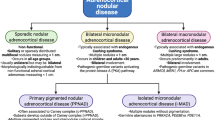
Abstract
Papillary thyroid cancer (PTC) is the most prevalent of all endocrine cancers. In recent studies, the presence of receptors for pituitary-type growth hormone-releasing hormone (pGHRH-R) has been demonstrated in various human cancers, including human prostate, brain, and other cancer lines. Thyroid malignancies, however, have not yet been investigated in this regard. In this study, we found that pGHRH-R and its functional splice variant, SV1, are present in normal thyroid and PTC cells. We also treated seven normal and PTC tumor thyroid cells in vitro with a GHRH antagonist, MIA-602, to compare its anti-proliferation and anti-invasion potential against vehicle-treated cells. We found that treatment with GHRH antagonist increases the expression of SV1 and pGHRH-R in tumor cells compared to tumor cells exposed to vehicle only, a response which may alter the sensitivity of signaling kinases within the cells. GHRH antagonist treatment of tumor cells also reduced activity of the tumor invasion marker, matrix metalloproteinase (MMP)-2, compared to tumor cells exposed to vehicle only. The expression of pGHRH-R and SV1, as well as MMP-2 activity, in normal thyroid cells remained unaffected by GHRH antagonist treatment. Similarly, cell proliferation rates for tumor or normal thyroid cells were not affected by GHRH antagonist treatment. Our findings have important implications for the therapeutic use of GHRH antagonist in cases of aggressive PTC refractory to conventional treatment modalities, and in which protein expression and MMP-2 activity in normal thyroid tissue is left unaltered.
Similar content being viewed by others
Introduction
Thyroid cancer is the most common endocrine malignancy and has one of the most rapidly increasing prevalences of any cancer in the USA. Of the differentiated variants, papillary thyroid cancer (PTC) accounts for 85–88 % of cases, while follicular cancers comprise 9–10 % and other variants such as Hürthle cell or oxyphil tumors comprise the remaining 3–5 % [1, 2]. Between 1973 and 2002, the incidence of PTC showed an increase from 3.6 to 8.7 per 100,000 in the USA, a 2.4-fold increase, partially attributed to its improved detection of PTC [1, 2]. While the standard of care is surgical resection followed by radioactive iodine therapy and thyroid hormone supplementation, a minority of cases may not respond adequately [3]. Effective treatments do not exist for patients with metastatic differentiated thyroid carcinomas that fail medical therapy, with the exception of vemurafenib in patients with advanced PTC harboring the BRAFV600E mutation [4, 5]. For these reasons, thyroid cancer has become an important public health issue [4].
Given the limited effectiveness of standard systemic chemotherapies, targeted molecular treatments have taken a major role in recent years. Matrix metalloproteinases (MMP), which are markers of a tumor’s invasion potential, have been shown to have a role in the prognosis associated with thyroid malignancy [6–8]. Their function may therefore be a therapeutic target, especially for aggressive or refractory variants. Antagonists of growth hormone-releasing hormone (GHRH) have been shown to effectively reduce MMP activity; however, this therapeutic pathway has not been investigated in PTC [9, 10]. In this study, we isolated cells from human PTC and normal thyroid tissues to confirm the presence of receptors for pituitary-type GHRH (pGHRH-R) and to evaluate responses to GHRH antagonist treatment.
Materials and Methods
Drugs and Chemicals
MIA-602, a GHRH antagonist, was synthesized in the laboratory of one of us (A.V.S.) by solid-phase methodology using Boc-chemistry as previously described [9, 11]. MIA-602 has the sequence: [(PhAc-Ada)0-Tyr1, D-Arg2, Fpa56, Ala8, Har9, Tyr(Me)10, His11, Orn12, Abu15, His20, Orn21, Nle27, D-Arg28, and Har29]hGHRH(1–29)NH2, where Ada is 12-aminododecanoic acid, Fpa5 is pentafluoro-phenylalanine, Orn is ornithine, Har is homoarginine, Nle is norleucine, PhAc is phenylacetyl, and Tyr(Me) is O-methyl-Tyr. For use, stock solutions of MIA-602 were dissolved in dimethyl sulfoxide (DMSO) at 1 μM concentration. The final concentration of DMSO after dilution with incubation medium was low (<0.1 %) to avoid any DMSO effect on growth in cell culture.
