Abstract
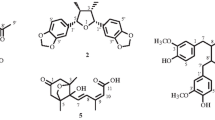
Brassica rapa ssp. campestris (Brassicaceae) is a conical, deep purple, edible root vegetable commonly known as a turnip. We initiated phytochemical and pharmacological studies to search for biological active compounds from the roots of B. rapa ssp. campestris. We isolated a novel phenanthrene derivative, 6-methoxy-1-[10-methoxy-7-(3-methylbut-2-enyl)phenanthren-3-yl]undecane-2,4-dione, named brassicaphenanthrene A (3) along with two known diarylheptanoid compounds, 6-paradol (1) and trans-6-shogaol (2), through the repeated silica gel (SiO2), octadecyl silica gel, and Sephadex LH-20 column chromatography. The chemical structures of the compounds were determined by spectroscopic data analyses including nuclear magnetic resonance, mass spectrometry, ultraviolet spectroscopy, and infra-red spectroscopy. All compounds exhibited high inhibitory activity against the growth of human cancer lines, HCT-116, MCF-7, and HeLa, with IC50 values ranging from 15.0 to 35.0 μM and against LDL-oxidation with IC50 values ranging from 2.9 to 7.1 μM.



Similar content being viewed by others
References
Agarwal, M., S. Walia, S. Dhingra, and B.P.S. Khambay. 2001. Insect growth inhibition, antioxidant and antifungal activity of compounds isolated/derived from Zingiber officinale Roscoe (ginger) rhizomes. Pest Management Science 57: 289–300.
An, S.J., J.I. Han, M.J. Kim, J.S. Park, J.M. Han, N.I. Baek, H.G. Chung, M.S. Choi, K.T. Lee, and T.S. Jeong. 2010. Ethanolic extracts of Brassica campestris ssp. rapa roots prevent high-fat diet-induced obesity via β 3-adrenergic regulation of white adipocyte lipolytic activity. Journal of Medicinal Food 13: 406–414.
Bang, M.H., D.Y. Lee, M.W. Han, Y.J. Oh, H.G. Chung, T.S. Jeong, K.T. Lee, M.S. Choi, and N.I. Baek. 2007. Development of biologically active compounds from edible plant sources-XX: Isolation of lipids from the roots of Brassica campestris ssp. rapa. Journal of the Korean Society for Applied Biological Chemistry 50: 233–237.
Bang, M.H., D.Y. Lee, Y.J. Oh, M.W. Han, H.J. Yang, H.G. Chung, T.S. Jeong, K.T. Lee, M.S. Choi, and N.I. Baek. 2008. Development of biological active compound from edible plant sources XXII. Isolation of indoles from the roots of Braccica campestris ssp. rapa and their hACAT inhibitory activity. Journal of the Korean Society for Applied Biological Chemistry 51: 65–69.
Chung, W.Y., Y.J. Jung, Y.J. Surh, S.S. Lee, and K.K. Park. 2001. Antioxidative and antitumor promoting effects of [6]-paradol and its homologs. Mutation Research-Genetic Toxicology and Environmental Mutagenesis 496: 199–206.
Jeong, T.S., Y.B. Ryu, H.Y. Kim, M.J. Curtis-Long, S.J. An, J.H. Lee, J.W.S. Lee, and K.H. Park. 2008. Low density lipoprotein (LDL)-antioxidant flavonoids from roots of Sophora flavescens. Biological and Pharmaceutical Bulletin 31: 2097–2102.
Jung, U.J., N.I. Baek, H.G. Chung, M.H. Bang, T.S. Jeong, K.T. Lee, Y.J. Kang, M.K. Lee, H.J. Kim, J.Y. Yeo, and M.S. Choi. 2008. Effects of the ethanol extract of the roots of Brassica rapa on glucose and lipid metabolism in C57BL/KsJ-db/db mice. Clinical Nutrition 2: 158–167.
Kim, J.S., Y.H. Choi, J.H. Seo, J.W. Lee, Y.S. Kim, S.Y. Ryu, J.S. Kang, Y.K. Kim, and S.H. Kim. 2004. Chemical constituents from the roots of Brassica campestris ssp. rapa. Journal of the Korean Society for Applied Biological Chemistry 35: 259–263.
Kim, Y.H., Y.W. Kim, Y.J. Oh, N.I. Baek, S.A. Chung, H.G. Chung, T.S. Jeong, M.S. Choi, and K.T. Lee. 2006. Protective effect of the ethanol extract of the roots of Brassica rapa on cisplatin-induced nephrotoxicity in LLC-PK1 cells and rats. Biological and Pharmaceutical Bulletin 29: 2436–2441.
Kuroyanagi, M., K. Umehara, T. Hayashi, and Y. Kirayama. 2002. Cell differentiation inducers and antiandrogenic active compounds from Zingiberis rhizoma. Journal of Natural Medicines 56: 47–50.
Lee, D.Y., M.H. Lee, T.S. Jung, B.M. Kwon, N.I. Baek, and Y.D. Rho. 2010. Triterpernoid and lignan from the fruits of Cornus kousa inhibit the activities of PRL-3 and LDL-oxidation. Journal of the Korean Society for Applied Biological Chemistry 53: 97–100.
Masuda, Y., H. Kikuzaki, M. Hisamoto, and N. Nakatani. 2004. Antioxidant properties of gingerol related compounds from ginger. BioFactors 21: 293–296.
Momann, T. 1983. Rapid clorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. Journal of Immunological Methods 65: 55–63.
Nurtjahja-Tjendraputra, E., A.J. Ammit, B.D. Roufogalis, V.H. Tran, and C.C. Duke. 2003. Effective anti-platelet and COX-1 enzyme inhibitors from pungent constituents of ginger. Thrombosis Research 111: 259–265.
Sang, S., J. Hong, H. Wu, J. Liu, C.S. Yang, M.H. Pan, V. Badmaev, and C.T. Ho. 2009. Increased growth inhibitory effects on human cancer cells and anti-inflammatory potency of shogaols from Zingiber offricinale relative to gingerols. Journal of Agricultural and Food Chemistry 57: 10645–10650.
Shino, K., N. Masui, Y. Yamada, M. Asano, and R. Nakamura. 2004. Isolation of antioxidative compounds from Ginger (Zingiber officinale Roscoe) and the effect of heat treatment. Japan Society of Home Economics 55: 375–380.
Wei, Q.-Y., J.-P. Ma, Y.-J. Cai, L. Yang, and Z.-L. Liu. 2005. Cytotoxic and apoptotic activities of diarylheptanoids and gingerol-related compounds from the rhizome of Chinese ginger. Journal of Ethnopharmacology 102: 177–184.
Wu, Q., M.H. Bang, D.Y. Lee, J.G. Cho, L.K. Jeong, S. Shrestha, K.T. Lee, H.G. Chung, E.M. Ahn, and N.I. Baek. 2012. New indoles from the roots of Brassica rapa ssp. campestris. Chemistry of Natural Compounds 48: 281–284.
Acknowledgments
This study was supported by a grant from the Next-Generation Bio-Green 21 Program (No. PJ008020), Rural Development Administration, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, Q., Cho, JG., Yoo, KH. et al. A new phenanthrene derivative and two diarylheptanoids from the roots of Brassica rapa ssp. campestris inhibit the growth of cancer cell lines and LDL-oxidation. Arch. Pharm. Res. 36, 423–429 (2013). https://doi.org/10.1007/s12272-013-0068-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-013-0068-8