Abstract
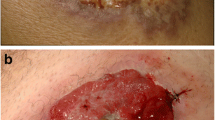
Inflammatory bowel disease (IBD) is associated with a number of extraintestinal complications, including skin lesions. Most reports have shown that skin lesions are found in 10–15% of IBD cases, although this depends on the definition of skin lesions. The representative skin lesions in patients with IBD are erythema nodosum, pyoderma gangrenosum, Sweet’s syndrome, and so on. These lesions are often associated with IBD progression, and intestinal lesions in particular require appropriate treatment. Recently, another clinical issue regarding skin lesions in patients with IBD, a so-called paradoxical reaction, during the treatment with anti-tumor necrosis factor (TNF)-α agents has emerged. These reactions are termed paradoxical reactions because the skin lesions sometimes resemble psoriasis, although the anti-TNF-α agents have been historically used to treat psoriasis. Paradoxical reactions are reportedly found in approximately 5–10% of patients using anti-TNF-α agents and are no longer rare. Now that the use of biologics is at its culmination, reports regarding paradoxical reactions are predicted to increase in number; thus, we must recognize skin lesions with IBD patients including this type of adverse events and manage them appropriately while consulting with dermatologists.




Similar content being viewed by others
References
Molodecky NA, Soon IS, Rabi DM, et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology. 2012;142:46–54.
Ottaviano G, Salvatore S, Salvatoni A, et al. Ocular manifestations of paediatric inflammatory bowel disease: a systematic review and meta-analysis. J Crohns Colitis. 2018;12:870–9.
Fine S, Nee J, Thakuria P, et al. Ocular, auricular, and oral manifestations of inflammatory bowel disease. Dig Dis Sci. 2017;62:3269–79.
Troncoso LL, Biancardi AL, de Moraes HV Jr, et al. Ophthalmic manifestations in patients with inflammatory bowel disease: a review. World J Gastroenterol. 2017;23:5836–48.
Lee HJ, Song HJ, Jeong JH, et al. Ophthalmologic manifestations in patients with inflammatory bowel disease. Intest Res. 2017;15:380–7.
Hammoudeh M, Elsayed E, Al-Kaabi S, et al. Rheumatic manifestations of inflammatory bowel diseases: a study from the Middle East. J Int Med Res. 2018;46:3837–47.
Pouillon L, Bossuyt P, Vanderstukken J, et al. Management of patients with inflammatory bowel disease and spondyloarthritis. Expert Rev Clin Pharmacol. 2017;10:1363–74.
Vavricka SR, Brun L, Ballabeni P, et al. Frequency and risk factors for extraintestinal manifestations in the Swiss inflammatory bowel disease cohort. Am J Gastroenterol. 2011;106:110–9.
Vavricka SR, Schoepfer A, Scharl M, et al. Extraintestinal manifestations of inflammatory bowel disease. Inflamm Bowel Dis. 2015;21:1982–92.
Monsén U, Sorstad J, Hellers G, et al. Extracolonic diagnoses in ulcerative colitis: an epidemiological study. Am J Gastroenterol. 1990;85:711–6.
Lo B, Julsgaard M, Vester-Andersen MK, et al. Disease activity, steroid use and extraintestinal manifestation are associated with increased disability in patients with inflammatory bowel disease using the inflammatory bowel disease disability index: a cross-sectional multicentre cohort study. Eur J Gastroenterol Hepatol. 2018;30:1130–6.
Godat S, Fournier N, Safroneeva E, et al. Frequency and type of drug-related side effects necessitating treatment discontinuation in the Swiss Inflammatory Bowel Disease Cohort. Eur J Gastroenterol Hepatol. 2018;30:612–20.
Cleynen I, Vermeire S. Paradoxical inflammation induced by anti-TNF agents in patients with IBD. Nat Rev Gastroenterol Hepatol. 2012;9:496–503.
Lichtenstein L, Ron Y, Kivity S, et al. Infliximab-related infusion reactions: systematic review. J Crohns Colitis. 2015;9:806–15.
Duron C, Goutte M, Pereira B, et al. Factors influencing acute infusion reactions in inflammatory bowel disease patients treated with infliximab in the era of scheduled maintenance therapy. Eur J Gastroenterol Hepatol. 2015;27:705–11.
Keane J, Gershon S, Wise RP, et al. Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent. N Engl J Med. 2001;345:1098–104.
