Abstract
The improvement of glycemic control in patients with type 2 diabetes (T2D) is an urgent need in Asia. East Asian T2D patients have distinct characteristics, including relatively low body weight, early impairment of islet beta cell function with reduced insulin secretion and a marked increase in postprandial blood glucose levels. Control of postprandial hyperglycemia and beta cell preservation are key elements of the therapeutic strategy for these patients. Glucagon-like peptide-1 receptor agonists (GLP-1RAs) reduce HbA1c, fasting plasma glucose, and (in particular) postprandial glucose levels, and slow gastric emptying, while minimizing the risk of hypoglycemia and weight gain. Compared with long-acting GLP-1RAs, short-acting GLP-1RAs produce greater slowing of gastric emptying (which is a key factor driving the reduction of postprandial glycemia) and induce beta cell rest rather than promoting postprandial insulin secretion when used under physiological conditions. GLP-1RAs have greater efficacy in Asian than Caucasian patients. GLP-1RA add-on therapy provides clinically meaningful reductions in HbA1c and postprandial glucose in Asians with T2D inadequately controlled by oral antidiabetic drugs (OADs) or basal insulin ± OADs. Premixed insulin is often prescribed for T2D patients in China. A short-acting GLP-1RA plus basal insulin is an alternative to premixed insulin, resulting in better efficacy and a lower risk of hypoglycemia and weight gain. In conclusion, GLP-1RAs, especially short-acting GLP-1RAs, are a practical treatment option for East Asian patients with T2D inadequately controlled by OADs or basal insulin.
Funding: Sanofi.
Similar content being viewed by others
References
Unnikrishnan R, Pradeepa R, Joshi SR, Mohan V. Type 2 diabetes: demystifying the global epidemic. Diabetes. 2017;66:1432–42.
Blind E, Janssen H, Dunder K, de Graeff PA. The European Medicines Agency’s approval of new medicines for type 2 diabetes. Diabetes Obes Metab. 2018;20:2059–63.
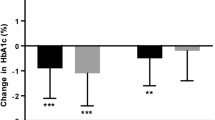
Shu H, Gu LN, Men LC, Lu JM. Lixisenatide improves glycemic control in Asian type 2 diabetic patients inadequately controlled with oral antidiabetic drugs: an individual patient data meta-analysis. Diabetes Ther. 2016;7:777–92.
Ji LN, Lu JM, Guo XH, et al. Glycemic control among patients in China with type 2 diabetes mellitus receiving oral drugs or injectables. BMC Public Health. 2013;13:602.
Ma RC, Chan JC. Type 2 diabetes in East Asians: similarities and differences with populations in Europe and the United States. Ann N Y Acad Sci. 2013;1281:64–91.
Liu Z, Fu C, Wang W, Xu B. Prevalence of chronic complications of type 2 diabetes mellitus in outpatients - a cross-sectional hospital based survey in urban China. Health Qual Life Outcomes. 2010;26(8):62.
Yabe D, Seino Y, Fukushima M, Seino S. β cell dysfunction versus insulin resistance in the pathogenesis of type 2 diabetes in East Asians. Curr Diab Rep. 2015;15(6):602.
Seino Y, Kuwata H, Yabe D. Incretin-based drugs for type 2 diabetes: focus on East Asian perspectives. J Diabetes Investig. 2016;7(Suppl 1):102–9.
Weng J, Ji L, Jia W, et al. Standards of care for type 2 diabetes in China. Diabetes Metab Res Rev. 2016;32:442–58.
Yang W, Weng J. Early therapy for type 2 diabetes in China. Lancet Diabetes Endocrinol. 2014;2:992–1002.
Sfairopoulos D, Liatis S, Tigas S, Liberopoulos E. Clinical pharmacology of glucagon-like peptide-1 receptor agonists. Hormones (Athens). 2018;17:333–50.
Guo XH. The value of short- and long-acting glucagon-like peptide-1 agonists in the management of type 2 diabetes mellitus: experience with exenatide. Curr Med Res Opin. 2016;32:61–76.
Linnebjerg H, Park S, Kothare PA, et al. Effect of exenatide on gastric emptying and relationship to postprandial glycemia in type 2 diabetes. Regul Pept. 2008;151:123–9.