Tissue Collection and Cell Culture
Surgical specimens of thyroid tumors and normal thyroid tissue were collected from women undergoing total thyroidectomy for PTC. To achieve homogeneity in our specimens, we limited our cohort to postmenopausal women ≥55 years of age. Seven normal thyroid samples and seven PTC samples were studied, with TNM stages ranging from T1N0M0 to T2N1M0. Informed consent was obtained per the requirements of the Institutional Review Board at the University of Miami. Primary thyroid cancer tissue was isolated and separated from normal thyroid tissue, which was obtained from the contralateral side of the thyroid gland. PTC was confirmed by final histopathology. Specimens were incubated for 15 min at 37 °C in 2 % dispase and phosphate-buffered saline. The tissue was gently minced to allow for collection of individual cells. Cells were suspended in Dulbecco’s modified Eagle medium (DMEM; Life Technologies Corporation, Grand Island, NY)/F:12 plus 10 % fetal bovine serum and incubated at 37 °C for 4–6 weeks in 10 % CO2 atmosphere. After achieving confluency, the cells were trypsinized and plated. All experiments were performed on cells between passages 1–2.
Proliferation Assays
Proliferation assays were performed for four experimental groups: thyroid tumor + MIA-602, thyroid tumor + vehicle, normal thyroid + MIA-602, and normal thyroid + vehicle. Tumor or normal thyroid cells (each 50,000 cells per well) were seeded in 24-well plates with 1 ml medium containing 10 % fetal bovine serum (FBS). After 24 h, culture media were replaced by DMEM/F:12 with 10% charcoal stripped media. 72 h later, cells were treated with 1 % charcoal stripped serum media containing MIA-602 (1 μM concentration) or vehicle only. The cells were then incubated for 24 h at 37 °C in 10 % CO2. An absolute cell count was performed using a Z1 Coulter particle counter (Beckman Coulter, Brea, CA). Duplicate counts of tumor and normal thyroid cells with and without MIA-602 were performed on trypsinized wells on days 1, 3 and 5.
Western Blot Analysis
Western blot analyses were performed for four experimental groups: thyroid tumor + MIA-602, thyroid tumor + vehicle, normal thyroid + MIA-602, and normal thyroid + vehicle. Tumor or normal thyroid cells (each 600,000 cells per well) were plated in each 75 cm2 flask (Nalge Nunc International, Rochester, NY) in DMEM/F:12 media with 10 % FBS. When the cells reached confluency, the media were switched to phenol red-free medium with 10 % charcoal-stripped serum (EFS) for 18 h. Cells were then exposed to MIA-602 (1 μM concentration) or vehicle only for 16–18 h. The supernatant was collected for zymography, and the cells were lysed for protein analysis. Protein expression was examined by Western blot as previously described [12]. Briefly, lysates were extracted and an assay was performed using the BCA protein assay kit (Pierce Biotechnology, Rockford, IL). Proteins (10 μg) were loaded in precast SDS polyacrylamide gels (Invitrogen, Carlsbad, CA) after boiling samples with Laemmli buffer and β-mercaptoethanol. Following electrophoresis, proteins were transferred to nitrocellulose membranes and blots exposed to GHRH receptor antibody (AbCam, Cambridge, MA; catalog no. 28692; 1:1,000) overnight. The primary antibody was washed and the secondary antibody (goat anti-rabbit; 1:1,000) was added for 1 h, followed by chemiluminescence solution and exposure to autoradiograph film. Films were scanned for densitometric analysis using Image J version 1.6 (National Institutes of Health [NIH], Bethesda, MD) as previously described [13]. Briefly, the number of pixels in each band was measured using Image J and was used for subsequent calculations. SV1, the main splice variant for pGHRH-R, which has a structure resembling a truncated variant of pGHRH-R, was analyzed in a similar fashion [14]. The bands for unglycosylated pGHRH-R (47 kDa) and SV1 (39.5 kDa) were sufficiently separated to allow for individual measurements [15, 16]. The glycosylation status of pGHRH-R is tissue-dependent and varies when analyzing different types of cancer histology. To achieve uniformity in our analyses, we refer to the unglycosylated variant of pGHRH-R in all of our experiments. Each Western blot experiment was carried out on at least four different cell lines (tumor and corresponding normal thyroid cells).
