Abstract
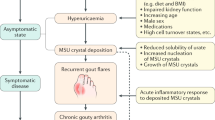
Gout is a disease caused by deposition of monosodium urate crystals in tissues. One of the limitations for successful treatment of gout is to consider it as an intermittent disease rather than a chronic inflammatory disease which, if improperly treated, leads to chronic clinical manifestations. In addition, gout is linked to increased cardiovascular morbidity and mortality.
Urate-lowering therapy comprises both nonpharmacologic and pharmacologic interventions, but most patients will need uratelowering drugs to achieve target therapeutic serum urate levels. Reaching target serum urate levels is associated with improvement in clinical outcomes, including a reduction of acute inflammation episodes, resolution of tophi, and improvement in health-related quality of life perception.
A number of urate-lowering drugs are available but a number of patients fail to achieve or maintain therapeutic serum urate levels and go on to develop refractory chronic gout. For such patients, efforts have been made to develop new treatments (e.g., febuxostat or pegloticase).
This review intends to increase the awareness of gout as a chronic deposition disease, and show that efforts should be made to properly control serum urate levels in order to achieve complete disappearance of urate crystal deposition.
Similar content being viewed by others
References
Perez-Ruiz F, Lioté F. Lowering serum uric acid levels: what is the optimal target for improving clinical outcomes in gout? Arthritis Rheum. 2007;57:1324–1328.
Puig JG, de Miguel E, Castillo MC, Rocha AL, Martinez MA, Torres RJ. Asymptomatic hyperuricemia: impact of ultrasonography. Nucleosides Nucleotides Nucleic Acids. 2008;27:592–595.
Pascual E. Persistence of monosodium urate crystals and low-grade inflammation in synovial fluid of patients with untreated gout. Arthritis Rheum. 1991;34:141–145.
Gutman AB. Treatment of primary gout: the present status. Arthitis Rheum. 1965;8:911–920.
Gutman AB. The past four decades of progress in the knowledge of gout, with an assessment of the present status. Arthritis Rheum. 1973;16:431–445.
Khanna PP, Perez-Ruiz F, Maranian P, Khanna D. Long-term therapy for chronic gout results in clinically important improvements in the healthrelated quality of life: short form-36 is responsive to change in chronic gout. Rheumatology (Oxford). 2011;50:740–745.
Perez-Ruiz F, Herrero-Beites AM, Gonzalez-Gay MA. Number of flares are associated with both gastrointestinal and vascular complications in patients with gout. Another issue for early intervention. Arthritis Rheum. 2010;62(Suppl.): S366.
Lottmann K, Chen X, Schadlich PK. Association between gout and all-cause as well as cardiovascular mortality: a systematic review. Curr Rheumatol Rep. 2012;14:195–203.
Kim SY, Guevara JP, Kim KM, Choi HK, Heitjan DF, Albert DA. Hyperuricemia and coronary heart disease: a systematic review and meta-analysis. Arthitis Rheum. 2010;62:170–180.
Kim SY, Guevara JP, Kim KM, Choi HK, Heitjan DF, Albert DA. Hyperuricemia and risk of stroke: a systematic review and meta-analysis. Arthritis Rheum 2009;61:885–892.
Krishnan E, Backer JF, Furst DE, Schumacher HR. Gout and the risk of acute myocardial infarction. Arthritis Rheum. 2006;54:2688–2696.
Krishnan E, Svendsen K, Neaton JD, Grandits G, Kuller LH. Long-term cardiovascular mortality among middle-aged men with gout. Arch Intern Med. 2008;168:1104–1110.
Choi HK, Curhan G. Independent impact of gout on mortality and risk for coronary heart disease. Circulation. 2007;116:894–900.
Chen SY, Chen CL, Shen ML. Severity of gouty arthritis is associated with Q-wave myocardial infarction: a large-scale, cross-sectional study. Clin Rheumatol. 2007;26:308–313.
Cappola TP, Kass DA, Nelson GS, et al. Allopurinol increases myocardial efficiency in patients with idiopathic dilated myocardiopathy. Circulation. 2001;104:2407–2411.
Crittenden DB, Lehmann RA, Schneck L, et al. Colchicine use is associated with decreased prevalence of myocardial infarction in patients with gout. J Rheumatol. 2012;39:1458–1464.
Choi HK, Liu S, Curhan G. Intake of purine-rich foods, protein, and dairy products and relationship to serum levels of uric acid: the Third National Health and Nutrition Examination Survey. Arthritis Rheum. 2005;52:283–289.
Gaffo AL, Roseman JM, Jacobs DR, Jr, et al. Serum urate and its relationship with alcoholic beverage intake in men and women: findings from the Coronary Artery Risk Development in Young Adults (CARDIA) cohort. Ann Rheum Dis. 2010;69:1965–1970.
Zhu Y, Zhang Y, Choi HK. The serum uratelowering impact of weight loss among men with a high cardiovascular risk profile: the Multiple Risk Factor Intervention Trial. Rheumatology (Oxford). 2010;49:2391–2399.