Park DI, Hisamatsu T, Chen M, et al. Asian Organization for Crohn’s and Colitis and Asian Pacific Association of Gastroenterology consensus on tuberculosis infection in patients with inflammatory bowel disease receiving anti-tumor necrosis factor treatment. Part 1: risk assessment. J Gastroenterol Hepatol. 2018;33:20–9.
Greenstein AJ, Janowitz HD, Sachar DB. The extra-intestinal complications of Crohn’s disease and ulcerative colitis: a study of 700 patients. Medicine. 1976;55:401–12.
Rankin GB, Watts HD, Melnyk CS, et al. National Cooperative Crohn’s Disease Study: extraintestinal manifestations and perianal complications. Gastroenterology. 1979;77:914–20.
Veloso FT, Carvalho J, Magro F. Immune-related systemic manifestations of inflammatory bowel disease. A prospective study of 792 patients. J Clin Gastroenterol. 1996;23:29–34.
Jose FA, Garnett EA, Vittinghoff E, et al. Development of extraintestinal manifestations in pediatric patients with inflammatory bowel disease. Inflamm Bowel Dis. 2009;15:63–8.
Ott C, Schölmerich J. Extraintestinal manifestations and complications in IBD. Nat Rev Gastroenterol Hepatol. 2013;10:585–95.
Marzano AV, Borghi A, Stadnicki A, et al. Cutaneous manifestations in patients with inflammatory bowel diseases: pathophysiology, clinical features, and therapy. Inflamm Bowel Dis. 2014;20:213–27.
Alreheili KM, Alsaleem KA, Almehaidib AI. Natural history and outcome of inflammatory bowel diseases in children in Saudi Arabia: a single-center experience. Saudi J Gastroenterol. 2018;24:171–6.
Burgdorf W. Cutaneous manifestations of Crohn’s disease. J Am Acad Dermatol. 1981;5:689–95.
Palamaras I, El-Jabbour J, Pietropaolo N, et al. Metastatic Crohn’s disease: a review. J Eur Acad Dermatol Venereol. 2008;22:1033–43.
Freeman HJ. Erythema nodosum and pyoderma gangrenosum in 50 patients with Crohn’s disease. Can J Gastroenterol. 2005;19:603–6.
Farhi D, Cosnes J, Zizi N, et al. Significance of erythema nodosum and pyoderma gangrenosum in inflammatory bowel diseases: a cohort study of 2402 patients. Medicine. 2008;87:281–93.
Saltman AP, Kuriya B. Löfgren syndrome in acute sarcoidosis. CMAJ. 2017;189:E1230.
Davatchi F, Chams-Davatchi C, Shams H, et al. Adult Behcet’s disease in Iran: analysis of 6075 patients. Int J Rheum Dis. 2016;19:95–103.
Passarini B, Infusino SD. Erythema nodosum. G Ital Dermatol Venereol. 2013;148:413–7.
Apgar JT. Newer aspects of inflammatory bowel disease and its cutaneous manifestations: a selective review. Semin Dermatol. 1991;10:138–47.
Timani S, Mutasim DF. Skin manifestations of inflammatory bowel disease. Clin Dermatol. 2008;26:265–73.
Lakatos PL, Lakatos L, Kiss LS, et al. Treatment of extraintestinal manifestations in inflammatory bowel disease. Digestion. 2012;86:28–35.
Clayton TH, Walker BP, Stables GI. Treatment of chronic erythema nodosum with infliximab. Clin Exp Dermatol. 2006;31:823–4.
Quin A, Kane S, Ulitsky O. A case of fistulizing Crohn’s disease and erythema nodosum managed with adalimumab. Nat Clin Pract Gastroenterol Hepatol. 2008;5:278–81.
Vavricka SR, Gubler M, Gantenbein C, et al. Anti-TNF treatment for extraintestinal manifestations of inflammatory bowel disease in the Swiss IBD Cohort Study. Inflamm Bowel Dis. 2017;23:1174–81.
Greuter T, Navarini A, Vavricka SR. Skin manifestations of inflammatory bowel disease. Clin Rev Allergy Immunol. 2017;53:413–27.
Polcz M, Gu J, Florin T. Pyoderma gangrenosum in inflammatory bowel disease: the experience at Mater Health Services’ Adult Hospital 1998–2009. J Crohns Colitis. 2011;5:148–51.