Zymography
Tumor and normal thyroid cells were grown in DMEM/F:12 media with 10 % FBS. Prior to collection, the medium was replaced with DMEM/F:12 with 0.1 % EFS and the cells were treated with MIA-602 (1 μM concentration) or vehicle only for 24 h. Cell supernatants were collected and loaded according to protein concentration as previously described for other cells [12]. Briefly, MMP-2 activity was assessed with 10 % zymogram gels (Invitrogen). After electrophoresis, gels were washed for 1 h in 2.5 % Triton X-100 and were then incubated for 24 h in incubation buffer (50 mM Tris–HCl, 5 mM CaCl2, 1 μM ZnCl2, and 0.05 % NaN3), allowing the determination of total proteolytic MMP-2 activity without interference from its associated tissue inhibitors. Gels were stained with Coomassie Blue and air dried. Densitometry, using Image J (NIH), was used to analyze relative MMP-2 activity.
Statistical Analysis
Significance of differences between experimental groups was determined by analysis of variance in combination with Student’s t test as appropriate. Significance was determined at p < 0.05.
Results
Expression of pGHRH-R and its Splice Variant, SV1, on Human Papillary Thyroid Cancer and Normal Thyroid Cells
Vehicle-treated tumor and normal thyroid cells were subjected to Western blot analysis to determine the expression of pGHRH-R and its main splice variant, SV1. Both pGHRH-R and SV1 could be detected using the same primary antibody. The expression of protein for unglycosylated pGHRH-R (47 kDa) and SV1 (39.5 kDa) on vehicle-treated tumor and normal thyroid cells was confirmed on Western blot (Fig. 1). The expression of pGHRH-R was higher in vehicle-treated tumor cells compared to vehicle-treated normal thyroid cells, *p < 0.05. In contrast, the expression of SV1 was similar in vehicle-treated tumor cells and vehicle-treated normal thyroid cells.
Primary thyroid tumor and normal thyroid cells were analyzed for the expression of pituitary-type growth hormone-releasing hormone receptor (pGHRH-R) and its main splice variant, SV1. a Representative scans of Western blots showing expression of pGHRH-R (47 kDa), SV1 (39.5 kDa), and β-actin. b Data are presented as mean ± SEM of percent of pGHRH-R, SV-1, and β-actin in tumor cells compared to corresponding normal thyroid cells. Tumor cells demonstrated higher expression of pGHRH-R vs. normal thyroid cells; *p < 0.05, n = 10. SV1 expression was not different between normal and tumor cells
Effect of GHRH Antagonist, MIA-602, on the Proliferation of Human Papillary Thyroid Cancer and Normal Thyroid Cells
In vitro assays of MIA-602 exposure (at 1 μM concentration) or exposure to vehicle only were performed on tumor and normal thyroid cells and conducted over a 6-day period. These experiments revealed that tumor cells proliferated at a higher rate than normal thyroid cells when exposed to vehicle only, ** p < 0.01. Exposure to MIA-602, however, did not alter the proliferation rate of either tumor cells or normal thyroid cells. Even after exposure of tumor and normal thyroid cells to MIA-602, the higher comparative proliferation rate of the tumor cells was maintained (Fig. 2).
Cell proliferation was followed for normal (black) and primary thyroid tumor (blue) cells during incubation with growth hormone-releasing hormone (GHRH) antagonist, MIA-602, at 1 μM concentration or with vehicle only. Data points represent absolute cell counts per milliliter on specified days after plating. Symbols:  normal thyroid + vehicle,
normal thyroid + vehicle,  normal thyroid + MIA-602,
normal thyroid + MIA-602,  thyroid tumor + vehicle,
thyroid tumor + vehicle,  thyroid tumor + MIA-602. Tumor cells proliferated at a higher rate compared to normal thyroid cells when exposed to vehicle only; **p < 0.01, n = 6. Treatment with MIA-602, however, did not alter the proliferation rate of either tumor cells or normal thyroid cells. After exposure of tumor and normal thyroid cells with MIA-602, tumor cells maintained a higher proliferation rate
thyroid tumor + MIA-602. Tumor cells proliferated at a higher rate compared to normal thyroid cells when exposed to vehicle only; **p < 0.01, n = 6. Treatment with MIA-602, however, did not alter the proliferation rate of either tumor cells or normal thyroid cells. After exposure of tumor and normal thyroid cells with MIA-602, tumor cells maintained a higher proliferation rate
Effect of MIA-602 on the Protein Expression of pGHRH-R and SV1 of Human Papillary Thyroid Cancer and Normal Thyroid Cells
Tumor and normal thyroid cells were subjected to Western blot analysis to evaluate protein expression of pGHRH-R and its splice variant, SV1. Tumor cells exposed to MIA-602 demonstrated a greater expression of pGHRH-R (47 kDa; Fig. 3a) and SV1 (39.5 kDa; Fig. 3b), as compared to tumor cells exposed to vehicle only, both *p < 0.05. Both pGHRH-R and SV1 could be detected using the same primary antibody. In contrast, MIA-602 did not alter the expression of pGHRH-R or SV1 in normal thyroid cells (Fig. 3a, b).
