Abstract
Introduction
In this study we aimed to investigate the effectiveness and safety profile of pirfenidone for the treatment of idiopathic pulmonary fibrosis (IPF) in a real-life setting.
Methods
Clinical records of patients diagnosed with mild-to-moderate IPF and receiving pirfenidone treatment across three centers in Turkey between January and September 2017 were retrospectively collected. Pulmonary function measurements, including percentage of forced vital capacity (FVC%) and percentage of diffusion capacity (DLCO%) were analyzed in patients who received pirfenidone treatment for at least 6 months. Decline in lung function, defined as an absolute decline of at least 10% in FVC from baseline, or death at 6 months was also analyzed. Safety data were included for all follow-up visits.
Results
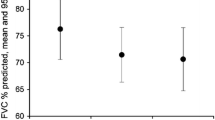
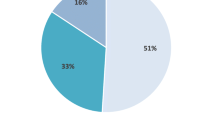
In the pooled cohort (n = 60), patients were mostly men (73.4%) and current or former smokers (61.7%). Average baseline FVC% and DLCO% were 68.4% and 48.7%, respectively. Forty-seven patients (78.3%) had a high-resolution computed tomography scan with a definite interstitial pneumonia (UIP) pattern, and 18 patients (30%) had a surgically proven UIP pattern. Forty-six (76.7%) patients with IPF remained stable and 14 (23.3%) patients had progressed according to decline in FVC of at least 10% during the therapy course. After 6 months of therapy, cough decreased in 58.3% of patients. At least one side effect due to therapy was encountered in 33 (55.0%) IPF patients. Dyspepsia (36.4%), nausea (27.3%), and rash/photosensitivity (24.2%) were the most frequent side effects in our cohort. Sixteen patients (26.7%) needed dose adjustment, one patient (1.7%) discontinued therapy, and one patient (1.7%) died in the study period.
Conclusions
This study shows that pirfenidone seems to be an effective treatment for IPF and also had tolerable and relatively acceptable side effects.
Funding
Roche.
Similar content being viewed by others
References
Raghu G. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med. 2011;183:788–824.
Raghu G. An official ATS/ERS/JRS/ALAT clinical practice guideline: treatment of idiopathic pulmonary fibrosis an update of the 2011 clinical practice guideline. Am J Respir Crit Care Med. 2015;192:e3–e19.
Azuma A, Nukiwa T, Tsuboi E, et al. Double-blind, placebo-controlled trial of pirfenidone in patients with idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2005;171:1040–7.
Taniguchi H, Ebina M, Kondoh Y, et al. Pirfenidone in idiopathic pulmonary fibrosis. Eur Respir J. 2010;35:821–9.
Noble PW, Albera C, Bradford WZ, et al. Pirfenidone in patients with idiopathic pulmonary fibrosis (CAPACITY): two randomised trials. Lancet. 2011;377:1760–9.
King TE Jr, Bradford WZ, Castro-Bernardini S, et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med. 2014;370:2083–92.
Lancaster L, Albera C, Bradford WZ, et al. Safety of pirfenidone in patients with idiopathic pulmonary fibrosis: integrated analysis of cumulative data from 5 clinical trials. BMJ Open Respir Res. 2016;3:e000105.
Costabel U, Albera C, Lancaster LH, et al. An open-label study of the long term safety of pirfenidone in patients with idiopathic pulmonary fibrosis (RECAP). Respiration. 2017;94:408–15.
Nathan SD, Albera C, Bradford WZ. Effect of continued treatment with pirfenidone following clinically meaningful declines in forced vital capacity: analysis of data from three phase 3 trials in patients with idiopathic pulmonary fibrosis. Thorax. 2016;71:429–35.
Ley B, Swigris J, Day BM. Pirfenidone reduces respiratory-related hospitalizations in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2017;196(6):756–61.
Van Manen MJG, Birring SS, Vancheri C, et al. Effect of pirfenidone on cough in patients with idiopathic pulmonary fibrosis. Eur Respir J. 2017;50(4):1701157.
Oltmanns U, Kahn N, Palmowski K, Trager A, Wenz H, Heussel CP. Pirfenidone in idiopathic pulmonary fibrosis: real-life experience from a German tertiary referral center for interstitial lung diseases. Respiration. 2014;88:199–207.
Lancaster L, de Andrade JA, Zibrak JD, Padilla ML, Albera C, Nathan SD. Pirfenidone safety and adverse event management in idiopathic pulmonary fibrosis. Eur Respir Rev. 2017;26(146):170057.
Tzouvelekis A, Karampitsakos T, Ntolios P, Tzilas V, Bouros E, Markozannes E. Longitudinal “real-world” outcomes of pirfenidone in idiopathic pulmonary fibrosis in Greece. Front Med (Lausanne). 2017;29(4):213.
Salih GN, Shaker SB, Madsen HD, Bendstrup E. Pirfenidone treatment in idiopathic pulmonary fibrosis: nationwide Danish results. Eur Clin Respir J. 2016;9(3):32608.
Skold CM, Janson C, Elf AK, Fiaschi M, Wiklund K, Persson HL. A retrospective chart review of pirfenidone-treated patients in Sweden: the REPRIS study. Eur Clin Respir J. 2016;18(3):32035.
Chaudhuri N, Duck A, Frank R, Holme J, Leonard C. Real world experiences: pirfenidone is well tolerated in patients with idiopathic pulmonary fibrosis. Respir Med. 2014;108:224–6.
Cottin V, Koschel D, Günther A. Long-term safety of pirfenidone: results of the prospective, observational PASSPORT study. ERJ Open Res. 2018:19:00084-2018. https://doi.org/10.1183/23120541.00084-2018.
Acknowledgements
Funding
The study and the article processing charges were funded by an unrestricted grant from Roche. The funding source had no input in the design, data collection and analysis and writing of the manuscript. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
İsmail Hanta, Aykut Cilli and Can Sevinc have nothing to disclose.
Compliance with Ethics Guidelines
This multicenter, retrospective research was conducted on the archive records of three chest diseases departments. For this kind of research, to obtain ethical committee approval was not an obligation, legally in Turkey and institutionally in Çukurova University.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced Digital Features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.7813736.
Rights and permissions
About this article
Cite this article
Hanta, I., Cilli, A. & Sevinc, C. The Effectiveness, Safety, and Tolerability of Pirfenidone in Idiopathic Pulmonary Fibrosis: A Retrospective Study. Adv Ther 36, 1126–1131 (2019). https://doi.org/10.1007/s12325-019-00928-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-019-00928-3