Abstract
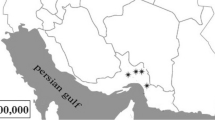
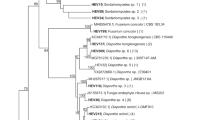
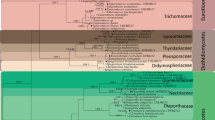
Ecological niches in the rhizosphere and phyllosphere of grasses capable of sustaining endophytes have been extensively studied. In contrast, little information regarding the identity and functions of endophytic fungi in stems is available. In this study, we investigated the taxonomic affinities, diversity, and host specificities of culturable endophytes in stems of wild rice (Oryza granulata) in China. Seventy-four isolates were recovered. Low recovery rate (11.7%) indicated that there were relatively few sites for fungal infection. Identification using morphology, morphospecies sorting, and molecular techniques resulted in classification into 50 taxa, 36 of which were recovered only once. Nucleotide sequence similarity analysis indicated that 30% of the total taxa recovered were highly divergent from known species and thus may represent lineages new to science. Most of the taxa were classified as members of the classes Sordariomycetes or Dothideomycetes (mainly in Pleosporales). The presence of Arthrinium and Magnaporthaceae species, most often associated with poaceous plants, suggested a degree of host specificity. A polyphasic approach was employed to identify two Muscodor taxa based on (i) ITS and RPB2 phylogenies, (ii) volatile compounds produced, and (iii) an in vitro bioassay of antifungal activity. This to our knowledge is only the second report regarding the isolation of Muscodor spp. in China. Therefore, we hypothesize that wild plants represent a huge reservoir of unknown fungi. The prevalence, novelty, and species-specificity of unique isolates necessitate a reevaluation of their contribution to ecosystem function and fungal biodiversity.
Similar content being viewed by others
References
Arnold, A.E. 2007. Understanding the diversity of foliar fungal endophytes: progress, challenges, and frontiers. Fungal Biol. Rev. 21, 51–66.
Arnold, A.E., D.A. Henk, R.L. Eells, F. Lutzoni, and R. Vilgalys. 2007. Diversity and phylogenetic affinities of foliar fungal endophytes in loblolly pine inferred by culturing and environmental PCR. Mycologia 99, 185–206.
Arnold, A.E., L.C. Mejía, D. Kyllo, E.I. Rojas, Z. Maynard, N. Robbins, and E.A. Herre. 2003. Fungal endophytes limit pathogen damage in a tropical tree. Proc. Natl. Acad. Sci. USA 100, 15649–15654.
Chaintreuil, C., E. Giraud, Y. Prin, J. Lorquin, A. Ba, M. Gillis, P. de Lajudie, and B. Dreyfus. 2000. Photosynthetic bradyrhizobia are natural endophytes of the African wild rice Oryza breviligulata. Appl. Environ. Microbiol. 66, 5437–5447.
Clay, K. 2004. Fungi and the food of the gods. Nature 427, 401–402.
Daisy, B., G.A. Strobel, D. Ezra, U. Castillo, G. Baird, and W.M. Hess. 2002. Muscodor vitigenus anam. sp. nov., an endophyte from Paullinia paullinioides. Mycotaxon 84, 39–50.
Davis, E.C., J.B. Franklin, A.J. Shaw, and R. Vilgalys. 2003. Endophytic Xylaria (Xylariaceae) among liverworts and angiosperms: phylogenetics, distribution and symbiosis. Am. J. Bot. 90, 1661–1667.
Deshmukh, S., R. Hückelhoven, P. Schäfer, J. Imani, M. Sharma, M. Weiss, F. Waller, and K.H. Kogel. 2005. The endophytic fungus Piriformospora indica reprograms barley to salt-stress tolerance, disease resistance, and higher yield. Proc. Natl. Acad. Sci. USA 102, 13386–13391.
Gallery, R., J.W. Dalling, and A.E. Arnold. 2007. Diversity, host affinity, and distribution of seed-infecting fungi: a case study with neotropical Cecropia. Ecology 88, 582–588.
Ganley, R.J., S.J. Brunsfeld, and G. Newcombe. 2004. A community of unknown, endophytic fungi in western white pine. Proc. Natl. Acad. Sci. USA 101, 10107–10112.