Western blot analysis of expression of a pituitary-type growth hormone-releasing hormone receptor (pGHRH-R) and b its main splice variant (SV1) in normal (black) and primary thyroid tumor (blue) cells after exposure to GHRH antagonist, MIA-602, at 1 μM concentration or vehicle only. Data are plotted as mean ± SEM of pGHRH-R and SV1 expression of tumor cells and normal thyroid cells compared to corresponding vehicle only controls. Tumor cells demonstrated higher pGHRH-R and SV1 expression after exposure to MIA-602 at 1 μM concentration compared to corresponding vehicle only tumor cells; *p < 0.05, n = 7 (pGHRH-R) and n = 5 (SV1). Normal thyroid cell pGHRH-R and SV1 expression were not affected by exposure to MIA-602. Abbreviations: M molecular weight marker, N+V normal thyroid + vehicle, N+MIA-602 normal thyroid+MIA-602, T+V thyroid tumor + vehicle, T+MIA-602 thyroid tumor + MIA-602
Effect of MIA-602 on the Invasion Potential of Human Papillary Thyroid Cancer and Normal Thyroid Cells
Tumor and normal thyroid cells were subjected to zymographic analysis to evaluate MMP-2 activity, a marker for invasion potential. MMP-2 activity did not differ between tumor and normal thyroid cells when exposed to vehicle only (data not shown). Tumor cells exposed to MIA-602 demonstrated decreased MMP-2 activity compared to tumor cells exposed to vehicle only, ** p < 0.01. Exposure to MIA-602 did not affect the MMP-2 activity of normal thyroid cells (Fig. 4).
Zymographic analysis was performed to examine matrix metalloproteinase (MMP)-2 activity in normal (black) and primary thyroid tumor (blue) cells, after exposure to GHRH antagonist, MIA-602, at 1 μM concentration or vehicle only. Data are graphed as mean ± SEM of MMP-2 activity of tumor and normal thyroid cells compared to corresponding vehicle only controls. Tumor cells demonstrated lower MMP-2 activity after exposure to MIA-602 at 1 μM concentration compared to corresponding vehicle only tumor cells; ** p < 0.01, n = 8. Normal thyroid cell line MMP-2 activity was unaffected by exposure to MIA-602. Abbreviations: M molecular weight marker, N+V normal thyroid + vehicle, N+MIA-602 normal thyroid + MIA-602, T+V thyroid tumor + vehicle, T+MIA-602 thyroid tumor + MIA-602
Discussion
The cancer-related mortality in patients with PTC without metastasis evident at diagnosis is approximately 6 % [17]. Given the rising incidence of thyroid cancer and its aggressive variants, however, investigations focusing on early detection and alternative treatment options have become an important goal of recent thyroid cancer research [18, 19]. Due to the limited effectiveness and poor tolerability of systemic chemotherapy, the current trend in surgical oncology is shifting away from it and toward therapy targeted at the molecular level. Clinical trials are currently underway with drugs targeted for the signaling kinases implicated in oncogenesis [4]. This approach shows considerable promise in treating aggressive or advanced thyroid carcinoma.
To achieve homogeneity in our study population, we chose to evaluate the effect of targeted therapy in papillary thyroid cancer specimens derived from post-menopausal women. The gender disparity with regard to invasive potential and outcomes has been well documented, and papillary thyroid cancer has been demonstrated to occur at rates 2.9 to 3.8 times greater in females vs. males [20]. More specifically, studies have found that estrogen receptor plays an important role in advanced papillary thyroid cancer [21]. By limiting our study cohort to post-menopausal women with papillary thyroid cancer, we are framing our experiments within homogeneous pathologic features and thereby refining our indication for targeted therapy.