DeFronzo RA, Okerson T, Viswanathan P, Guan X, Holcombe JH, MacConell L. Effects of exenatide versus sitagliptin on postprandial glucose, insulin and glucagon secretion, gastric emptying, and caloric intake: a randomized, cross-over study. Curr Med Res Opin. 2008;24:2943–52.
Cervera A, Wajcberg E, Sriwijitkamol A, et al. Mechanism of action of exenatide to reduce postprandial hyperglycemia in type 2 diabetes. Am J Physiol Endocrinol Metab. 2008;294:E846–52.
Meier JJ, Rosenstock J, Hincelin-Méry A, et al. Contrasting effects of lixisenatide and liraglutide on postprandial glycemic control, gastric emptying, and safety parameters in patients with type 2 diabetes on optimized insulin glargine with or without metformin: a randomized, open-label trial. Diabetes Care. 2015;38:1263–73.
Becker RH, Stechl J, Steinstraesser A, Golor G, Pellissier F. Lixisenatide reduces postprandial hyperglycaemia via gastrostatic and insulinotropic effects. Diabetes Metab Res Rev. 2015;31:610–8.
Lorenz M, Pfeiffer C, Steinsträsser A, et al. Effects of lixisenatide once daily on gastric emptying in type 2 diabetes–relationship to postprandial glycemia. Regul Pept. 2013;185:1–8.
van Raalte DH, Verchere CB. Improving glycaemic control in type 2 diabetes: stimulate insulin secretion or provide beta-cell rest? Diabetes Obes Metab. 2017;19:1205–13.
Kolterman OG, Buse JB, Fineman MS, et al. Synthetic exendin-4 (exenatide) significantly reduces postprandial and fasting plasma glucose in subjects with type 2 diabetes. J Clin Endocrinol Metab. 2003;88:3082–9.
van Raalte DH, Bunck MC, Smits MM, et al. Exenatide improves β-cell function up to 3 years of treatment in patients with type 2 diabetes: a randomised controlled trial. Eur J Endocrinol. 2016;175:345–52.
Drab SR. Glucagon-like peptide-1 receptor agonists for type 2 diabetes: a clinical update of safety and efficacy. Curr Diabetes Rev. 2016;12:403–13.
Kim HJ, Park SO, Ko SH, et al. Glucagon-like peptide-1 receptor agonists for the treatment of type 2 diabetes mellitus: a position statement of the Korean Diabetes Association. Diabetes Metab J. 2017;41:423–9.
Haneda M, Noda M, Origasa H, et al. Japanese clinical practice guideline for diabetes 2016. J Diabetes Investig. 2018. https://doi.org/10.1111/jdi.12810.
Chinese Diabetes Society. Chinese guideline for the prevention and treatment of type 2 diabetes mellitus (2017 edition). Chin J Diabetes Mellitus. 2018;10(1):4–67.
Kim YG, Hahn S, Oh TJ, Park KS, Cho YM. Differences in the HbA1c-lowering efficacy of glucagon-like peptide-1 analogues between Asians and non-Asians: a systematic review and meta-analysis. Diabetes Obes Metab. 2014;16:900–9.
Kim YG, Hahn S, Oh TJ, Kwak SH, Park KS, Cho YM. Differences in the glucose-lowering efficacy of dipeptidyl peptidase-4 inhibitors between Asians and non-Asians: a systematic review and meta-analysis. Diabetologia. 2013;56:696–708.
Park H, Park C, Kim Y, Rascati KL. Efficacy and safety of dipeptidyl peptidase-4 inhibitors in type 2 diabetes: meta-analysis. Ann Pharmacother. 2012;46:1453–69.
Cho YM. Incretin physiology and pathophysiology from an Asian perspective. J Diabetes Investig. 2015;6:495–507.
Zhang F, Tang L, Zhang Y, Lü Q, Tong N. Glucagon-like peptide-1 mimetics, optimal for Asian type 2 diabetes patients with and without overweight/obesity: meta-analysis of randomized controlled trials. Sci Rep. 2017;7(1):15997.
Ma RC, Hu C, Tam CH, et al. Genome-wide association study in a Chinese population identifies a susceptibility locus for type 2 diabetes at 7q32 near PAX4. Diabetologia. 2013;56:1291–305.
Wang JS, Tu ST, Lee IT, et al. Contribution of postprandial glucose to excess hyperglycaemia in Asian type 2 diabetic patients using continuous glucose monitoring. Diabetes Metab Res Rev. 2011;27:79–84.