González, M.C., A.L. Anaya, A.E. Glenn, M.L. Macías-Rubalcava, B.E. Hernández-Bautista, and R.T. Hanlin. 2009. Muscodor yucatanensis, a new endophytic ascomycete from Mexican chakah, Bursera simaruba. Mycotaxon 110, 363–372.
Henson, J.M. 1989. DNA probe for identification of the take-all fungus, Gaeumannomyces graminis. Appl. Environ. Microbiol. 55, 284–288.
Hung, P.Q., S.M. Kumar, V. Govindsamy, and K. Annapurna. 2009. Isolation and characterization of endophytic bacteria from wild and cultivated soybean varieties. Biol. Fert. Soils 44, 155–162.
Jeffries, P. 2004. Microbial symbioses with plants, pp. 204–210. In A.T. Bull (ed.), Microbial Diversity and Bioprospecting, American Society for Microbiology, Washington, USA.
Joshee, S., B.C. Paulus, D. Park, and P.R. Johnston. 2009. Diversity and distribution of fungal foliar endophytes in New Zealand Podocarpaceae. Mycol. Res. 113, 1003–1015.
Kageyama, S.A., K.G. Mandyam, and A. Jumpponen. 2009. Diversity, function and potential applications of the root-associated endophytes, pp. 29–57. In A. Varma (ed.), Mycorrhiza-State of the Art, Genetics and Molecular Biology, Eco-Function, Biotechnology, Eco-Physiology, Structure and Systematics, Springer-Verlag, Berlin Heidelberg, Germany.
Kharkwal, A.C., H. Kharkwal, I. Sherameti, R. Oelmuller, and A. Varma. 2008. Novel symbiotrophic endophytes, pp. 753–766. In A. Varma (ed.), Mycorrhiza-State of the Art, Genetics and Molecular Biology, Eco-Function, Biotechnology, Eco-Physiology, Structure and Systematics, Springer-Verlag, Berlin Heidelberg, Germany.
Kuldau, G. and C. Bacon. 2008. Clavicipitaceous endophytes: Their ability to enhance resistance of grasses to multiple stresses. Biol. Control. 46, 57–71.
Larena, I., O. Salazar, V. González, M.C. Julián, and V. Rubio. 1999. Design of a primer for ribosomal DNA internal transcribed spacer with enhanced specificity for ascomycetes. J. Biotechnol. 75, 187–194.
Liu, Y., S. Whelen, and B.D. Hall. 1999. Phylogenetic relationships among ascomycetes: evidence from an RNA polymerase II subunit. Mol. Biol. Evol. 16, 1799–1808.
Maciá-Vicente, J.G., H.B. Jansson, S.K. Abdullah, E. Descals, J. Salinas, and L.V. Lopez-Llorca. 2008. Fungal root endophytes from natural vegetation in Mediterranean environments with special reference to Fusarium spp. FEMS Microbiol. Ecol. 64, 90–105.
Malkus, A., P.L. Chang, S.M. Zuzga, K. Chung, J. Shao, B.M. Cunfer, E. Arseniuk, and P.P. Ueng. 2006. RNA polymerase II gene (RPB2) encoding the second largest protein subunit in Phaeosphaeria nodorum and P. avenaria. Mycol. Res. 110, 1152–1164.
Mitchell, A.M., G.A. Strobel, W.M. Hess, P.N. Vargas, and D. Ezra. 2008. Muscodor crispans, a novel endophyte from Ananas ananassoides in the Bolivian Amazon. Fung. Divers. 31, 37–44.
Morakotkarn, D., H. Kawasaki, and T. Seki. 2007. Molecular diversity of bamboo-associated fungi isolated from Japan. FEMS Microbiol. Lett. 266, 10–19.
Naik, B.S., J. Shashikala, and Y.L. Krishnamurthy. 2009. Study on the diversity of endophytic communities from rice (Oryza sativa L.) and their antagonistic activities in vitro. Microbiol. Res. 164, 290–296.
Peng, G., Q. Yuan, H. Li, W. Zhang, and Z. Tan. 2008. Rhizobium oryzae sp. nov., isolated from the wild rice Oryza alta. Int. J Syst. Evol. Microbiol. 58, 2158–2163.
Porras-Alfaro, A., J. Herrera, R.L. Sinsabaugh, K.J. Odenbach, T. Lowrey, and D.O. Natvig. 2008. Novel root fungal consortium associated with a dominant desert grass. Appl. Environ. Microbiol. 74, 2805–2813.
