions: From experiment to first-principles. ACS Catal. 4(12), 4364 (2014). doi:10.1021/cs501312v
Y.H. Fang, Z.P. Liu, First principles tafel kinetics of methanol oxidation on Pt(111). Surf. Sci. 631, 42 (2015). doi:10.1016/j.susc.2014.05.014
R. Reichert, J. Schnaidt, Z. Jusys, R.J. Behm, The influence of reactive side products in electrocatalytic reactions: Methanol oxidation as case study. ChemPhysChem. 14(16), 3678 (2013). doi:10.1002/cphc.201300726
K. Franaszczuk, E. Herrero, P. Zelenay, A. Wieckowski, J. Wang, R.I. Masel, A comparison of electrochemical and gas-phase decomposition of methanol on platinum surfaces. J. Phys. Chem. 96(21), 8509 (1992). doi:10.1021/j100200a056
J. Wintterlin, R. Schuster, G. Ertl, Existence of a ”hot” atom mechanism for the dissociation of O 2 on Pt(111). Phys. Rev. Lett. 77, 123 (1996)
A. Eichler, J. Hafner, Molecular precursors in the dissociative adsorption of O 2 on Pt(111). Phys. Rev. Lett. 79, 4481 (1997). doi:10.1103/PhysRevLett.79.4481
A. Groß, A. Eichler, J. Hafner, M.J. Mehl, D.A. Papaconstantopoulos, Ab initio based tight-binding molecular dynamics simulation of the sticking and scattering of O 2/Pt(111). J. Chem. Phys. 174713, 124 (2006)
L.C. Grabow, A.A. Gokhale, S.T. Evans, J.A. Dumesic, M. Mavrikakis, Mechanism of the water gas shift reaction on Pt: First principles, experiments, and microkinetic modeling. J. Phys. Chem. C. 112(12), 4608 (2008). doi:10.1021/jp7099702
E.M. Karp, C.T. Campbell, F. Studt, F. Abild-Pedersen, J.K. Nørskov, Energetics of oxygen adatoms, hydroxyl species and water dissociation on Pt(111). J. Phys. Chem. C. 116(49), 25772 (2012). doi:10.1021/jp3066794
J.C. Fajín, M.N.D.S. Cordeiro, J.B. Gomes, Density functional theory study of the water dissociation on platinum surfaces: General trends. J. Phys. Chem. A. 118(31), 5832 (2014). doi:10.1021/jp411500j
A. Groß, Dynamical quantum processes of molecular beams at surfaces: dissociative adsorption of hydrogen on metal surfaces. Surf. Sci. 363, 1 (1996)
X. Lin, F. Gossenberger, A. Groß, Ionic adsorbate structures on metal electrodes calculated from first-principles. Ind. Eng. Chem. Res. 55(42), 11107 (2016). doi:10.1021/acs.iecr.6b03087
Acknowledgements
This research has been supported by the German Research Foundation (DFG) through contract GR 1503/21-2. The authors acknowledge the computer time supported by the state of Baden-Württemberg through the bwHPC project and the Germany Research Foundation (DFG) through grant number INST 40/467-1 FUGG.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakong, S., Groß, A. Methanol Oxidation on Pt(111) from First-Principles in Heterogeneous and Electrocatalysis. Electrocatalysis 8, 577–586 (2017). https://doi.org/10.1007/s12678-017-0370-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12678-017-0370-1