Khanna D, FitzGerald JD, Khanna PP, et al. 2012 American College of Rheumatology Guidelines for management of gout Part I: Systematic nonpharmacologic and pharmacologic therapeutic approaches to hyperuricemia. Arthitis Rheum. 2012;64:1431–1446.
Perez-Ruiz F. New treatments for gout. Bone Joint Spine. 2007;74:313–315.
Cea Soriano L, Rothenbacher D, Choi HK, Garcia Rodriguez LA. Contemporary epidemiology of gout in the UK general population. Arthritis Res Ther. 2011;13:R39.
Annemans L, Spaepen E, Gaskin M, et al. Gout in the UK and Germany: prevalence, comorbidities and management in general practice 2000–2005. Ann Rheum Dis. 2007;67:960–966.
Zhang W, Doherty M, Pascual E, et al; EULAR Committee for International Clinical Studies including Therapeutics. EULAR evidence based recommendations for gout. Part II. Management. Report of a Task Force of the EULAR Standing Committee for international clinical studies including therapeutics (ESCISIT). Ann Rheum Dis. 2006;65:1312–1324.
Jordan KM, Cameron JS, Snaith M, et al. British Society for Rheumatology and British Health Professionals in Rheumatology Guideline for the Management of Gout. Rheumatology (Oxford). 2007;46:1372–1374.
Hamburger M, Baraf HS, Adamson TC, et al. 2011 recommendations for the diagnosis and management of gout and hyperuricemia. Phys Sportsmed. 2011;39:98–123.
Reinders MK, Haggsma C, Jansen TL, et al. A randomized-controlled trial on the efficacy and tolerability with dose escalation of allopurinol 300–600 mg/day versus benzbromarone 100–200 mg/day in patients with gout. Ann Rheum Dis. 2008;68:892–897.
Becker MA, Schumacher HR Jr, Wortmann RL, et al. Febuxostat compared with allopurinol in patients with hyperuricemia and gout. N Engl J Med. 2005;353:2450–2461.
Sundy JS, Baraf HSB, Yood RA, et al. Efficacy and tolerability of pegloticase for the treatment of chronic gout in patients refractory to conventional treatment. Two randomized controlled trials. JAMA. 2011;306:711–720.
Perez-Ruiz F, Calabozo M, Fernandez-Lopez MJ, et al. Treatment of chronic gout in patients with renal function impairment. An open, randomized, actively controlled. J Clin Rheumatol. 1999;5:49–55.
Perez-Ruiz F, Alonso-Ruiz A, Calabozo M, Herrero-Beites A, Garcia-Erauskin G, Ruiz-Lucea E. Efficacy of allopurinol and benzbromarone for the control of hyperuricaemia. A pathogenic approach to the treatment of primary chronic gout. Ann Rheum Dis. 1998;57:545–549
Reinders MK, Van Roon EN, Houtman PM, Brouwers JR, Jansen TL. Biochemical effectiveness of allopurinol and allopurinol-probenecid in previously benzbromarone-treated gout patients. Clin Rheumatol. 2007;26:1459–1465.
Perez-Ruiz F. Treating to target: an strategy to cure gout. Rheumatology (Oxford). 2009;48(Suppl. 2):ii9–ii14.
Stamp LK, Khanna PP, Dalbeth N, et al. Serum urate in chronic gout — will it be the first validated soluble biomarker in rheumatology? J Rheumatol. 2011;38:1462–1466.
Perez-Ruiz F, Calabozo M, Pijoan JI, Herrero-Beites AM, Ruibal A. Effect of urate-lowering therapy on the velocity of size reduction of tophi in chronic gout. Arthritis Rheum. 2002;47:356–360.
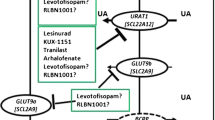
Enomoto A, Endou H. Roles of organic anion transporters (OATs) and a urate transporter (URAT1) in the pathophysiology of human disease. Clin Exp Nephrol. 2005;9:195–205.
Perez-Ruiz F, Herrero-Beites AM, Carmona L. A twostage approach to the treatment of hyperuricemia in gout: The “Dirty Dish” hypothesis. Arthritis Rheum. 2011;63:4002–4006.
Perez-Ruiz F, Atxotegi J, Hernando I, Calabozo M, Nolla JM. Using serum urate levels to determine the period free of gouty symptoms after withdrawal of long-term urate-lowering therapy: a prospective study. Arthritis Rheum. 2006;55:786–790.
Wortmann RL, MacDonald PA, Hunt B, Jackson RL. Effect of prophylaxis on gout flares after the initiation of urate-lowering therapy: analysis of data from three phase III trials. Clin Ther. 2011;32:2386–2397.
Schumacher HR Jr, Becker MA, Lloyd E, MacDonald PA, Lademacher C. Febuxostat in the treatment of gout: 5-yr findings of the FOCUS efficacy and safety study. Rheumatology (Oxford). 2009;48:188–194.