Antagonists of GHRH have been shown to augment apoptosis and decrease proliferation of multiple cancer types, including lung, breast, and prostate [9, 22–25]. Increasing evidence suggests that in addition to inhibiting the pituitary-GH-hepatic IGF-I axis, the effect of GHRH antagonists is related to a direct autocrine/paracrine inhibition of tumor growth [26]. Essentially, the effect of the GHRH antagonist is via inhibition of GHRH within tumor tissue [9]. However, the role of GHRH antagonists in thyroid cancer has not been heretofore investigated. In an effort to identify potentially novel treatment mechanisms, we show for the first time that pGHRH-R and its main splice variant, SV1, are expressed in primary PTC cells. The effect of GHRH antagonists on decreasing the invasion potential of tumor cells may be a promising therapeutic pathway, though its effect on proliferation in vitro was not statistically significant. The findings on upregulation of pGHRH-R in papillary thyroid cancer cells by exposure to GHRH antagonist, MIA-602, are in agreement with the results reported for human PC-3 prostate cancers [27] and DBTRG-06 human glioblastomas [16] treated with other GHRH antagonists.
Recent data suggest that MMPs not only degrade extracellular matrices, but also stimulate secretion of growth factors that increase mitogenic activity and inhibit apoptotic pathways [28]. Specifically, MMPs have been shown to be involved in the invasion potential of thyroid cancer [6–8]. Rajoria et al. showed that a natural dietary compound with anti-estrogenic activity, 3,3′-diindolylmethane, inhibits the metastatic potential of thyroid cancer by eliminating MMP-2 and MMP-9 expression in vitro [6]. In addition, Campo et al. demonstrated an increase in MMP-2 in all histologic types of primary thyroid carcinomas and their metastases [7]. The same study found increased MMP-2 expression in metastatic tissue compared to primary tumor cells. This finding suggests upregulation of enzymes in metastastic cells, which correlates with higher tumor grade. In our study of slow-growing primary thyroid cancer cells, it was not surprising that we did not find a difference in MMP-2 activity when compared to normal thyroid tissue, when exposed to vehicle only. In this setting, we have demonstrated a clear reduction of MMP-2 activity in tumor cells compared to normal thyroid cells consequent to exposure to the GHRH antagonist, MIA-602.
Conclusion
In this study, we found that pGHRH-R and its main splice variant are expressed in thyroid tissue. The expression of pGHRH-R was upregulated in tumor cells compared to normal thyroid cells, while SV1 expression was not different. These differences in tumor cell receptor expression may affect sensitivity to GHRH antagonists, as shown by the marked changes observed in expression of pGHRH-R and SV1 after drug treatment in tumor cells, while concurrently treated normal thyroid cells remained unaltered. The decreased MMP-2 activity found in tumor cells treated with GHRH antagonists suggests a reduction in the invasion potential of PTC cells; meanwhile, normal thyroid cell MMP-2 activity was unaffected. The use of potent GHRH antagonists may have a significant therapeutic potential in patients with aggressive variants of thyroid cancer that are refractory to current treatment modalities. Further, that normal thyroid cells were unaffected by such treatment with antagonists of GHRH, suggests a selective advantage. Additional research is warranted to further delineate the effect of treatment with antagonists of GHRH on tumor cells, given its likelihood to influence future clinical outcomes for patients with PTC.