Qiu, W.H. and H. Z. Chen. 2008. An alkali-stable enzyme with laccase activity from endophytic fungus and the enzymatic modification of alkali lignin. Bioresour. Technol. 99, 5480–5484.
Rodriguez, R.J., J.F. White, Jr., A.E. Arnold, and R.S. Redman. 2009. Fungal endophytes: diversity and functional roles. New Phytol. 182, 314–330.
Rosa, L.H., A.B.M. Vaz, R.B. Caligiorne, S. Campolina, and C.A. Rosa. 2009. Endophytic fungi associated with the Antarctic grass Deschampsia antarctica Desv. (Poaceae). Polar Biol. 32, 161–167.
Samuels, G.J., E.H.C. McKenzie, and D.E. Buchanan. 1981. Ascomycetes of New Zealand two new species of Apiospora and their Arthrinium anamorphs on bamboo. N.Z. J. Bot. 19, 137–149.
Sánchez Márquez, S., G.F. Bills, and I. Zabalgogeazcoa. 2007. The endophytic mycobiota of the grass Dactylis glomerata. Fung. Divers. 27, 171–195.
Sánchez Márquez, S., G.F. Bills, and I. Zabalgogeazcoa. 2008. Diversity and structure of the fungal endophytic assemblages from two sympatric coastal grasses. Fung. Divers. 33, 87–100.
Schulz, B. and C. Boyle. 2005. The endophytic continuum. Mycol. Res. 109, 661–686.
Schulz, B., C. Boyle, S. Draeger, A.K. Römmert, and K. Krohn. 2002. Endophytic fungi: a source of novel biologically active secondary metabolites. Mycol. Res. 106, 996–1004.
Sieber, T.N. 2002. Fungal root endophytes, pp. 887–917. In Y. Wasel, A. Eshel, and U. Kafkafi (eds.), Plant roots: the hidden half, 3rd edition, Marcel Dekker, New York, N.Y., USA.
Sopalun, K., G.A. Strobel, W.M. Hess, and J. Worapong. 2003. A record of Muscodor albus, an endophyte from Myristica fragrans in Thailand. Mycotaxon 88, 239–247.
Stadler, M. and J. Fournier. 2006. Pigment chemistry, taxonomy and phylogeny of the Hypoxyloideae (Xylariaceae). Rev. Iberoam. Micol. 23, 160–170.
Stadler, M., J. Fournier, T. Laessoe, C. Lechat, H.V. Tichy, and M. Piepenbring. 2008. Recognition of hypoxyloid and xylarioid Entonaema species allied Xylaria species from a comparison of holomorphic morphology, HPLC profiles, and ribosomal DNA sequences. Mycol. Progress 7, 53–73.
Stadler, M. and N.P. Keller. 2008. Paradigm shifts in fungal secondary metabolite research. Mycol. Res. 112, 251–270.
Stone, J.K., J.D. Polishook, and J.F. White, Jr. 2004. Endophytic fungi, pp. 241–270. In G.M. Mueller, G.F. Bills, and M.S. Foster (eds.), Biodiversity of fungi: inventory and monitoring methods. Elsevier Academic Press, Burlington, Massachusetts, USA.
Strobel, G.A. 2006. Muscodor albus and its biological promise. J. Ind. Microbiol. 33, 514–522.
Strobel, G.A. 2007. Plant-associated microorganisms (endophytes) as a new source of bioactive natural products, pp. 49–70. In O. Kayser and W.J. Quax (eds.), Medicinal plant biotechnology: from basic research to industrial applications. Wiley-VCH.
Strobel, G.A., E. Dirske, J. Sears, and C. Markworth. 2001. Volatile antimicrobials from Muscodor albus, a novel endophytic fungus. Microbiology 147, 2943–2950.
Strobel, G.A., B. Knighton, K. Kluck, Y. Ren, T. Livinghouse, M. Griffin, D. Spakowicz, and J. Sears. 2008. The production of mycodiesel hydrocarbons and their derivatives by the endophytic fungus Gliocladium roseum (NRRL 50072). Microbiology 154, 3319–3328.
Tang, A.M.C., R. Jeewon, and K.D. Hyde. 2009. A re-evaluation of the evolutionary relationships within the Xylariaceae based on ribosomal and protein-coding gene sequences. Fung. Divers. 34, 127–155.