Bennett ML, Jackson JM, Jorizzo JL, et al. Pyoderma gangrenosum. A comparison of typical and atypical forms with an emphasis on time to remission. Case review of 86 patients from 2 institutions. Medicine (Baltimore). 2000;79:37–46.
Powell FC, Su WP, Perry HO. Pyoderma gangrenosum: classification and management. J Am Acad Dermatol. 1996;34:395–409.
Lora V, Cerroni L, Cota C. Skin manifestations of rheumatoid arthritis. G Ital Dermatol Venereol. 2018;153:243–55.
Chasset F, Francès C. Cutaneous manifestations of medium- and large-vessel vasculitis. Clin Rev Allergy Immunol. 2017;53:452–68.
Ahn C, Negus D, Huang W. Pyoderma gangrenosum: a review of pathogenesis and treatment. Expert Rev Clin Immunol. 2018;14:225–33.
Callen JP. Pyoderma gangrenosum. Lancet. 1998;351:581–5.
Rothfuss KS, Stange EF, Herrlinger KR. Extraintestinal manifestations and complications in inflammatory bowel diseases. World J Gastroenterol. 2006;12:4819–31.
Huang W, McNeely MC. Neutrophilic tissue reactions. Adv Dermatol. 1997;13:33–64.
Weizman A, Huang B, Berel D, et al. Clinical, serologic, and genetic factors associated with pyoderma gangrenosum and erythema nodosum in inflammatory bowel disease patients. Inflamm Bowel Dis. 2014;20:525–33.
Thomas KS, Ormerod AD, Craig FE, et al. UK Dermatology Clinical Trials Network’s STOP GAP Team. Clinical outcomes and response of patients applying topical therapy for pyoderma gangrenosum: a prospective cohort study. J Am Acad Dermatol. 2016;75:940–9.
Rice SA, Woo PN, El-Omar E, et al. Topical tacrolimus 0.1% ointment for treatment of cutaneous Crohn’s disease. BMC Res Notes. 2013;6:19.
Jolles S, Niclasse S, Benson E. Combination oral and topical tacrolimus in therapy-resistant pyoderma gangrenosum. Br J Dermatol. 1999;140:564–5.
Friedman S, Marion JF, Scherl E, et al. Intravenous cyclosporine in refractory pyoderma gangrenosum complicating inflammatory bowel disease. Inflamm Bowel Dis. 2001;7:1–7.
Carp JM, Onuma E, Das K, et al. Intravenous cyclosporine therapy in the treatment of pyoderma gangrenosum secondary to Crohn’s disease. Cutis. 1997;60:135–8.
Sapienza MS, Cohen S, Dimarino AJ. Treatment of pyoderma gangrenosum with infliximab in Crohn’s disease. Dig Dis Sci. 2004;49:1454–7.
Brooklyn TN, Dunnill MG, Shetty A, et al. Infliximab for the treatment of pyoderma gangrenosum: a randomised, double blind, placebo controlled trial. Gut. 2006;55:505–9.
Vavricka SR, Scharl M, Gubler M, et al. Biologics for extraintestinal manifestations of IBD. Curr Drug Targets. 2014;15:1064–73.
Hurabielle C, Schneider P, Baudry C, et al. Certolizumab pegol - A new therapeutic option for refractory disseminated pyoderma gangrenosum associated with Crohn’s disease. J Dermatolog Treat. 2016;27:67–9.
Afifi L, Sanchez IM, Wallace MM, et al. Diagnosis and management of peristomal pyoderma gangrenosum: a systematic review. J Am Acad Dermatol. 2018;78:1195–204.e1.
Ohmori T, Yamagiwa A, Nakamura I, et al. Treatment of pyoderma gangrenosum associated with Crohn’s disease. Am J Gastroenterol. 2003;98:2101–2.
Ohno M, Koyama S, Ohara M, et al. Pyoderma gangrenosum with ulcerative colitis successfully treated by the combination of granulocyte and monocyte adsorption apheresis and corticosteroids. Intern Med. 2016;55:25–30.
Sweet D. An acute febrile neutrophilic dermatosis. Br J Dermatol. 1964;76:350–6.
Salmon P, Rademaker M, Edwards L. A continuum of neutrophilic disease occurring in a patient with ulcerative colitis. Australas J Dermatol. 1998;39:116–8.