Becker MA, Schumacher HR, MacDonald PA, Lloyd E, Lademacher C. Clinical efficacy and safety of successful longterm urate lowering with febuxostat or allopurinol in subjects with gout. J Rheumatol. 2009;36:1273–1282.
Dalbeth N, Schauer C, MacDonald P, et al. Methods of tophus assessment in clinical trials of chronic gout: a systematic literature review and pictorial reference guide. Ann Rheum Dis. 2011;70:597–604.
Perez-Ruiz F, Martin I, Canteli B. Ultrasonographic measurement of tophi as an outcome measure for chronic gout. J Rheumatol. 2007;34:1888–1893.
Perez-Ruiz F, Schlesinger N, Dalbeth N, Urresola A, De Miguel E. Imaging of gout: findings and utility. Arthritis Res Ther. 2009;11:232.
Edwards LN. Treatment-failure gout: a moving target. Arthritis Rheum. 2008;58:2587–2590.
Lipworth W, Kerridge I, Brett J, Day R. How clinical and research failures lead to suboptimal prescribing: the example of chronic gout. BMJ. 2011;343:d7459.
Perez-Ruiz F, Carmona L, Yebenes MJ, et al. An audit of the variability of diagnosis and management of gout in the rheumatology setting: the gout evaluation and management study. J Clin Rheumatol. 2011;17:349–355.
Dalbeth N, Petrie KJ, House M, et al. Illness perceptions in patients with gout and the relationship with progression of musculoskeletal disability. Arthritis Care Res (Hoboken). 2011;63:1605–1612.
Rees F, Jenkins W, Doherty M. Patients with gout adhere to curative treatment if informed appropriately: proof-of-concept observational study. Ann Rheum Dis. 2012 [Epub ahead of print].
Edwards LN, Sundy JS, Forsythe A, Blume S, Pan F, Becker MA. Work productivity loss dueto flares in patient swith chronic gout refractory to conventional therapy. J Med Econ. 2011;14:10–15.
Brook RA, Forsythe A, Smeeding JE, Edwards LN. Chronic gout: epidemiology, disease progression, treatment and disease burden. Cur Med Res Opin. 2010;26:2813–2821.
Wu EQ, Forsythe A, Guérin A, Yu AP, Latremouille-Viau D, Tsaneva M. Comorbidity burden, healthcare resource utilization, and costs in chronic gout patients refractory to conventional urate-lowering therapy. Am J Ther. 2011 [Epub ahed of print].
Richette P, Briere C, Honene-Claviert V, Loeuille D, Bardin T. Rasburicase for tophaceous gout not treatable with allopurinol: an exploratory study. J Rheumatol. 2007;34:1093–2098.
Sherman MR, Saifer MGP, Perez-Ruiz F. PEGuricase in the management of treatment-resistant gout and hyperuricemia. Adv Drug Deliv Rev. 2008;60:59–68.
Yue CS, Huang W, Alton M, et al. Population pharmacokinetic and pharmacodynamic analysis of pegloticase in subjects with hyperuricemia and treatment-failure gout. J Clin Pharmacol. 2008;48:708–718.
Sundy JS, Becker MA, Baraf HSB, et al; Pegloticase Phase 2 Study Investigators. Reduction of plasma urate levels following treatment with multiple doses of pegloticase (Polyethylene glycolconjugated uricase) in patients with treatment-failure gout. Results of a phase II randomized study. Arthritis Rheum. 2008;58:2882–2891.
Hershfield MS, Roberts LJ, Ganson NJ, et al. Treating gout with pegloticase, a PEGylated urate oxidase, provides insight into the importance of uric acid as an antioxidant in vivo. Proc Natl Acad Sci U S A. 2010;107:14351–14356.
Perez-Ruiz F, Sundy J, Krishnan E, et al. Efficacy and safety of Lesinurad (RDEA594), a novel uricosuric agent, given in combination with allopurinol in allopurinol-refractory gout patients: randomized, double-blind, placebo-controlled, Phase 2B study. Ann Rheum Dis. 2011;70(Suppl. 3):104.
Fleishmann R, Shen Z, Yeh LT, et al. Lesinurad (RDEA594), a novel uricosuric agent, in combination with febuxostat shows significant additive urate-lowering effects in gout patients with 100% response achieved for all combination dose regimens. Ann Rheum Dis. 2011;70(Suppl. 3):188.
Hollister AS, Becker M, Terkeltaub R, et al. BCX4208 shows synergistic reductions in serum uric acid in gout patients when combined with allopurinol. Ann Rheum Dis. 2011;70(Suppl. 3):183.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Perez-Ruiz, F., Herrero-Beites, A.M. Evaluation and Treatment of Gout as a Chronic Disease. Adv Therapy 29, 935–946 (2012). https://doi.org/10.1007/s12325-012-0059-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-012-0059-z