References
Davies L, Gilbert Welch H (2006) Increasing incidence of thyroid cancer in the United States, 1973–2002. JAMA 295(18):2164–2167. doi:10.1001/jama.295.18.2164
Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, Mandel SJ, Mazzaferri EL, McIver B, Pacini F, Schlumberger M, Sherman SI, Steward DL, Michael Tuttle R (2009) Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 19(11):1167–1214. doi:10.1089/thy.2009.0110
Sipos JA, Mazzaferri EL (2008) The therapeutic management of differentiated thyroid cancer. Expert Opin Pharmacother 9(15):2627–2637. doi:10.1517/14656566.9.15.2627
Sherman SI (2011) Targeted therapies for thyroid tumors. Mod Pathol 24(Suppl 2):S44–S52. doi:10.1038/modpathol.2010.165
Dadu R, Shah K, Busaidy NL, Waguespack SG, Habra MA, Ying AK, Hu MI, Bassett R, Jimenez C, Sherman SI, Cabanillas ME (2015) Efficacy and tolerability of vemurafenib in patients with BRAF(V600E)-positive papillary thyroid cancer: M.D. Anderson Cancer Center Off Label Experience. J Clin Endocrinol Metab 100(1):E77–E81. doi:10.1210/jc.2014-2246
Rajoria S, Suriano R, George A, Shanmugam A, Schantz SP, Geliebter J, Tiwari RK (2011) Estrogen induced metastatic modulators MMP-2 and MMP-9 are targets of 3,3′-diindolylmethane in thyroid cancer. PLoS One 6(1):e15879. doi:10.1371/journal.pone.0015879
Campo E, Merino MJ, Liotta L, Neumann R, Stetler-Stevenson W (1992) Distribution of the 72-kd type IV collagenase in nonneoplastic and neoplastic thyroid tissue. Hum Pathol 23(12):1395–1401
Baldini E, Toller M, Graziano FM, Russo FP, Pepe M, Biordi L, Marchioni E, Curcio F, Ulisse S, Ambesi-Impiombato FS, D’Armiento M (2004) Expression of matrix metalloproteinases and their specific inhibitors in normal and different human thyroid tumor cell lines. Thyroid 14(11):881–888. doi:10.1089/thy.2004.14.881
Schally AV, Varga JL, Engel JB (2008) Antagonists of growth-hormone-releasing hormone: an emerging new therapy for cancer. Nat Clin Pract Endocrinol Metab 4(1):33–43. doi:10.1038/ncpendmet0677
Siejka A, Barabutis N, Schally AV (2012) GHRH antagonist inhibits focal adhesion kinase (FAK) and decreases expression of vascular endothelial growth factor (VEGF) in human lung cancer cells in vitro. Peptides 37(1):63–68. doi:10.1016/j.peptides.2012.07.010
Varga JL, Schally AV, Horvath JE, Kovacs M, Halmos G, Groot K, Toller GL, Rekasi Z, Zarandi M (2004) Increased activity of antagonists of growth hormone-releasing hormone substituted at positions 8, 9, and 10. Proc Natl Acad Sci USA 101(6):1708–1713. doi:10.1073/pnas.0307288101
Glassberg MK, Elliot SJ, Fritz J, Catanuto P, Potier M, Donahue R, Stetler-Stevenson W, Karl M (2008) Activation of the estrogen receptor contributes to the progression of pulmonary lymphangioleiomyomatosis via matrix metalloproteinase-induced cell invasiveness. J Clin Endocrinol Metab 93(5):1625–1633. doi:10.1210/jc.2007-1283
Catanuto P, Doublier S, Lupia E, Fornoni A, Berho M, Karl M, Striker GE, Xia X, Elliot S (2009) 17 beta-estradiol and tamoxifen upregulate estrogen receptor beta expression and control podocyte signaling pathways in a model of type 2 diabetes. Kidney Int 75(11):1194–1201. doi:10.1038/ki.2009.69
Rekasi Z, Czompoly T, Schally AV, Halmos G (2000) Isolation and sequencing of cDNAs for splice variants of growth hormone-releasing hormone receptors from human cancers. Proc Natl Acad Sci USA 97(19):10561–10566. doi:10.1073/pnas.180313297
Gaylinn BD, Lyons CE, Zysk JR, Clarke IJ, Thorner MO (1994) Photoaffinity cross-linking to the pituitary receptor for growth hormone-releasing factor. Endocrinology 135(3):950–955. doi:10.1210/endo.135.3.8070391
Kovacs M, Schally AV, Hohla F, Rick FG, Pozsgai E, Szalontay L, Varga JL, Zarandi M (2010) A correlation of endocrine and anticancer effects of some antagonists of GHRH. Peptides 31(10):1839–1846. doi:10.1016/j.peptides.2010.07.006