Becuwe C, Delaporte E, Colombel JF, et al. Sweet’s syndrome associated with Crohn’s disease. Acta Derm Venereol. 1989;69:444–5.
Travis S, Innes N, Davies MG, et al. Sweet’s syndrome: an unusual cutaneous feature of Crohn’s disease or ulcerative colitis. Eur J Gastroenterol Hepatol. 1997;9:715–20.
Ali M, Duerksen DR. Ulcerative colitis and Sweet’s syndrome: a case report and review of the literature. Can J Gastroenterol. 2008;22:296–8.
Shin OR, Lee Y, Bak S, et al. Gastroenterology: sweet’s syndrome in a patient with acutely exacerbated ulcerative colitis. J Gastroenterol Hepatol. 2015;30:965.
Lopes CR, Soares M, Cardoso C, et al. Sweet’s syndrome complicating ulcerative colitis: a rare association. BMJ Case Rep. 2016;2016:pii: bcr2015212990.
Calixto R, Menezes Y, Ostronoff M, et al. Favorable outcome of severe, extensive, granulocyte colony-stimulating factor-induced, corticosteroid-resistant Sweet’s syndrome treated with high-dose intravenous immunoglobulin. J Clin Oncol. 2014;32:e1–2.
Fukutoku M, Shimizu S, Ogawa Y, et al. Sweet’s syndrome during therapy with granulocyte colony-stimulating factor in a patient with aplastic anaemia. Br J Haematol. 1994;86:645–8.
Fujii A, Mizutani Y, Hattori Y, et al. Sweet’s syndrome successfully treated with granulocyte and monocyte adsorption apheresis. Case Rep Dermatol. 2017;9:13–8.
Yasuda F, Fujio Y, Kakuta R, et al. Use of cyclosporin A for successful management of steroid-resistant Sweet’s syndrome patient with possible myelodysplastic syndrome. J Dermatol. 2014;41:465–6.
Martínez Andrés B, Sastre Lozano V, Sánchez Melgarejo JF. Sweet syndrome after treatment with vedolizumab in a patient with Crohn’s disease. Rev Esp Enferm Dig. 2018;110:530.
Maeda K, Okada M, Yao T, et al. Intestinal and extraintestinal complications of Crohn’s disease: predictors and cumulative probability of complications. J Gastroenterol. 1994;29:577–82.
Letsinger JA, McCarty MA, Jorizzo JL. Complex aphthosis: a large case series with evaluation algorithm and therapeutic ladder from topicals to thalidomide. J Am Acad Dermatol. 2005;52:500–8.
Basu MK, Asquith P. Oral manifestations of inflammatory bowel disease. Clin Gastroenterol. 1980;9:307–21.
Kurtzman DJ, Jones T, Lian F, et al. Metastatic Crohn’s disease: a review and approach to therapy. J Am Acad Dermatol. 2014;71:804–13.
Guest GD, Fink RL. Metastatic Crohn’s disease: case report of an unusual variant and review of the literature. Dis Colon Rectum. 2000;43:1764–6.
Marotta PJ, Reynolds RP. Metastatic Crohn’s disease. Am J Gastroenterol. 1996;9:373–5.
Sabbadini C, Banzato C, Schena D, et al. Metastatic Crohn’s disease in childhood. J Dtsch Dermatol Ges. 2016;14:431–4.
Siroy A, Wasman J. Metastatic Crohn disease: a rare cutaneous entity. Arch Pathol Lab Med. 2012;136:329–32.
Chiba M, Iizuka M, Horie Y, et al. Metastatic Crohn’s disease involving the penis. J Gastroenterol. 1997;32:817–21.
Albuquerque A, Magro F, Rodrigues S, et al. Metastatic cutaneous Crohn’s disease of the face: a case report and review of the literature. Eur J Gastroenterol Hepatol. 2011;23:954–6.
Carranza DC, Young L. Successful treatment of metastatic Crohn’s disease with cyclosporine. J Drugs Dermatol. 2008;7:789–91.
Kiuru M, Camp B, Adhami K, et al. Treatment of metastatic cutaneous Crohn disease with certolizumab. Dermatol Online J. 2015;21.
Wylomanski S, Bouquin R, Dréno B, et al. Spectacular response of metastatic vulval Crohn’s disease to infliximab treatment. Int J Dermatol. 2016;55:1146–8.
Williams N, Scott NA, Watson JS, et al. Surgical management of perineal and metastatic cutaneous Crohn’s disease. Br J Surg. 1993;80:1596–8.