Thomas, S.E., J. Crozier, M.C. Aime, H.C. Evans, and K.A. Holmes. 2008. Molecular characterization of fungal endophytic morphospecies associated with the indigenous forest tree, Theobroma gileri in Ecuador. Mycol. Res. 112, 852–860.
Tian, X.L., L.X. Cao, H.M. Tan, Q.G. Zeng, Y.Y. Jia, W.Q. Han, and S.N. Zhou. 2004. Study on the communities of endophytic fungi and endophytic actinomycetes from rice and their antipathogenic activities in vitro. World J. Microbiol. Biotechnol. 20, 303–309.
Vallino, M., D. Greppi, M. Novero, P. Bonfante, and E. Lupotto. 2009. Rice root colonisation by mycorrhizal and endophytic fungi in aerobic soil. Ann. Appl. Biol. 154, 195–204.
Vandenkoornhuyse, P., S.L. Baldauf, C. Leyval, J. Straczek, and P. Young. 2002. Extensive fungal diversity in plant roots. Science 295, 2051.
White, T.J., S. Bruns, S. Lee, and J. Taylor. 1990. Amplification and direct sequencing of fungal genes for phylogenetics, pp. 315–322. In M.A. Innis, D.H. Gelfand, J.J. Sninsky, and T.J. White (eds.), PCR protocols: a guide to methods and applications. Academic Press, San Diego, USA.
Wong, P.T.W. 2002. Gaeumannomyces wongoonoo sp. nov., the cause of a patch disease of buffalo grass (syn. St Augustine grass). Mycol. Res. 106, 857–862.
Wong, M.K.M. and K.D. Hyde. 2001. Diversity of fungi on six species of Gramineae and one species of Cyperaceae in Hong Kong. Mycol. Res. 105, 1485–1491.
Wong, P.T.W. and J. Walker. 1975. Germinating phialidic conidia of Gaeumannomyces graminis and Phialophora-like fungi from Gramineae. Trans. Br. Mycol. Soc. 65, 41–47.
Worapong, J., G.A. Strobel, B. Daisy, U.F. Castillo, G. Baird, and W.M. Hess. 2002. Muscodor roseus anam. gen. et sp. nov., an endophyte from Grevillea pteridifolia. Mycotaxon 81, 463–475.
Worapong, J., G.A. Strobel, E.J. Ford, J.Y. Li, G. Baird, and W.M. Hess. 2001. Muscodor albus anam. gen. et sp. nov. an endophyte from Cinnamonum zeylanicum. Mycotaxon 79, 67–79.
Yuan, Z.L., F.C. Lin, C.L. Zhang, and C.P. Kubicek. 2010a. A new species of Harpophora (Magnaporthaceae) recovered from healthy wild rice (Oryza granulate) roots, representing a novel member of beneficial dark septate endophyte. FEMS Microbiol. Lett. 307, 94–101.
Yuan, Z.L., C.L. Zhang, F.C. Lin, and C.P. Kubicek. 2010b. Identity, diversity, and molecular phylogeny of the endophytic mycobiota in rare wild rice roots (Oryza granulate) from a nature reserve in Yunnan, China. Appl. Environ. Microbiol. 76, 1642–1652.
Zhang, G.X., G.X. Peng, E.T. Wang, H. Yan, Q.H. Yuan, W. Zhang, X. Lou, H. Wu, and Z.Y. Tan. 2008. Diverse endophytic nitrogen-fixing bacteria isolated from wild rice Oryza rufipogon and description of Phytobacter diazotrophicus gen. nov. sp. nov. Arch. Microbiol. 189, 431–439.
Zhang, C.L., G.P. Wang, L.J. Mao, M. Komon-Zelazowska, Z.L. Yuan, F.C. Lin, I.S. Druzhinina, and C.P. Kubicek. 2010. Muscodor fengyangensis sp. nov. from southeast China: morphology, physiology and production of volatile compounds. Fungal Biol. 114, 797–808.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplemental material for this article may be found at http://www.springer.com/content/120956
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Yuan, Zl., Su, Zz., Mao, Lj. et al. Distinctive endophytic fungal assemblage in stems of wild rice (Oryza granulata) in China with special reference to two species of Muscodor (xylariaceae). J Microbiol. 49, 15–23 (2011). https://doi.org/10.1007/s12275-011-0213-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-011-0213-3