Zhang XM, Li PF, Hou JN, Ji LN. Blood glucose profiles in East Asian and Caucasian injection-naive patients with type 2 diabetes mellitus inadequately controlled on oral antihyperglycemic medication: pooled analysis of individual patient data. Diabetes Metab Res Rev. 2018;15:e3062.
Sheu WH, Brunell SC, Blase E. Efficacy and tolerability of exenatide twice daily and exenatide once weekly in Asian versus White patients with type 2 diabetes mellitus: a pooled analysis. Diabetes Res Clin Pract. 2016;114:160–72.
Seino H, Onishi Y, Naito Y, Komatsu M. Lixisenatide improves glycemic outcomes of Japanese patients with type 2 diabetes: a meta-analysis. Diabetol Metab Syndr. 2016;1(8):36.
Bergenstal RM, Strock E, Mazze R, et al. Diurnal glucose exposure profiles of patients treated with lixisenatide before breakfast or the main meal of the day: an analysis using continuous glucose monitoring. Diabetes Metab Res Rev. 2017. https://doi.org/10.1002/dmrr.2879.
Lipska KJ, Yao X, Herrin J, et al. Trends in drug utilization, glycemic control, and rates of severe hypoglycemia, 2006-2013. Diabetes Care. 2017;40:468–75.
Eng C, Kramer CK, Zinman B, Retnakaran R. Glucagon-like peptide-1 receptor agonist and basal insulin combination treatment for the management of type 2 diabetes: a systematic review and meta-analysis. Lancet. 2014;384:2228–34.
Cimmaruta D, Maiorino MI, Scavone C, et al. Efficacy and safety of insulin-GLP-1 receptor agonists combination in type 2 diabetes mellitus: a systematic review. Expert Opin Drug Saf. 2016;15(Suppl. 2):77–83.
Maiorino MI, Chiodini P, Bellastella G, Capuano A, Esposito K, Giugliano D. Insulin and glucagon-like peptide 1 receptor agonist combination therapy in type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Diabetes Care. 2017;40:614–24.
Seino Y, Min KW, Niemoeller E, Takami A, EFC10887 GETGOAL-L Asia Study Investigators. Randomized, double-blind, placebo-controlled trial of the once-daily GLP-1 receptor agonist lixisenatide in Asian patients with type 2 diabetes insufficiently controlled on basal insulin with or without a sulfonylurea (GetGoal-L-Asia). Diabetes Obes Metab. 2012;14:910–7.
Yang W, Min K, Zhou Z, et al. Efficacy and safety of lixisenatide in a predominantly Asian population with type 2 diabetes insufficiently controlled with basal insulin: the GetGoal-L-C randomized trial. Diabetes Obes Metab. 2018;20:335–43.
Yoon KH, Hardy E, Han J. Exenatide versus insulin lispro added to basal insulin in a subgroup of Korean patients with type 2 diabetes mellitus. Diabetes Metab J. 2017;41(1):69–74.
Božek T, Bilić-Ćurčić I, Berković MC, et al. The effectiveness of lixisenatide as an add on therapy to basal insulin in diabetic type 2 patients previously treated with different insulin regimes: a multi-center observational study. Diabetol Metab Syndr. 2018;13(10):16.
Acknowledgements
Funding
This review and the article processing charges were funded by Sanofi. The author had full access to the information used to compile this work and takes complete responsibility for the integrity and accuracy of this review.
Medical Writing and Editorial Assistance
Medical writing and editorial assistance was provided by Kathy Croom and Steve Clissold of Content Ed Net and was funded by Sanofi.
Authorship
The author meets the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article and takes responsibility for the integrity of the work as a whole, and has given his approval for this version to be published.
Disclosures
In addition to this project for Sanofi, Ju-Ming Lu reported receiving consulting fees from Novo Nordisk and lecture fees from Bayer, Merck, and Lilly.
Compliance with Ethics Guidelines
This mini review is based on previously conducted studies and does not contain any studies with human participants or animals performed by the authors.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced Digital Features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.7745426.
Rights and permissions
About this article
Cite this article
Lu, JM. The Role of Glucagon-Like Peptide-1 Receptor Agonists in Type 2 Diabetes in Asia. Adv Ther 36, 798–805 (2019). https://doi.org/10.1007/s12325-019-00914-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-019-00914-9