Zouboulis CC, Desai N, Emtestam L, et al. European S1 guideline for the treatment of hidradenitis suppurativa/acne inversa. J Eur Acad Dermatol Venereol. 2015;29:619–44.
Zouboulis CC, Del Marmol V, Mrowietz U, et al. Hidradenitis suppurativa/acne inversa: criteria for diagnosis, severity assessment, classification and disease evaluation. Dermatology. 2015;231:184–90.
van der Zee HH, de Winter K, van der Woude CJ, et al. The prevalence of hidradenitis suppurativa in 1093 patients with inflammatory bowel disease. Br J Dermatol. 2014;171:673–5.
Yadav S, Singh S, Edakkanambeth Varayil J, et al. Hidradenitis suppurativa in patients with inflammatory bowel disease: a population-based cohort study in Olmsted County, Minnesota. Clin Gastroenterol Hepatol. 2016;14:65–70.
van der Zee HH, van der Woude CJ, Florencia EF, et al. Hidradenitis suppurativa and inflammatory bowel disease: are they associated? Results of a pilot study. Br J Dermatol. 2010;162:195–7.
Deckers IE, Benhadou F, Koldijk MJ, et al. Inflammatory bowel disease is associated with hidradenitis suppurativa: Results from a multicenter cross-sectional study. J Am Acad Dermatol. 2017;76:49–53.
Lukach AJ, Saul MI, Ferris LK, et al. Risk factors for hidradenitis suppurativa in patients with inflammatory bowel disease. Dig Dis Sci. 2018;63:755–60.
Ortiz-Masiá D, Cosín-Roger J, Calatayud S, et al. M1 Macrophages activate notch signalling in epithelial cells: relevance in Crohn’s disease. J Crohns Colitis. 2016;10:582–92.
Schlapbach C, Hänni T, Yawalkar N, et al. Expression of the IL-23/Th17 pathway in lesions of hidradenitis suppurativa. J Am Acad Dermatol. 2011;65:790–8.
Ingram JR. The genetics of hidradenitis suppurativa. Dermatol Clin. 2016;34:23–8.
Alikhan A, Lynch PJ, Eisen DB. Hidradenitis suppurativa: a comprehensive review. J Am Acad Dermatol. 2009;60:539–61 (quiz 562–3).
Knowles SR, Graff LA, Wilding H, et al. Quality of life in inflammatory bowel disease: a systematic review and meta-analyses—part I. Inflamm Bowel Dis. 2018;24:742–51.
Knowles SR, Graff LA, Wilding H, et al. Quality of life in inflammatory bowel disease: a systematic review and meta-analyses—part II. Inflamm Bowel Dis. 2018;24:966–76.
Kimball AB, Kerdel F, Adams D, et al. Adalimumab for the treatment of moderate to severe Hidradenitis suppurativa: a parallel randomized trial. Ann Intern Med. 2012;157:846–55.
Gottlieb A, Menter A, Armstrong A, et al. Adalimumab treatment in women with moderate-to-severe hidradenitis suppurativa from the placebo-controlled portion of a phase 2, randomized, double-blind study. J Drugs Dermatol. 2016;15:1192–6.
Kimball AB, Okun MM, Williams DA, et al. Two phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375:422–34.
Kerbleski JF, Gottlieb AB. Dermatological complications and safety of anti-TNF treatments. Gut. 2009;58:1033–9.
Fréling E, Baumann C, Cunty JF, et al. Cumulative incidence of, risk factors for, and outcome of dermatological complications of anti-TNF therapy in inflammatory bowel disease: a 14-year experience. Am J Gastroenterol. 2015;110:1186–96.
Cleynen I, Moerkercke WV, Billiet T, et al. Characteristics of skin lesions associated with anti-tumor necrosis factor therapy in patients with inflammatory bowel disease. Ann Intern Med. 2016;164:10–22.
Verea MM, Del Pozo J, Yebra-Pimental MT, et al. Psoriasiform eruption induced by infliximab. Ann Pharmacother. 2004;38:54–5.
Sfikakis PP, Ilipoulos A, Elezoglou A, et al. Psoriasis induced by anti-tumor necrosis factor therapy: a paradoxical adverse reaction. Arthritis Rheum. 2005;52:2513–8.