Mazzaferri EL, Jhiang SM (1994) Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am J Med 97(5):418–428
Ahmed M, Uddin S, Hussain AR, Alyan A, Jehan Z, Al-Dayel F, Al-Nuaim A, Al-Sobhi S, Amin T, Bavi P, Al-Kuraya KS (2012) FoxM1 and its association with matrix metalloproteinases (MMP) signaling pathway in papillary thyroid carcinoma. J Clin Endocrinol Metab 97(1):E1–E13. doi:10.1210/jc.2011-1506
Loh K-C, Greenspan FS, Gee L, Miller TR, Yeo PP (1997) Pathological tumor-node-metastasis (pTNM) staging for papillary and follicular thyroid carcinomas: a retrospective analysis of 700 patients. J Clin Endocrinol Metab 82(11):3553–3562
Aschebrook-Kilfoy B, Ward MH, Sabra MM, Devesa SS (2011) Thyroid cancer incidence patterns in the United States by histologic type, 1992–2006. Thyroid 21(2):125–134. doi:10.1089/thy.2010.0021
Rahbari R, Zhang L, Kebebew E (2010) Thyroid cancer gender disparity. Future Oncol 6(11):1771–1779. doi:10.2217/fon.10.127
Munoz-Moreno L, Isabel Arenas M, Jose Carmena M, Schally AV, Prieto JC, Bajo AM (2014) Growth hormone-releasing hormone antagonists abolish the transactivation of human epidermal growth factor receptors in advanced prostate cancer models. Invest New Drugs 32(5):871–882. doi:10.1007/s10637-014-0131-4
Siejka A, Barabutis N, Schally AV (2011) GHRH antagonist MZ-5-156 increases the expression of AMPK in A549 lung cancer cells. Cell Cycle 10(21):3714–3718. doi:10.4161/cc.10.21.17904
Seitz S, Rick FG, Schally AV, Treszl A, Hohla F, Szalontay L, Zarandi M, Ortmann O, Engel JB, Buchholz S (2013) Combination of GHRH antagonists and docetaxel shows experimental effectiveness for the treatment of triple-negative breast cancers. Oncol Rep 30(1):413–418. doi:10.3892/or.2013.2435
Fahrenholtz CD, Rick FG, Garcia MI, Zarandi M, Cai R-Z, Block NL, Schally AV, Burnstein KL (2014) Preclinical efficacy of growth hormone-releasing hormone antagonists for androgen-dependent and castration-resistant human prostate cancer. Proc Natl Acad Sci USA 111(3):1084–1089. doi:10.1073/pnas.1323102111
Busto R, Schally AV, Varga JL, Garcia-Fernandez MO, Groot K, Armatis P, Szepeshazi K (2002) The expression of growth hormone-releasing hormone (GHRH) and splice variants of its receptor in human gastroenteropancreatic carcinomas. Proc Natl Acad Sci USA 99(18):11866–11871. doi:10.1073/pnas.182433099
Heinrich E, Schally AV, Buchholz S, Rick FG, Halmos G, Mile M, Groot K, Hohla F, Zarandi M, Varga JL (2008) Dose-dependent growth inhibition in vivo of PC-3 prostate cancer with a reduction in tumoral growth factors after therapy with GHRH antagonist MZ-J-7-138. Prostate 68(16):1763–1772. doi:10.1002/pros.20843
Stamenkovic I (2000) Matrix metalloproteinases in tumor invasion and metastasis. Semin Cancer Biol 10(6):415–433. doi:10.1006/scbi.2000.0379
Acknowledgments
In memory of Dr. Michael Karl, who was instrumental in the initiation of this study. A.V.S. was supported, in part, by the Research Service of the Veterans Affairs Department, Sylvester Comprehensive Cancer Center, and the Division of Hematology and Oncology, Department of Medicine, University of Miami Miller School of Medicine. N.L.B. was supported by the Weeks Family Endowment for Research.
Disclosure Statement
The authors have nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Paola Catanuto and Jun Tashiro contributed equally to this work.
Rights and permissions
About this article
Cite this article
Catanuto, P., Tashiro, J., Rick, F.G. et al. Expression of Receptors for Pituitary-Type Growth Hormone-Releasing Hormone (pGHRH-R) in Human Papillary Thyroid Cancer Cells: Effects of GHRH Antagonists on Matrix Metalloproteinase-2. HORM CANC 6, 100–106 (2015). https://doi.org/10.1007/s12672-015-0217-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12672-015-0217-2