Gottlieb AB, Evans R, Li S, et al. Infliximab induction therapy for patients with severe plaque-type psoriasis: a randomized, double-blind, placebo-controlled trial. J Am Acad Dermatol. 2004;51:534–42.
Asahina A, Nakagawa H, Etoh T, Adalimumab M04-688 Study Group, et al. Adalimumab in Japanese patients with moderate to severe chronic plaque psoriasis: efficacy and safety results from a Phase II/III randomized controlled study. J Dermatol. 2010;37:299–310.
Andrade P, Lopes S, Gaspar R, et al. Anti-tumor necrosis factor-α-induced dermatological complications in a large cohort of inflammatory bowel disease patients. Dig Dis Sci. 2018;63:746–54.
Bae JM, Lee HH, Lee BI, et al. Incidence of psoriasiform diseases secondary to tumour necrosis factor antagonists in patients with inflammatory bowel disease: a nationwide population-based cohort study. Aliment Pharmacol Ther. 2018;48:196–205.
Afzali A, Wheat CL, Hu JK, Olerud JE, Lee SD. The association of psoriasiform rash with anti-tumor necrosis factor (anti-TNF) therapy in inflammatory bowel disease: a single academic center case series. J Crohns Colitis. 2014;8:480–8.
Rahier JF, Buche S, Peyrin-Biroulet L, et al. Groupe d’Etude Thérapeutique des Affections Inflammatoires du Tube Digestif (GETAID). Severe skin lesions cause patients with inflammatory bowel disease to discontinue anti-tumor necrosis factor therapy. Clin Gastroenterol Hepatol. 2010;8:1048–55.
Collamer AN, Battafarano DF. Psoriatic skin lesions induced by tumor necrosis factor antagonist therapy: clinical features and possible immunopathogenesis. Semin Arthritis Rheum. 2010;40:233–40.
Shmidt E, Wetter DA, Ferguson SB, et al. Psoriasis and palmoplantar pustulosis associated with tumor necrosis factor-α inhibitors: the Mayo Clinic experience, 1998 to 2010. J Am Acad Dermatol. 2012;67:e179-85.
Tillack C, Ehmann LM, Friedrich M, et al. Anti-TNF antibody-induced psoriasiform skin lesions in patients with inflammatory bowel disease are characterized by interferon-γ expressing Th1 cells and IL-17A/IL-22-expressing Th17 cells and respond to anti-IL-12/IL-23 antibody treatment. Gut. 2014;63:567–77.
Pugliese D, Guldi L, Ferraro PM, et al. Paradoxical psoriasis in a large cohort of patients with inflammatory bowel disease receiving treatment with anti-TNF alpha: 5-year follow-up study. Aliment Pharmacol Ther. 2015;42:880–8.
Ko JM, Gottlieb AB, Kerbleski JF. Induction and exacerbation of psoriasis with TNF-blockade therapy: a review and analysis of 127 cases. J Dermatolog Treat. 2009;20:100–8.
Numakura T, Tamada T, Nara M, et al. Simultaneous development of sarcoidosis and cutaneous vasculitis in a patient with refractory Crohn’s disease during infliximab therapy. BMC Pulm Med. 2016;16:30.
Gîlcă GE, Diaconescu S, Bălan GG, et al. Sarcoidosis associated with infliximab therapy in ulcerative colitis: a case report. Medicine (Baltimore). 2017;96:e6156.
Decock A, Van Assche G, Vermeire S, et al. Sarcoidosis-like lesions: another paradoxical reaction to anti-TNF therapy? J Crohns Colitis. 2017;11:378–83.
Amano H, Matsuda R, Shibata T, et al. Paradoxical SAPHO syndrome observed during anti-TNFα therapy for Crohn’s disease. Biologics. 2017;11:65–9.
Marzano AV, Tavecchio S, Berti E, et al. Paradoxical autoinflammatory skin reaction to tumor necrosis factor alpha blockers manifesting as amicrobial pustulosis of the folds in patients with inflammatory bowel diseases. Medicine (Baltimore). 2015;94:e1818.
Keohane SG, Hayes PC, Hunter JA. Lichen planus and Crohn’s disease. Acta Derm Venereol. 1997;77:488.
Kumar KM, Nachiammai N, Madhushankari GS. Association of oral manifestations in ulcerative colitis: a pilot study. J Oral Maxillofac Pathol. 2018;22:199–203.
Alice Sy N, Khalidi N, Dehghan, et al. Vasculitis in patients with inflammatory bowel diseases: a study of 32 patients and systematic review of the literature. Semin Arthritis Rheum. 2016;45:475–82.
Humbert S, Guilpain P, Puéchal X, et al. Inflammatory bowel diseases in anti-neutrophil cytoplasmic antibody-associated vasculitides: 11 retrospective cases from the French Vasculitis Study Group. Rheumatology. 2015;54:1970–5.
Reddy H, Shipman AR, Wojnarowska F. Epidermolysis bullosa acquisita and inflammatory bowel disease: a review of the literature. Clin Exp Dermatol. 2013;38:225–9 (quiz 229–30).
Raab B, Fretzin DF, Bronson DM, et al. Epidermolysis bullosa acquisita and inflammatory bowel disease. JAMA. 1983;250:1746–8.
Fernández-Guarino M, Sáez EM, Gijón RC, et al. Linear IGA dermatosis associated with ulcerative colitis. Eur J Dermatol. 2006;16:692–3.
Taniguchi T, Maejima H, Saito N, et al. Case of linear IgA bullous dermatosis-involved ulcerative colitis. Inflamm Bowel Dis. 2009;15:1284–5.
Vedak P, Kroshinsky D, St John J, et al. Genetic basis of TNF-α antagonist associated psoriasis in inflammatory bowel diseases: a genotype-phenotype analysis. Aliment Pharmacol Ther. 2016;43:697–704.
Glimcher LH, Townsend MJ, Sullivan BM, et al. Recent developments in the transcriptional regulation of cytolytic effector cells. Nat Rev Immunol. 2004;4:900–11.
Rácz E, Prens EP. Molecular pathophysiology of psoriasis and molecular targets of antipsoriatic therapy. Expert Rev Mol Med. 2009;11:e38.
Nestle FO, Gilliet M. Defining upstream elements of psoriasis pathogenesis: an emerging role for interferon alpha. J Investig Dermatol. 2005;125:xiv–xv.
Niess JH, Danese S. Anti-TNF and skin inflammation in IBD: a new paradox in gastroenterology? Gut. 2014;63:533–5.
Collamer AN, Guerrero KT, Henning JS, et al. Psoriatic skin lesions induced by tumor necrosis factor antagonist therapy: a literature review and potential mechanisms of action. Arthritis Rheum. 2008;59:996–1001.
Matsumoto S, Mashima H. Efficacy of ustekinumab against infliximab-induced psoriasis and arthritis associated with Crohn’s disease. Biologics. 2018;12:69–73.
Delobeau M, Abdou A, Puzenat E, et al. Observational case series on adalimumab-induced paradoxical hidradenitis suppurativa. J Dermatolog Treat. 2016;27:251–3.
Benzaquen M, Flachaire B, Rouby F, et al. Paradoxical pustular psoriasis induced by ustekinumab in a patient with Crohn’s disease-associated spondyloarthropathy. Rheumatol Int. 2018;38:1297–9.
Lee HY, Woo CH, Haw S. Paradoxical Flare of Psoriasis after Ustekinumab Therapy. Ann Dermatol. 2017;29:794–5.
Fiorino G, Allez M, Malesci A, et al. Review article: anti TNF-alpha induced psoriasis in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2009;29:921–7.
Soh JS, Yun WJ, Kim KJ, et al. Concomitant use of azathioprine/6-mercaptopurine decreases the risk of anti-TNF-induced skin lesions. Inflamm Bowel Dis. 2015;21:832–9.
Funding
This work was partly supported by Health and Labour Sciences Research Grants for research on intractable diseases from the Ministry of Health, Labour and Welfare of Japan (Investigation and Research for intractable Inflammatory Bowel Disease) (to H.N.), and Japan Society for the Promotion of Science (JSPS) Grants-in-Aid for Scientific Research (KAKENHI) Grant Number JP17J02428 (to T.I.) and JP18H02799 (to H.N.). The funders of the study had no role in the study design, data collection, data analysis, data interpretation, or writing of the report.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Human rights statement
All procedures followed have been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Informed consent was obtained from all patients for being included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Iida, T., Hida, T., Matsuura, M. et al. Current clinical issue of skin lesions in patients with inflammatory bowel disease. Clin J Gastroenterol 12, 501–510 (2019). https://doi.org/10.1007/s12328-019-00958-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12328-019-00958-y